10-K: Annual report [Section 13 and 15(d), not S-K Item 405]
Published on February 24, 2026
UNITED STATES
SECURITIES AND EXCHANGE COMMISSION
Washington, D.C. 20549
____________________________________________________________________________
Form 10-K
____________________________________________________________________________
(Mark One)
| ANNUAL REPORT PURSUANT TO SECTION 13 OR 15(d) OF THE SECURITIES EXCHANGE ACT OF 1934 | |||||
For the fiscal year ended December 31 , 2025
Or
| TRANSITION REPORT PURSUANT TO SECTION 13 OR 15(d) OF THE SECURITIES EXCHANGE ACT OF 1934 | |||||
For the transition period from ____________ to______________
Commission File Number: 001-36046

(Exact name of registrant as specified in its charter)
(State or other jurisdiction of
incorporation or organization)
(Address of principal executive offices)
(I.R.S. Employer
Identification No.)
(Zip Code)
Registrant’s telephone number, including area code: (386 ) 462-6800
Securities registered pursuant to Section 12(b) of the Act:
| Title of each class | Trading Symbol(s) | Name of exchange on which registered | ||||||||||||
Securities registered pursuant to Section 12(g) of the Act:
None
(Title of class)
Indicate by check mark if the registrant is a well-known seasoned issuer, as defined in Rule 405 of the Securities Act. Yes ☒ No ☐
Indicate by check mark if the registrant is not required to file reports pursuant to Section 13 or Section 15(d) of the Act. Yes ☐ No ☒
Indicate by check mark whether the registrant (1) has filed all reports required to be filed by Section 13 or 15(d) of the Securities Exchange Act of 1934 during the preceding 12 months (or for such shorter period that the registrant was required to file such reports), and (2) has been subject to such filing requirements for the past 90 days. Yes ☒ No ☐
Indicate by check mark whether the registrant has submitted electronically every Interactive Data File required to be submitted pursuant to Rule 405 of Regulation S-T (§ 232.405 of this chapter) during the preceding 12 months (or for such shorter period that the registrant was required to submit such files). Yes ☒ No ☐
Indicate by check mark whether the registrant is a large accelerated filer, an accelerated filer, a non-accelerated filer, a smaller reporting company, or an emerging growth company. See the definitions of “large accelerated filer,” “accelerated filer”, “smaller reporting company” and “emerging growth company” in Rule 12b-2 of the Exchange Act.
Large accelerated filer ☐
Non-accelerated filer ☐
Smaller reporting company ☐
Emerging growth company ☐
If an emerging growth company, indicate by check mark if the registrant has elected not to use the extended transition period for complying with any new or revised financial accounting standards provided pursuant to Section 13(a) of the Exchange Act. ☐
Indicate by check mark whether the registrant has filed a report on and attestation to its management’s assessment of the effectiveness of its internal control over financial reporting under Section 404(b) of the Sarbanes-Oxley Act (15 U.S.C. 7262(b)) by the registered public accounting firm that prepared or issued its audit report. ☒
If securities are registered pursuant to Section 12(b) of the Act, indicate by check mark whether the financial statements of the registrant included in the filing reflect the correction of an error to previously issued financial statements. ☐
Indicate by check mark whether any of those error corrections are restatements that required a recovery analysis of incentive-based compensation received by any of the registrant’s executive officers during the relevant recovery period pursuant to § 240.10D-1(b). Yes ☐ No ☒
Indicate by check mark whether the registrant is a shell company (as defined in Rule 12b-2 of the Act). Yes ☐ No ☒
As of June 30, 2025, the last day of the registrant’s most recently completed second quarter, the aggregate market value of the voting and non-voting common equity held by non-affiliates of the registrant was approximately $430.7 million based upon the last reported sale price of the common stock on the Nasdaq Capital Market.
The number of shares outstanding of the Registrant’s common stock as of February 20, 2026, was 51,897,682 shares.
DOCUMENTS INCORPORATED BY REFERENCE
Portions of the Registrant’s definitive proxy statement to be filed pursuant to Regulation 14A within 120 days after the end of the Registrant’s fiscal year are incorporated by reference into Part III of this Form 10-K.
| Table of Contents | ||||||||
| Page | ||||||||
i
FORWARD-LOOKING STATEMENTS
From time to time, in reports filed with the U.S. Securities and Exchange Commission (the “SEC”) (including this Annual Report on Form 10-K), in press releases, and in other communications to shareholders or the investment community, Axogen, Inc. (including Axogen, Inc.’s wholly owned subsidiaries, Axogen Corporation, Axogen Processing Corporation, Axogen Europe GmbH, and Axogen Germany GmbH, (the “Company,” “Axogen,” “we,” “our,” or “us”) may provide forward-looking statements, as defined in the Private Securities Litigation Reform Act of 1995, concerning possible or anticipated future results of operations or business developments. These statements are based on management's current expectations or predictions of future conditions, events, or results based on various assumptions and management’s estimates of trends and economic factors in the markets in which we are active, as well as its business plans. Words such as “expects,” “anticipates,” “intends,” “plans,” “believes,” “seeks,” “estimates,” “projects,” “forecasts,” “continue,” “may,” “should,” “will,” “goals,” and variations of such words and similar expressions are intended to identify such forward-looking statements.
The forward-looking statements in this Form 10-K include, but are not limited to the following:
•Our expectations regarding our ability to market Avive+ Soft Tissue Matrix and our expectations that Avive+ Soft Tissue Matrix will, and will continue to be, regulated solely under Section 361 of the Public Health Service Act (“PHS Act”);
•Statements regarding estimates of the total addressable market for our current portfolio, our belief that our total addressable market is comprised of four categories: (i) extremities, (ii) oral maxillofacial and head and neck, (iii) breast reconstruction neurotization, and (iv) urology and statements regarding estimates of the market for our current portfolio in each of these four categories;
•Statements regarding our beliefs that expanding our products into lower extremity surgery, urology, and the surgical treatment of pain could offer us expanded revenue opportunities;
•Our belief that further innovation and product development is needed to maintain our leadership position and provide expansion opportunities;
•Our belief that losing the ability to sell Axoguard products obtained through the Evergen agreements, even though we may be able to develop or obtain replacements, could materially adversely affect our business, results of operations, financial condition, and prospects;
•Our belief that additional opportunities exist to develop or acquire complementary products in peripheral nerve repair as well as opportunities to expand our existing portfolio of products in new applications of peripheral nerve repair;
•Our belief that adequate coverage and reimbursement from third-party payors, including government payors such as Medicare and Medicaid, are important for achieving product acceptance and widespread adoption;
•Our belief that there is a global need for surgical repair of damaged or transected nerves and that a global market exists for our product; and
•Statements regarding our ability to monitor product utilization within accounts and generate improved estimates of our revenue by application.
The forward-looking statements are and will be subject to risks and uncertainties, which may cause actual results to differ materially from those expressed or implied in such forward-looking statements. Forward-looking statements contained in this Form 10-K should be evaluated together with the many uncertainties that affect our business and its market, particularly those discussed in the risk factors and cautionary statements set forth in our filings with the SEC, including as described in “Risk Factors” included in Item 1A of this Form 10-K and “Risk Factor Summary” included in this Form 10-K. Forward-looking statements are not guarantees of future performance, and actual results may differ materially from those projected. The forward-looking statements contained in this report are based on information that is currently available to us and expectations and assumptions that we deem reasonable at the time the statements were made. The forward-looking statements are representative only as of the date they are made and, except as required by applicable law, we assume no responsibility to publicly update or revise any forward-looking statements, whether as a result of new information, future events, changed circumstances or otherwise.
MARKET, INDUSTRY AND OTHER DATA
This Annual Report on Form 10-K contains estimates, projections, and other information concerning our industry, and our business, including data regarding the estimated size of those markets. Information that is based on estimates, forecasts, projections, or similar methodologies is inherently subject to uncertainties and actual events or circumstances may differ materially from events and circumstances reflected in this information. Unless otherwise expressly stated, we obtained this
1
industry, business, market, and other data from reports, research surveys, studies, and similar data prepared by third parties, industry, medical and general publications, government data, and similar sources. In some cases, we do not expressly refer to the sources from which this data is derived. In that regard, when we refer to one or more sources of this type of data in any paragraph, you should assume that other data of this type appearing in the same paragraph is derived from the same sources, unless otherwise expressly stated or the context otherwise requires.
2
PART I
ITEM 1. BUSINESS
General
We are the leading company focused specifically on the science, development, and commercialization of technologies for peripheral nerve regeneration and repair. We are passionate about providing the opportunity to restore nerve function and quality of life for patients with peripheral nerve injuries. We provide innovative, clinically proven, and economically effective repair solutions for surgeons and healthcare providers. Peripheral nerves provide the pathways for both motor and sensory signals throughout the body. Every day, people suffer traumatic injuries or undergo surgical procedures that impact the function of their peripheral nerves. Physical damage to a peripheral nerve or the inability to properly reconnect peripheral nerves can result in the loss of muscle or organ function, the loss of sensory feeling, or the initiation of pain.
Our platform for peripheral nerve repair features a comprehensive portfolio of products, including:
•Avance® (acellular nerve allograft-arwx) an acellular nerve scaffold approved by the United States (“U.S.”) Food and
Drug Administration (“FDA”) on December 3, 2025 for the treatment of adult and pediatric patients aged one month or older with sensory, mixed, and motor peripheral nerve discontinuities (“Avance”) and is expected to be commercially available early in the second quarter of 2026 as a replacement for Avance® Nerve Graft;
•Avance® Nerve Graft, a biologically active off-the-shelf processed human nerve allograft for bridging severed peripheral nerves without the comorbidities associated with a second surgical site (“Avance Nerve Graft” and together with Avance, the “Avance Products”);
•Axoguard Nerve Connector®, a porcine (pig) submucosa extracellular matrix (“ECM”) coaptation aid for tensionless repair of severed peripheral nerves (“Axoguard Nerve Connector”);
•Axoguard Nerve Protector®, a porcine submucosa ECM product used to wrap and protect damaged peripheral nerves and reinforce the nerve reconstruction while minimizing soft tissue attachments (“Axoguard Nerve Protector”);
•Axoguard HA+ Nerve Protector™, a porcine submucosa ECM base layer coated with a proprietary hyaluronate-alginate gel, a next-generation technology designed to enhance nerve gliding and provide short- and long-term protection for peripheral nerve injuries (“Axoguard HA+ Nerve Protector”);
•Axoguard Nerve Cap®, a porcine submucosa ECM product used to protect a peripheral nerve end and separate the nerve from the surrounding environment to reduce the development of symptomatic or painful neuroma (“Axoguard Nerve Cap”); and
•Avive+ Soft Tissue Matrix™, a multi-layer amniotic membrane allograft used to protect and separate tissues in the surgical bed during the critical phase of tissue healing (“Avive+ Soft Tissue Matrix”).
On June 24, 2024, we announced the launch of Avive+ Soft Tissue Matrix. Avive+ Soft Tissue Matrix is processed and distributed in accordance with FDA requirements for Human Cellular and Tissue-based Products (“HCT/P”) under the Code of Federal Regulations (“CFR”) Title 21 (“21 CFR”) Part 1271 regulations and U.S. Public Health Service regulations as a Section 361 human tissue product. Products regulated solely under Section 361 of the PHS Act are a product category under close scrutiny by the FDA for compliance with the regulatory requirements and are potentially subject to regulatory change in the future.
Our portfolio of products is currently available in the U.S., Canada, Germany, the United Kingdom (“U.K.”), Spain and several other countries.
Revenue from the distribution of our Avance Nerve Graft, Axoguard Nerve Connector, Axoguard Nerve Protector, Axoguard HA+ Nerve Protector, Axoguard Nerve Cap and Avive+ Soft Tissue Matrix in the U.S. is the main contributor to our total reported sales and have been the key component of our growth to date.
Nerves can be damaged in several ways. When a nerve is cut due to a traumatic injury or inadvertently during a surgical procedure, functionality of the nerve may be compromised, causing the nerve to no longer carry the signals to and from the brain to the muscles and skin, thereby reducing or eliminating functionality. The loss of function can impact a person’s ability
3
to work and perform daily tasks, to properly be aware and respond to their environment (e.g., heat, cold or other dangers), and could negatively impact their ability to experience and enjoy life.
Nerve damage or transection of the type described above generally requires a surgical repair. Traditionally, the standard has been to either suture the nerve ends together directly without tension or to bridge the gap between the nerve ends with a less important nerve surgically removed from elsewhere in the patient’s own body, referred to as a nerve autograft. More recently, synthetic or collagen conduits have been used for the repair of short gaps. Nerves that are not repaired or heal abnormally may result in a permanent loss of motor and/or sensory function. Additionally, abnormal healing can form a neuroma that may send altered signals to the brain resulting in the sensation of pain. This abnormal section of the nerve can, under certain circumstances, be surgically removed and the nerve can be managed by capping, burying, or surgically repairing the nerve.
In addition, compression on a nerve, blunt force trauma or other physical irritations to a nerve can cause nerve damage that may alter the signal conduction of the nerve, resulting in pain, and may, in some instances, require surgical intervention to address the resulting nerve compression. Finally, when a patient undergoes a mastectomy due to breast cancer or prophylactically due to a genetic predisposition for breast cancer, the nerves are cut to allow the removal of the breast tissue. This can result in a loss of sensation, the potential risk of a symptomatic neuroma, and could negatively impact the patient’s quality of life. When a patient chooses a breast reconstruction after a mastectomy, sensation and quality of life can, in certain cases, be returned through surgical nerve repair.
To improve the options available for the surgical repair and regeneration of peripheral nerves, we have developed and licensed regenerative medicine technologies. Our innovative approach to regenerative medicine has resulted in first-in-class products that we believe are redefining the peripheral nerve repair market. Our products are used by surgeons during surgical interventions to repair a wide variety of physical nerve damage throughout the body, which can range from a simple laceration of a finger to a complex brachial plexus injury (an injury to the network of nerves that control the movement and sensation of the shoulder, arm, and hand) as well as nerve injuries caused by dental, orthopedic, and other surgical procedures.
Peripheral Nerve Regeneration Market Overview
Peripheral nerve injury (“PNI”) through damage or transection is a major source of physical disability impairing the ability to move muscles or to feel normal sensations. Patients suffer traumatic bodily injuries every day that may result in damage or transection to peripheral nerves severe enough to require surgical treatment. We break our total addressable market into four categories: (i) extremities, (ii) oral maxillofacial and head and neck, (iii) breast reconstruction neurotization, and (iv) urology, which we refer to collectively as the total addressable market (“TAM”).
We previously estimated that U.S. PNI has a potential TAM for our current product portfolio and believe it is presently at least $5.6 billion. Estimating the TAM for nerve repair is challenging as there is not a simple data source for the incidence of peripheral nerve issues. This is further complicated by the fact that nerves can be injured through a variety of traumatic and surgical injuries and can impact a patient from head to toe. In addition, we believe nerves are often one of many structures injured in a trauma (e.g., amputation) or in surgery and the incidence of these nerve injuries are often not coded or tracked. Quantifying the procedures involving nerve repair may also be challenging. While selected trauma and surgical procedures are dedicated to the repair of nerves, most of the incidence of nerve repair is a step in a larger trauma or surgical procedure. Current Procedural Terminology (“CPT”) codes exist for surgeons to code for nerve repair; however, we believe the data substantially underestimates the total number of nerves repaired. Physicians are encouraged to document all steps of procedures, but open trauma often involves many surgical steps, and CPT codes may be inclusive of each other or may not be documented or reported in billing records. As a result, we believe CPT coding underrepresents the total number of nerve repairs performed in trauma. Because we believe CPT claims are not fully representative of the true volumes of nerve repair surgery, we follow an “empirical” methodology to estimate the TAM, using published clinical literature and procedure databases to make what we believe are the most objective assumptions.
Extremities
The extremities business is comprised of traumatic nerve injuries and chronic nerve injuries. The traumatic nerve injuries portion of the TAM encompasses traumatic PNI throughout the body, with approximately 95% of injuries affecting upper and lower extremity nerves. We previously estimated the trauma portion of the TAM and presently believe it is at least $2.9 billion annually based upon epidemiological studies regarding the general number of trauma patients, clinical literature review reporting PNI incidence, and physician interviews. We have estimated the portion of these nerve repair procedures due to trauma that would require gap repair, primary repair and/or nerve protection and applied, as we believed was appropriate in each procedure segment, the number of units and average sales price of Avance Products and the average market price for nerve connectors and nerve protectors to determine the probable TAM.
4
Oral Maxillofacial and Head and Neck
We previously estimated the oral maxillofacial portion of the TAM and presently believe it is at least $1.2 billion annually, based upon research indicating that approximately 54,000 PNI occur in the U.S. each year related to third molar surgeries, anesthetic injections, dental implants, orthognathic surgery, and mandibular resection procedures. We have applied the average sales price of the Avance Products, Axoguard Nerve Connector, and Axoguard Nerve Protector that address such PNI to derive the OMF portion of the estimated TAM.
In head and neck, we are focused on addressing nerve injuries in parotidectomy, thyroidectomy and radical neck dissections, which we believe presently represent a significant opportunity with more than 200,000 procedures performed annually, highly concentrated in large academic hospital centers.
Breast
We previously estimated the breast portion of the TAM based on autologous flap reconstructions (i.e. DIEP flaps) and presently believe it is at least $417 million annually. In 2023, we launched Resensation® to implant-based procedures with neurotization of the nipple area complex. We estimate that there is an additional 10 - 15% of the breast reconstructions done with implants that can also be neurotized which adds at least $260 million to increase the estimated TAM to at least $677 million annually. Currently, when a patient undergoes autologous or implant-based breast reconstruction after a mastectomy, the patient receives the shape of a natural breast, but often times experiences little to no return of sensory feeling. In certain cases, sensation can be returned to the breast area with the use of our products through an innovative surgical technique called Resensation. We believe that the ideal breast reconstruction should restore breast size, shape, symmetry, and softness, as well as sensation, without the potential risks and comorbidity associated with autograft. We believe the Resensation technique incorporates a patient’s desire for the opportunity to return sensation to their breasts with a reproducible and efficient surgical approach for reconstructive plastic surgery.
Urology
Prostate cancer is the second most common cancer for men in the U.S. with 1 in 8 men diagnosed over their lifetime. Robotic assisted radical prostatectomy, which involves the surgical removal of the prostate gland, is the most common surgical treatment. Despite its efficacy in cancer control and development of nerve sparing techniques, the procedure often leads to problematic side effects, such as incontinence and erectile dysfunction, due to nerve injury to the cavernous nerve. Our nerve repair portfolio can be used to reconstruct the transected cavernous nerve as well as provide nerve protection in nerve sparing radical prostatectomies. We currently estimate the TAM is at least $754.0 million or 110,000 procedures. To derive the estimated TAM we multiplied the average market sales price of our portfolio products used in the procedure by the number of estimated procedures.
Although distribution and sales of products in the extremities, oral maxillofacial and head and neck, breast and urology portions of the TAM constitute our primary revenue sources today, multiple market expansion opportunities in other clinical applications could offer us expanded revenue opportunities.
Biologics License Application Approval
On December 3, 2025, the FDA approved the BLA for Avance as an acellular nerve scaffold for the treatment of adult and pediatric patients aged one month or older with sensory nerve discontinuity (≤25mm). The FDA granted accelerated approval to Avance for the treatment of sensory nerve discontinuity (>25mm) and mixed and motor nerve discontinuity based on extrapolation of the sensory results from our completed RECON study entitled “A Multicenter, Prospective, Randomized, Patient and Evaluator Blinded Comparative Study of Nerve Cuffs and Avance Nerve Graft Evaluating Recovery Outcomes for the Repair of Nerve Discontinuities.” Continued approval for indications of sensory nerve discontinuities (>25mm) and for mixed and motor nerve discontinuities depends on verification and description of clinical benefit in confirmatory studies. The FDA requires a post-marketing requirement study with a final protocol due February 5, 2026, study completion by December 5, 2030, and a final report due June 5, 2031, as well as progress reports every 180 days, beginning on May 31, 2026. Under this post-marketing requirement, we will conduct a study comparing Avance to nerve autograft.
Commercial availability of Avance is expected early in the second quarter of 2026. In alignment with prior agreements with the FDA, we will transition Avance Products from a human tissue product under Section 361 of the PHS Act (“361 HCT/P”) to a licensed biologic product under Section 351 of the PHS Act. Products that began manufacturing prior to the BLA approval will continue to be distributed as a 361 HCT/P under 21 CFR Part 1271 requirements. Per our agreed upon transition plan with the FDA, this product may remain in the supply chain until its labeled expiration date. Production of Avance under BLA standards began following BLA approval. In the meantime, Avance remains available under the current tissue framework.
5
Our Product Portfolio
Avance Nerve Graft
Avance Nerve Graft is a biologically active nerve implant with more than 17 years of comprehensive clinical evidence and more than 120,000 implants since launch. Avance Nerve Graft is processed nerve allograft intended for the surgical repair of peripheral nerve discontinuities to support regeneration across the defect. It is intended to act as a structural bridge in order to guide and support axonal regeneration across a peripheral nerve gap caused by traumatic injury or surgical intervention. Avance Nerve Graft is decellularized and sterile processed human peripheral nerve tissue. We developed Avance Nerve Graft by following the guiding principle that the human body created the optimal peripheral nerve structure. We, through our licensing efforts and research, developed our Avance Method®, a proprietary method for processing recovered human peripheral nerve tissue in a manner that preserves the essential structure of the ECM while cleansing away cellular and noncellular debris. Avance Nerve Graft provides the natural peripheral nerve structure of a nerve, including the native laminin, to guide the regenerating nerve fibers. The nerve ECM is additionally processed to remove a natural inhibitor to regeneration called chondroitin sulphate proteoglycan.
We believe that Avance Nerve Graft is the first off-the-shelf nerve allograft for bridging nerve transections. Avance Nerve Graft is comprised of bundles of small diameter endoneurial tubes that are held together by an outer sheath called the epineurium. Avance Nerve Graft has been processed to remove cellular and noncellular factors such as cells, fat, blood, and axonal debris, while preserving the three-dimensional laminin lined tubular bioscaffold (i.e., microarchitecture), epineurium, and microvasculature of the peripheral nerve. After processing, Avance Nerve Graft is flexible and pliable, and its epineurium can be sutured in place allowing for a tension-free approximation of the proximal and distal peripheral nerve stumps. During the healing process, the body revascularizes and gradually remodels the graft into the patient’s own tissue while allowing the processed peripheral nerve allograft to physically support axonal regeneration across the peripheral nerve discontinuities. Avance Nerve Graft does not require immunosuppression for use.
With lengths up to 70mm and diameters up to 5mm, Avance Nerve Graft allows surgeons to choose and trim the implant to the correct length for reconstructing the relevant peripheral nerve gap, as well as to match the diameter to the proximal and distal end of the severed peripheral nerve. Avance Nerve Graft is stored frozen and utilizes packaging that maintains the implant in a sterile condition. The packaging is typical for medical products, so the surgical staff is familiar with opening the package for transfer of Avance Nerve Graft into the sterile surgical field. The packaging also provides protection during shipment and storage and a reservoir for the addition of sterile fluid to aid in thawing the product. Avance Nerve Graft thaws in less than ten minutes, and once thawed, it is ready for implantation.
Avance Nerve Graft provides the following key advantages:
•A three-dimensional bioscaffold for bridging a peripheral nerve gap;
•A biologically active nerve therapy with more than 17 years of comprehensive clinical evidence;
•No patient donor-nerve surgery, therefore no comorbidities associated with a secondary surgical site;
•Available in a variety of diameters up to 5mm to meet a range of anatomical needs;
•Available in a variety of lengths up to 70mm to meet a range of gap lengths;
•Decellularized and cleansed ECM;
•Implanted without the need for immunosuppression, remodels into patient’s own tissue;
•Structurally supports the body’s own regeneration process;
•Handles similar to an autograft, and is flexible and pliable;
•Alleviates tension at the repair site;
•Three-year shelf life; and
•Supplied sterile.
6
Axoguard Nerve Connector
Axoguard Nerve Connector is a coaptation aid used to align and connect severed peripheral nerve ends in a tensionless repair. The product is in a tubular shape with an open lumen on each end where the severed peripheral nerve ends are inserted. It is typically used when the gap between the peripheral nerve ends is 5mm or less in length. Axoguard Nerve Connector is made from a processed porcine ECM that allows the body’s natural healing process to repair the peripheral nerve while its tube shape isolates and protects the transected nerves during the healing process. During healing, the patient’s own cells incorporate into the ECM product to remodel and form a tissue similar to the outermost layer of the peripheral nerve (nerve epineurium). Axoguard Nerve Connector is provided sterile, for single use only, and in a variety of sizes to meet the surgeon’s needs.
Axoguard Nerve Connector can be used:
•As an alternative to direct suture repair;
•As a peripheral nerve coaptation; Connector-Assisted Repair®;
•To aid coaptation in direct repair, grafting, or cable grafting repairs; and
•To reinforce the coaptation site.
Axoguard Nerve Connector has the following advantages:
•Processed intact porcine ECM with an open, porous structure that allows for cell infiltration and remodeling;
•Designed as a coaptation aid for tensionless repair of transected or severed peripheral nerves;
•Alleviates tension at the repair site;
•Remodels into the patient’s own tissue;
•Reduces the number of required sutures (versus direct repair with suture);
•Allows surgeon to move sutures away from the repair site which may minimize inflammation and aid nerve regeneration;
•Reduces potential for fascicular mismatch;
•Allows visualization of underlying peripheral nerve ends;
•Available in seven different diameters and two different lengths to address a variety of nerve repair situations;
•Strong and flexible, easy to suture; and
•Stored at room temperature with a minimum of 18-month shelf life for the sizes with six-layers and 24-month shelf life for those with four-layers.
Axoguard Nerve Protector
Axoguard Nerve Protector is a product used to protect and wrap damaged peripheral nerves and reinforce reconstructed nerve gaps while minimizing soft tissue attachments. It is designed to protect and isolate the peripheral nerve during the healing process after surgery by creating a barrier between the nerve tissue and the surrounding tissue bed. The product is delivered in a slit tube format allowing it to be wrapped around peripheral nerve structures. Axoguard Nerve Protector is made from a processed porcine ECM. During healing, the ECM remodels allowing the protector to separate the peripheral nerve from the surrounding tissue. Axoguard Nerve Protector competes against off-the-shelf biomaterials such as reconstituted bovine collagen as well as the use of the patient’s own tissue such as vein and hypothenar fat pad wrapping. Axoguard Nerve Protector is provided sterile, for single use only, and in a variety of sizes to meet the surgeon’s needs.
7
Axoguard Nerve Protector can be used to:
•Separate and protect the nerve from the surrounding tissue during the healing process;
•Minimize risk of soft tissue attachments and entrapment in compressed peripheral nerves;
•Protect peripheral nerves in a traumatized wound bed; and
•Reinforce a coaptation site.
Axoguard Nerve Protector has the following advantages:
•Processed porcine submucosa ECM used to reinforce a coaptation site, wrap a partially severed peripheral nerve or protect peripheral nerve tissue;
•Creates a protective layer that isolates and protects the peripheral nerve in a traumatized wound bed;
•Remodels to form a new soft tissue layer;
•Easily conforms and provides 360-degree wrapping of damaged peripheral nerve tissue;
•Allows the body’s natural healing process to repair the nerve;
•Minimizes the potential for soft tissue attachments and peripheral nerve entrapment by physically isolating the nerve during the healing process;
•Allows peripheral nerve gliding;
•Strong and flexible, plus easy to suture;
•Is available in five different widths and two different lengths to address a variety of peripheral nerve repair situations; and
•Stored at room temperature with a minimum of 24-month shelf life.
Axoguard HA+ Nerve Protector
Axoguard HA+ Nerve Protector is a surgical implant that provides non-constricting protection for peripheral nerves. It is designed to be an interface between the nerve and the surrounding tissue. Axoguard HA+ Nerve Protector is comprised of an ECM and is fully remodeled during the healing process. The lubricant coating on Axoguard HA+ Nerve Protector is composed of sodium hyaluronate and sodium alginate. When hydrated, the lubricant coating reduces friction between the nerve and the surrounding tissue. Axoguard HA+ Nerve Protector is flexible to accommodate movement of the joint and associated tendons and has sufficient mechanical strength to hold sutures. Axoguard HA+ Nerve Protector is provided sterile, for single use only, and in a variety of sizes to meet surgeons’ needs.
Axoguard HA+ Nerve Protector can be used to:
•Separate and protect the nerve from the surrounding tissue during the healing process;
•Minimize risk of soft tissue attachments and entrapment in compressed peripheral nerves;
•Protect peripheral nerves in a traumatized wound bed; and
•Provide tension relief when used in aiding a coaptation.
Axoguard HA+ Nerve Protector has the following advantages:
•Processed porcine submucosa ECM layer is vascularized and remodeled by the patient into new site-specific tissue;
•Double-sided HA+ gel coating to reduce friction and enhance nerve gliding through traumatic tissue beds;
8
•Formulated for optimized handling and flexibility of surgical application—quick hydration, flat sheet configuration and is easy to suture if needed;
•Allows the body’s natural healing process to repair the nerve;
•Minimizes the potential for soft tissue attachments and peripheral nerve entrapment by physically isolating the nerve during the healing process;
•Allows peripheral nerve gliding;
•Is available in five different sizes up to 4cm x 8cm to address a variety of peripheral nerve repair situations; and
•Stored at room temperature with a minimum of 24-month shelf life.
Axoguard Nerve Cap
Axoguard Nerve Cap is a proprietary porcine submucosa ECM product used to protect a peripheral nerve end and separate the nerve from the surrounding environment to reduce the development of symptomatic or painful neuroma.
Nerves are often cut in a variety of surgeries and a nerve that is cut and not reconstructed may form an entangled mass of disorganized nerve and fibrous tissue that could cause debilitating pain called a symptomatic neuroma. Neuromas are a potential cause of pain for those patients who complain of chronic post-surgical pain, including in amputees, which may lead to an inability to use their prosthesis. Despite more than 30 different treatment methods, it is our belief that neuromas continue to be an unresolved problem in microsurgery. We believe the Axoguard Nerve Cap can address these painful neuromas and address nerve pain without the complications of traditional methods, including pharmacotherapy and chemical injections, among others. Axoguard Nerve Cap can be used to reduce the development of symptomatic or painful neuroma formation.
Axoguard Nerve Cap has the following advantages:
•Separates the nerve end from surrounding tissue, neurotrophic factors and mechanical stimulation;
•Reduces painful neuroma formation;
•Allows for anchoring of a nerve end or stump to nearby tissue structure;
•Material gradually remodels into the patient’s own tissue to protect the nerve end;
•Semi-translucence allows for visualization of nerve ends or stumps and easy visualization for suture placement;
•Is available in six different sizes to address a variety of situations; and
•Stored at room temperature with a minimum of 18-month shelf life.
Avive+ Soft Tissue Matrix
Avive+ Soft Tissue Matrix, a multi-layer amniotic membrane allograft used to protect and separate tissues in the surgical bed during the critical phase of tissue repair.
Avive+ Soft Tissue Matrix has the following advantages:
•Keeps adjacent tissues separated and acts as a barrier to soft tissue attachment, which may reduce the potential of nerve tethering and impaired nerve function;
•Present for at least 16 weeks during the critical phase of tissue repair, as confirmed in an animal study model;
•Multi-layered design allows for ideal handling, easy repositioning, and the ability to suture or secure into place; and
•Unique, layered design features epithelial cells on both sides.
9
Tissue Recovery and Processing
Avance Nerve Graft Processing Overview
We developed our Avance Method®, an advanced and proprietary technique to process Avance Nerve Graft from donated human peripheral nerve tissue. Our Avance Method requires special training over several months for each manufacturing associate who processes Avance Nerve Grafts. The processing and manufacturing system for Avance Nerve Graft has required significant capital investment, and we seek to continually improve our manufacturing and quality assurance processes and systems. Our Avance Method is depicted as follows:
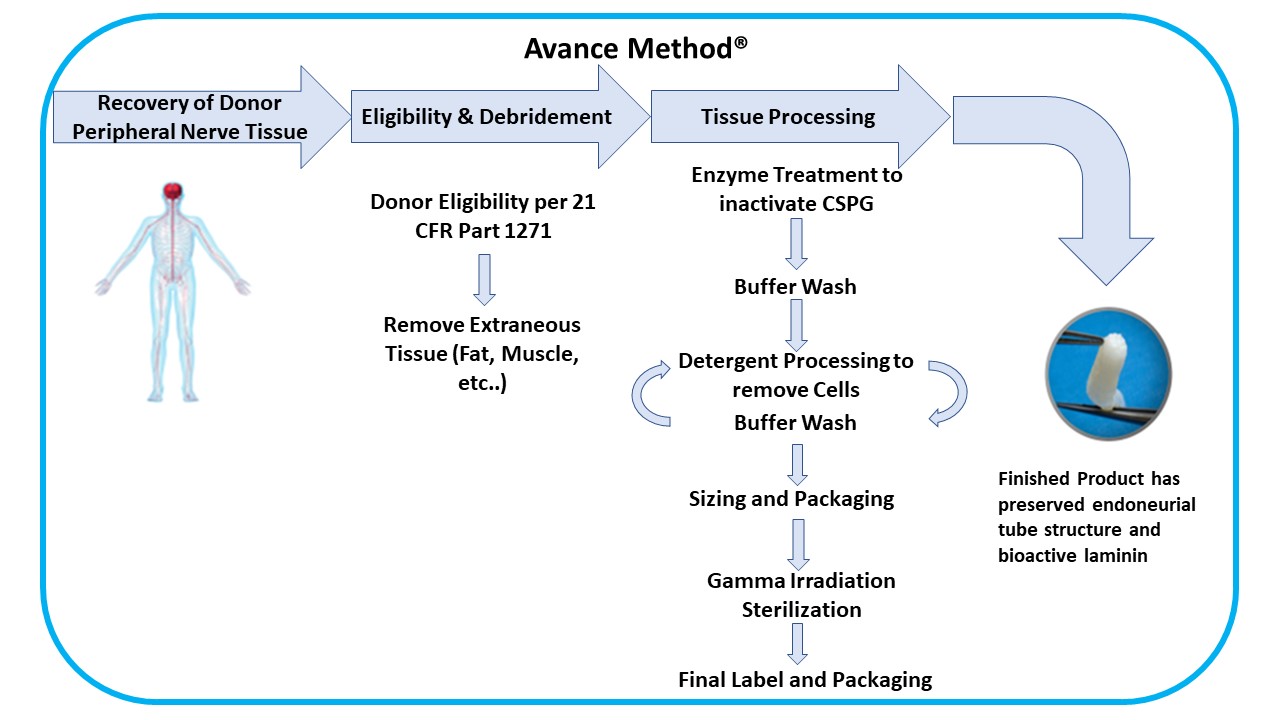
Biologic Tissue Processing
Our Avance Method comprises peripheral nerve tissue recovery/acquisition and testing, donor medical review and release, debridement and other processing steps, packaging, and sterilization to meet or exceed all applicable FDA, state, and international regulations and the American Association of Tissue Banks (“AATB”) standards. Our supply agreements with recovery and acquisition agencies for peripheral nerve tissue and our ability to enter into additional supply contracts, as necessary will provide us with the tissue volumes we require to meet the demand for our Avance implants. As an FDA registered tissue establishment, we use both our own personnel and subcontractors for recovery/acquisition, storage, testing, processing, and sterilization of the donated peripheral nerve tissue and placental tissues. Additionally, we and our subcontractors, have contracted with independent current Good Manufacturing Practice (“cGMP”) and Good Laboratory Practice (“GLP”) compliant laboratories to perform testing for product release. The safety of Avance Nerve Graft is supported by donor screening, process validation, process controls, and validated terminal sterilization methods. The Axogen Quality System has built in redundancies that are meant to ensure product release only after such product meets our quality control and product requirements.
Tissue Recovery and Biologic Processing Facilities
We partner with other FDA registered tissue establishments and AATB accredited recovery agencies or recovery agencies in compliance with FDA, state and international regulations and AATB standards for human tissue recovery. After consent for donation is obtained, donations are screened and tested in detail for safety in compliance with FDA, state and international regulations and AATB standards on communicable disease transmission.
In 2023, we successfully transferred the Avance Nerve Graft tissue processing and packaging to our Axogen Processing Center facility (the “APC Facility”), which is comprised of a 107,000 square foot building on approximately 8.5 acres of land
10
located in Vandalia, Ohio. It is expected that the APC Facility, along with the ability for expansion, will provide processing capabilities that will meet our intended sales growth. On December 3, 2025, the FDA approved the BLA for Avance. The APC Facility is now registered with the FDA as a biologics manufacturing facility.
We obtained certain economic development grants from state and local authorities totaling up to $2.7 million including $1.3 million of cash grants to offset costs to acquire and develop the APC Facility. Certain economic development grants were subject to fixed asset investments and job creation milestones with clawback clauses if we do not meet the minimum requirements for these milestones. In October 2025, we received notification from certain grant authorities that we are expected to satisfy our job creating milestone, assuming we continue to maintain certain headcount and payroll amount thresholds through December 31, 2026. If we are unable to maintain minimum requirements under our grant agreements, we could be obligated to pay back up to approximately $950 thousand as of December 31, 2025 related to these grants. See Part II, Item 8. Financial Statements and Supplementary Data – Notes to Consolidated Financial Statements - Note 15 - Commitments and Contingencies - Service Agreements.
We process and package Avive+ Soft Tissue Matrix using our employees and equipment pursuant to a License and Services Agreement, as amended (the “License and Services Agreement”) with a third party (the “Service Provider”), in Dayton, Ohio at their facility (the “Dayton Facility”). We expect to continue to rely on the Dayton Facility for the processing of Avive+ Soft Tissue Matrix. The Service Provider is a FDA registered tissue establishment and an AATB accredited organization.
The current License and Services Agreement extends through December 31, 2026. Under the License and Services Agreement, we pay the Service Provider a facility fee for clean-room, manufacturing, storage, and office space. The Service Provider also provides services in support of our manufacturing such as routine sterilization of daily supplies, providing disposable supplies and microbial services, and office support. The service fee is based on a per donor batch rate. The Dayton Facility provides a cost effective, quality controlled and licensed facility. Our processing methods and process controls have been developed and validated to ensure product uniformity and quality. Pursuant to the License and Services Agreement, we pay license fees on a monthly basis. See Note 15 - Commitments and Contingencies - Service Agreements in the Notes to the Consolidated Financial Statements in Part II, Item 8 of this Form 10-K.
Tissue Packaging
After processing, the packaging operation for Avance Nerve Graft is performed in a controlled environment at the APC Facility and the packaging operation for Avive+ Soft Tissue Matrix is performed in a controlled environment at the Dayton Facility. Each Avance Nerve Graft and Avive+ Soft Tissue Matrix are visually inspected and organized by size into finished product codes. The tissue implants are then packaged in primary packaging. The outer pouch acts as the primary sterility and moisture barrier.
Tissue Sterilization and Labeling
After being processed and packaged, Avance Nerve Graft and Avive+ Soft Tissue Matrix are then terminally sterilized at a contract sterilization facility. After sterilization, Avance Nerve Graft and Avive+ Soft Tissue Matrix are shipped back to Axogen where the product lots will undergo quality review to ensure the lots meets specifications and then final packaging and labeling. Orders for Avance Nerve Graft and Avive+ Soft Tissue Matrix are placed with our customer care team, and the products are packaged and shipped from our distribution facilities.
Tissue Product Release
We have established quality procedures for review of tissue recovery, relevant donor medical record review, and release to processing that meet or exceed FDA requirements as defined in 21 CFR Part 1271, state regulations, international regulations, and AATB standards. The Axogen Quality System meets the requirements set forth under 21 CFR Part 1271 for HCT/Ps, including Good Tissue Practices (“GTP”) and is compliant with the 21 CFR Part 820 Quality System Regulation (“QSR”). Furthermore, we utilize validated processes for the handling of raw material components, environmental control, processing, packaging, and terminal sterilization. In addition to ongoing monitoring activities for product conformity to specifications and sterility, shipping methods have been validated in accordance with applicable industry standards.
11
Manufacturing of Our Medical Device Classified Products
Manufacturing for the Axoguard Product Line
Axoguard Nerve Connector, Axoguard Nerve Protector, Axoguard HA+ Nerve Protector, and Axoguard Nerve Cap (collectively, the “Axoguard Product Line”) are manufactured by Cook Biotech Incorporated (acquired on January 31, 2024 by RTI Surgical, Inc. and rebranded on December 17, 2024 as Evergen), in West Lafayette, Indiana (“Evergen”), which was established in 1995 to develop and manufacture implants utilizing porcine ECM. We do not expect this acquisition to have a material impact on our relationship with Evergen or on our operations. We decided to expand our portfolio of products and felt that the unique ECM material offered by Evergen provided the combination of properties needed in nerve reconstruction. Evergen’s ECM material is pliable, capable of being sutured, and translucent and allows the patient’s own cells to incorporate into the ECM to remodel and form a tissue similar to the nerve’s epineurium. Evergen has its own source of the raw material for the ECM material and manufactures products in the Axoguard Product Line from such sources.
Axoguard Nerve Connector and Axoguard Nerve Protector
On August 27, 2008, we entered into an agreement with Evergen, as amended on February 24, 2012, October 10, 2014, February 26, 2018, and August 4, 2023 (the “Distribution Agreement”), to distribute its ECM technology in the form of the Surgisis® Nerve Cuff, in the form of a nerve wrap or patch, or any other mutually agreed to configuration. The Surgisis products were rebranded under our Axoguard name and consist of the Axoguard Nerve Connector and Axoguard Nerve Protector. Our distribution rights are worldwide in the field of the peripheral and central nervous system but excluding use of the products in the oral cavity for endodontic and periodontal applications and oral maxillofacial surgery solely as they relate to dental, soft or hard tissue repair, or reconstruction. We believe the exclusion does not limit our identified oral maxillofacial market, but expansion into certain additional oral maxillofacial market areas could be limited to our other products not subject to the Distribution Agreement.
The Distribution Agreement terminates on December 31, 2030. Although the agreement requires certain minimum purchases, through mutual agreement, the parties have not established such minimums and to date have not enforced such provision. The Distribution Agreement also establishes a formula for the transfer cost of the Axoguard Nerve Connector and Axoguard Nerve Protector.
Axoguard Nerve Cap and Axoguard HA+ Nerve Protector
We developed, filed several patent applications, and, on August 8, 2017, obtained FDA 510(k) regulatory clearance for Axoguard Nerve Cap. We developed, filed several patent applications, and, on April 7, 2023, obtained FDA 510(k) regulatory clearance for Axoguard HA+ Nerve Protector and a second 510(k) regulatory clearance expanding the indication for use of Axoguard HA+ Nerve Protector on October 12, 2023. These devices are made with Evergen’s ECM material.
Pursuant to the Nerve End Cap Supply Agreement dated June 27, 2017, as amended on April 6, 2020 and August 4, 2023, (the “Amended Supply Agreement”), Evergen is the exclusive contract manufacturer of the Axoguard Nerve Cap and both parties have provided the other party the necessary licenses to their technologies for operation of the Amended Supply Agreement. The Amended Supply Agreement has a term through December 31, 2030.
Pursuant to the Axoguard HA+ Nerve Protector Supply Agreement dated May 2, 2023 (the “Axoguard HA+ Nerve Protector Supply Agreement”), Evergen is the exclusive contract manufacturer of Axoguard HA+ Nerve Protector and both parties have provided the other party with the necessary licenses to their technologies for operation under the agreement. The Axoguard HA+ Nerve Protector Supply Agreement has a term through June 30, 2030. Consistent with the Axoguard Nerve Connector and Axoguard Nerve Protector products, we are able to sell, subject to regulatory approvals, the Axoguard Nerve Cap and Axoguard HA+ Nerve Protector worldwide in the field of the peripheral and central nervous system, but subject to the same exclusions as Axoguard Nerve Connector and Axoguard Nerve Protector.
Sales and Marketing
Overview
We are focused on developing the peripheral nerve repair and regeneration market, committed to improving awareness of new surgical peripheral nerve repair options and building additional scientific and clinical data to assist surgeons and patients in making informed choices with respect to the repair of peripheral nerve injuries. We believe that there is an opportunity to improve current approaches to peripheral nerve repair and that our approach will solidify our position as a leader in the field of peripheral nerve repair products. The following provides the key elements of our sales and marketing strategy.
12
Increase Awareness of Our Products
Prior to the introduction of our portfolio of peripheral nerve repair products, surgeons had a limited number of options available to surgically repair damaged or transected peripheral nerves. We entered the market to improve the standard of care for nerve injury patients. We intend to increase market penetration and share by increasing awareness of the impact of nerve damage on quality of life and improving the adoption of nerve repair techniques and our products through the continued use of educational conferences and presentations, training for surgical residents, fellows and attending physicians, scientific publications, digital communication, and a knowledgeable and professional sales team. We work to increase awareness and the use of our products within high potential accounts that include hospitals that are level 1 trauma centers, academic affiliated and have a large number of microsurgical trained surgeons and large procedural volumes across our focus clinical specialties. Our customer call points are focused on plastic reconstructive surgeons, orthopedic and plastic hand surgeons who perform surgeries on patients suffering traumatic nerve damage or transection, on oral maxillofacial and head and neck surgeons who repair damaged oral and facial nerves, on plastic reconstructive surgeons who perform breast reconstruction and neurotization and on urologists who perform robotically assisted radical prostatectomies.
Expand Clinical and Scientific Data Regarding the Performance of Our Products
Generating robust clinical and scientific data is a cornerstone of our product development, regulatory, and commercialization strategy. As of December 31, 2025, there have been over 350 peer-reviewed clinical publications related to our products, with some publications including data across multiple product offerings. This expanding body of evidence reflects sustained and growing adoption of our technologies and continued clinical interest in peripheral nerve repair and protection.
Our RANGER® clinical study (defined below in Government Regulations - Clinical Trials), a comprehensive utilization registry of the Avance Nerve Graft, has enrolled more than 2,800 Avance Nerve Graft repairs to date. Enrollment and follow-up in the primary RANGER arms and the MATCH arm (defined below in Government Regulations - Clinical Trials) - evaluating autograft and conduit repair comparators - were completed in December 2023. Analyses from these studies supported our BLA submission. An additional arm of the RANGER study, Sensation-NOW®, tracks neurotization outcomes in autologous DIEP flap breast reconstruction and completed enrollment in November 2025. To date, the RANGER registry has generated 11 peer-reviewed publications and more than 100 scientific conference presentations, contributing to the clinical understanding of nerve repair across sensory, mixed, and motor nerve injuries and advancing the field of nerve repair.
In March 2025, enrollment was completed for COVERED (defined below in Government Regulations - Clinical Trials), a multi-center series evaluating Axoguard HA+ Nerve Protector for nerve protection in first revision cubital tunnel decompression.
We have also supported numerous investigator-initiated studies, case reports, and peer-reviewed publications across clinical areas including breast neurotization, head and neck reconstruction, compressive neuropathies, and the surgical treatment of pain. Additional case series are ongoing or in development for urology, brachial plexus, breast reconstruction neurotization, compression injuries, and the surgical treatment of pain. We continue to collaborate with external investigators and partners on clinically impactful research.
The RECON study (defined below in Government Regulations - Clinical Trials), a pivotal Phase III, multicenter, prospective, randomized, comparative study of hollow tube conduits and Avance Nerve Graft has successfully completed enrollment, follow-up, and analysis. Three peer-reviewed publications from this study have been published.
The REPOSE study pilot phase (defined below in Government Regulations - Clinical Trials), a multicenter, prospective, randomized, and subject-blinded study of Axoguard Nerve Cap as compared to neurectomy alone for the treatment of symptomatic neuroma, has also been published. The comparative phase has completed enrollment and follow-up, providing critical data to guide treatment strategies. Additionally, enrollment in REPOSE XL was completed in January 2025. This study is evaluating the tolerability and feasibility of a large diameter Axoguard Nerve Cap for protecting and preserving terminated nerve ends.
Through continued investment in clinical research, real-world evidence generation, and scientific dissemination, we aim to strengthen the clinical foundation supporting our products, inform regulatory and reimbursement strategies, and advance the standard of care in peripheral nerve repair and protection.
13
Commitment to the Education of Best Practices in Peripheral Nerve Repair
We have established educational conferences and presentations and surgical resident and fellow training that we believe have positioned us as a leader in providing peripheral nerve repair best practices. We have historically provided education on peripheral nerve repair through in-person national programs, including our “Advances and Best Practices in Nerve Repair” as well as local and regional educational events. In 2025, we offered multiple educational programs including virtual and in-person surgeon education programs.
Focused on Developing Deeper Penetration with Our Existing Accounts Through Development of Long-term Users of Our Algorithm in High Potential Accounts Across Our Targeted Surgical Procedures
We provide full sales and distribution services. As of December 31, 2025, we had 138 direct sales professionals in the U.S. Our direct sales force continues to be supplemented by independent sales agencies that represent approximately 8% of our total revenue. We believe that near-term growth can be supported first through expanded productivity of our existing sales force as they go into more depth with existing accounts and then by adding additional accounts. We expect the number of direct sales professionals to increase over time. Additionally, we have successfully utilized a hybrid commercial approach that includes the use of independent agencies in more remote geographies to provide appropriate local support for surgeons, without the travel time required of a direct sales representative.
Our products are available and sold in 18 countries outside the U.S. through a number of independent in-country distributors. We provide support and resources for independent agencies and distributors both within and outside the U.S. We provide our products to hospitals, surgery centers and military hospitals, calling on surgeons, including plastic reconstructive surgeons, orthopedic and plastic hand surgeons, and certain oral and maxillofacial surgeons to review the benefits of our products. While surgeons make the decision to implant our products in appropriate patients, hospitals make the decision to purchase the products from us. In today’s budget constrained environment, hospital committees review new technologies for cost effectiveness as well as quality. We believe that we have been successful in meeting the needs of these hospital committees by demonstrating the cost/benefit of our products and providing a fair value to the hospital.
Increased Patient Access via Improved Coverage and Payment
We actively support our sales and marketing strategy by ensuring a strong reimbursement foundation for our nerve repair products. We work closely with public and private payors so that hospitals and surgeons receive prompt, adequate payment for procedures using the Avance Products and Axoguard products. Dedicated Category I CPT codes for nerve allograft repair (CPT 64912, with add-on +64913) are in place, allowing providers to bill specifically for surgeries using Avance Products. Importantly, the Centers for Medicare & Medicaid Services (CMS) recognizes the complexity and cost of advanced nerve repairs – Avance Nerve Graft procedures carry a “device-intensive” designation and, effective January 1, 2026, CMS created a new Level 3 Ambulatory Payment Classification (APC 5433) for these nerve repair procedures. As a result, Medicare outpatient reimbursement rates have risen significantly, aligning payment with our products’ clinical value. For example, the national average facility payment for a nerve allograft repair procedure in a hospital outpatient setting is now 96% higher than in 2019 (221% higher in ambulatory surgery centers over the same period). This improved payment adequacy reduces economic barriers for providers and supports wider adoption of our nerve repair portfolio.
Concurrently, we are expanding coverage among private insurers through evidence-driven engagement. In partnership with leading surgeons and healthcare providers, our team works to reverse legacy non-coverage policies for Avance Products and Axoguard products. Many commercial payers that once deemed these innovations “investigational” have updated their coverage after reviewing clinical data and real-world outcomes. We have already achieved policy revisions with several regional insurance plans, adding coverage for millions of newly insured lives. Today, approximately 65% of commercially insured patients in the U.S. have coverage for procedures utilizing our products, and we continue to collaborate with payers to close remaining gaps. By ensuring that insurers cover our nerve repair solutions, we enhance patient access and give hospitals confidence that using Avance Products and Axoguard products will be reimbursed, directly supporting our sales efforts.
Expand the Product Pipeline and Applications in Peripheral Nerve Repair
We have developed and continue to develop new and next generation products to support surgeons in their needs for repairing damaged or transected peripheral nerves. We believe additional opportunities exist to develop or acquire complementary products in peripheral nerve repair. In addition, there are opportunities to expand the existing portfolio of products in new applications of peripheral nerve repair in applications such as lower extremity surgery, head and neck surgery, urology, and the surgical treatment of pain.
14
Avance Nerve Graft Performance
We have worked with leading institutions, researchers, and surgeons to support innovation in the field of surgical peripheral nerve repair. We believe our RANGER study is the largest multi-center clinical study conducted in peripheral nerve gap repair with more than 2,800 enrolled repairs. We have completed the RECON study. This study was a Phase III trial to support our BLA for Avance. See Government Regulations - Clinical Trials - Our Clinical Trials below.
International Opportunity for Revenue
We currently focus primarily on the U.S. market, with additional foreign distribution and sales in Canada, Germany, the U.K., Spain and several other countries. The need for the surgical repair of damaged or transected nerves is a global opportunity. Through our revenue outside the U.S., we have demonstrated the capability to take our current peripheral nerve repair surgical portfolio into new geographical markets.
We currently have European Union (“E.U.”) wide registration only for Axoguard Nerve Connector and Axoguard Nerve Protector as approval/registration for Avance Nerve Graft as human tissue is required in each individual country. Avance Nerve Graft was granted marketing authorization in Germany and direct commercial operations began in 2022. As a result of the BLA approval for Avance, some foreign regulators may request updated notifications or documentation regarding the FDA regulatory status of Avance and this may require a Certificate of Foreign Government which we would obtain from the FDA.
Currently, Axoguard Nerve Cap is available in the U.S. and New Zealand. Further Introduction of our portfolio of products into foreign markets is subject to meeting the appropriate regulatory standards of particular countries and any appropriate regional regulation or directive. In addition to regulatory approval, reimbursement approval is necessary to achieve material product adoption in most countries. Avance Nerve Graft has received a recommendation from the National Institute for Health and Care Excellence in the U.K. for digital nerve repair. To date, revenue from international distribution and sales have not been material, there are no material risks associated with foreign operations, and we do not have dependencies as to international revenue. See Item 1A. Risk Factors – Our operations must comply with FDA and other governmental requirements.
Research and Development
We are committed to advancing the field of peripheral nerve repair and offer the most comprehensive portfolio of solutions for peripheral nerve injuries. Our development efforts are focused on expanding the clinical evidence base for nerve repair surgical applications, introducing product line extensions of the Avance Products and Axoguard products, and innovating new technologies and products to address unmet needs in peripheral nerve repair.
In collaboration with leading academic institutions, we actively support the development and evaluation of novel approaches in peripheral nerve injury treatment.
For the fiscal year ended December 31, 2025, we invested approximately $32.9 million in research and development (“R&D”) activities. This investment includes costs associated with product development, clinical research, and the transition of Avance Nerve Graft to a biological product, reflecting our dedication to both scientific advancement and regulatory excellence.
Competition
The medical device and biotechnology industries are characterized by rapidly advancing technologies, intense competition, and a strong emphasis on proprietary products. As such, we cannot predict what products may be offered in the future that may compete with our products. In the peripheral nerve repair market, we compete primarily against all transected and non-transected peripheral nerve repair approaches, including direct suture repair, autograft, and hollow-tube nerve conduits and materials used to wrap and protect damaged peripheral nerve tissue.
Because the requirements of the biomaterials used in peripheral nerve repair can vary based on the severity and location of the damaged nerve, the size and function of the nerve, surgical technique, and patient preference, our peripheral nerve repair products compete against both autograft materials (nerve in the case of a bridging repair and vein or fat in the case of a nerve protection repair), and a limited number of off-the-shelf alternatives for repairing, protecting and nerve termination. Competitive aspects of our products focus on their overall value proposition and suitability for specific applications and can include composition and structure of the material, ease of use, clinical evidence, handling, and price. Our major competitors’ products include off-the-shelf repair options in hollow-tube conduits, coaptation aids and bio-absorbable wraps.
15
We believe any current or future competitors face the following important barriers to market entry as it relates to its peripheral nerve repair products. Our intellectual property (“IP”), and that of our partners, including patents, patents-pending, trade secrets, and unique, internal subject matter expertise, is believed to be an important barrier for our Avance Products and Axoguard products. We have developed knowledge and experience in understanding and meeting FDA regulatory requirements for Avance Nerve Graft, including having made a substantial investment in conducting the pre-clinical and clinical testing necessary to support a submission for an FDA BLA. Additionally, we believe our ability to offer a portfolio of products focused on peripheral nerve repair and the breadth of clinical data associated with the products provides a unique competitive position versus other entities that do not have this breadth of product offering. However, due to our limited resources, our smaller size, and our relatively early stage, we believe we may face competitive challenges from larger entities and market factors that could negatively impact our growth, including competitors’ introduction of new products and competitors’ bundling of products to achieve pricing benefits. See Item 1A. Risk Factors – Technological change and competition for newly developed products could reduce demand for our products. and Risk Factors – Our operating results could be adversely impacted if we are unable to effectively manage and sustain our future growth or scale our operations.
Intellectual Property
Overview
We protect our IP through a combination of patents, trademarks, trade secrets, and copyrights. In addition, we safeguard our trade secrets and other confidential know-how, and carefully protect these and other IP rights when engaging with third parties. For example, we require vendors, contract organizations, consultants, advisors, and employees to execute confidentiality and nondisclosure agreements, and to appropriately protect any information disclosed to them by us so as to preserve confidential and/or trade secret status. We also require consultants, advisors, and employees to assign their rights to any relevant IP arising out of their relationship with us to us.
License Agreements
We have previously entered into license agreements with the University of Florida Research Foundation (the “UFRF”) and the University of Texas at Austin (“UTA”). Under the terms of these license agreements, we hold exclusive worldwide licenses to underlying technologies used by us in our Avance Nerve Graft. The license agreements include the right to certain patents and patents pending in the U.S. and international markets. The effective term of the license agreements extends through the term of the related patents. The patents for which royalty obligations exist under the UFRF license agreement expired in December 2023, and the UTA license agreement expired when the last patents licensed thereunder expired in September 2023.
Patents
As of December 31, 2025, we own 45 issued U.S. patents, more than 50 pending U.S. patent applications (including those for which we have received a notice of allowance) and more than 500 international patents and patent applications with regard to our peripheral nerve products and other related technologies.
In connection with Avance, per Section 351(k)(7) and 351(i)(4) of the PHS Act, from the date of BLA approval, we believe we will have a period of 12 years of exclusivity in the U.S. from commercial competition from biosimilars using Avance Nerve Graft as the reference product. Finally, we have Enforcement Discretion from the FDA regarding continued distribution under controls applicable to HCT/Ps with an agreed transition plan to a BLA. We believe a competitive processed peripheral nerve allograft (non-biosimilar) would need to successfully complete BLA Phase I, II and III clinical studies prior to clinical release, the completion of which we believe would take at least eight years.
Each of Axogen’s other products in the U.S. and abroad, is also protected by multiple patents and local laws providing protection for IP, which provides further barriers to entry for potentially competitive products. Axogen’s Axoguard Nerve Cap is protected by numerous issued Axogen patents in the U.S. and globally. Additional allowed Axogen patent applications, as well as other pending Axogen patent applications that are expected to be issued in the U.S. and abroad, will provide further protection of Axoguard Nerve Cap and thus act as additional obstacles to the commercial introduction of competitive products.
Our Axoguard HA+ Nerve Protector is also the subject of multiple pending Axogen patent applications in the U.S. and abroad. The potential for products competitive with Axogen’s Axoguard line of products, including our Axoguard Nerve Connector and Axoguard Nerve Protector, is further encumbered by the additional IP protections related to their methods of manufacture, as discussed further below in the Trademarks, Trade Secrets and Copyrights section below.
Our policy is to seek patent protection for, or where strategically preferable, maintain as trade secret, the inventions that we consider important to our products and the development of our business. We have sought, and will continue to seek, patent
16
protection for select proprietary technologies and other inventions emanating from our R&D, including with respect to uses, methods, and compositions, in an effort to further fortify our IP in areas of importance to us and our growing product portfolio. In instances that patent protection is not possible, product value to our portfolio can still be derived.
Trademarks, Trade Secrets and Copyrights
We hold a significant portfolio of hundreds of registered trademarks and applied-for trademarks in the U.S. and worldwide. Protection of our trademarks allows us to prevent competitors from, for example, using the same or a confusingly similar company name, or the same or confusingly similar product names within identified classes of goods that could otherwise wrongfully allow such competitors to capitalize on our brand, reputation, and goodwill, and thereby improperly bolster their sales or reputations through, for example, consumer confusion, a false indication of our endorsement, or of a false indication of corporate or contractual relationship with us. We police and enforce our marks.
We possess trade secrets and material know-how in the following general subject matters: nerve and tissue processing, nerve repair, product testing methods, and pre-clinical and clinical expertise. We have registered copyrights for training tools and artistic renderings. Additionally, we entered into the Distribution Agreement and Amended Supply Agreement with Evergen for the Axoguard products. Evergen believes it has know-how and trade secrets with respect to its ECM technology that provides certain additional competitive obstacles to third parties, in addition to those obstacles existing in view of Axogen-owned IP.
Government Regulations
U.S. Government Regulation Overview
Our products are subject to regulation throughout their lifecycle by the FDA, as well as other federal and state regulatory bodies in the U.S. and comparable authorities in other countries. In addition, our Avance Products must comply with the standards of the tissue bank industry’s accrediting organization, the AATB.
We distribute Axoguard Nerve Connector and Axoguard Nerve Protector products for Evergen, and Evergen is responsible for the regulatory compliance of these products. These Axoguard products are regulated as medical devices and subject to pre-market notification requirements under section 510(k) of the Federal Food, Drug, and Cosmetic Act (the “FD&C Act”), 21 CFR Part 820 QSR, and related laws and regulations. Evergen has obtained a 510(k) pre-market clearance for Axoguard Nerve Connector from the FDA for the use of porcine small intestine submucosa for the repair of peripheral nerve transections where gap closure can be achieved by flexion of the extremity. Evergen has also obtained a 510(k) pre-market clearance for Axoguard Nerve Protector for the repair of peripheral nerve damage in which there is no gap or where a gap closure is achieved by flexion of the extremity. We sell the 510(k) cleared devices under the trade names Axoguard Nerve Connector and Axoguard Nerve Protector.
We are the specification developer and authorization holder of the Axoguard Nerve Cap product, which is classified by the FDA as a Class II device. The Axoguard Nerve Cap was cleared for market under 510(k) K163446. It is classified by FDA under 21 CFR § 882.5275 (Nerve Cuff, product code: JXI). Evergen is the contract manufacturer for our Axoguard Nerve Cap product, and we are responsible for the regulatory compliance, distribution, and sale of this product.
We are the specification developer and authorization holder of the Axoguard HA+ Nerve Protector product, which is classified by the FDA as a Class II device. Axoguard HA+ Nerve Protector was cleared for market under 510(k) K223640 on April 7, 2023, and a second 510(k) K231708 regulatory clearance was obtained on October 12, 2023, expanding the indication for use of Axoguard HA+ Nerve Protector. The products are classified by the FDA under 21 CFR § 882.5275 (Nerve Cuff, product code: JXI). Evergen is the contract manufacturer, and we are responsible for the regulatory compliance, distribution, and sale of this product.
Avive+ Soft Tissue Matrix Regulation
We launched Avive+ Soft Tissue Matrix in the second quarter of 2024 after engagement with the FDA through the Tissue Reference Group Rapid Inquiry Program, which resulted in feedback that the product appears to be regulated solely under Section 361 of the PHS Act and the regulations in 21 CFR Part 1271. Products regulated solely under Section 361 of the PHS Act are a product category under close scrutiny by the FDA for compliance with the regulatory requirements and potentially subject to regulatory change in the future. Failure to comply with applicable regulatory requirements could expose us to potential compliance actions by the FDA or state regulators and could risk the commercial availability of the product.
17
FDA — General
FDA regulations govern nearly all the activities that we perform, or that are performed on our behalf, to ensure that medical products distributed domestically or exported internationally are safe and effective for their intended uses. The activities the FDA regulates include the following:
•Product design, development, and manufacture;
•Product safety, testing, labeling, and storage;
•Pre-clinical testing in animals and in the laboratory;
•Clinical investigations in humans;
•Pre-marketing clearance, approval, or licensing;
•Record-keeping and document-retention procedures;
•Advertising and promotion;
•The import and export of products;
•Product marketing, sales, and distribution;
•Post-marketing vigilance, surveillance and medical device reporting, including reporting of deaths, serious injuries, communicable diseases, device malfunctions, or other adverse events; and
•Corrective actions, removals and recalls.
Failure to comply with applicable FDA regulatory requirements may subject us to a variety of administrative or judicially imposed penalties or sanctions and/or prevent us from obtaining or maintaining required approvals, clearances, or licenses to manufacture and market our products. It could also subject us to enforcement actions or sanctions, such as agency refusal to approve pending applications, warning letters, product recalls, product seizures, total or partial suspension of production or distribution of products, injunctions, consent decrees or civil monetary penalties or criminal prosecution.
FDA’s Pre-market Clearance and Approval Requirements - Medical Devices
Unless an exemption applies, each medical device distributed commercially in the U.S. requires either a 510(k) pre-market notification submission or a Pre-Market Approval (“PMA”) Application to the FDA, or other FDA regulatory authorization. Medical devices are classified into one of three classes—Class I, Class II, or Class III—depending on the degree of risk, the level of control necessary to assure the safety and effectiveness of each medical device and how much is known about the type of device. For devices first intended for marketing after May 28, 1976, pre-market review and clearance by the FDA for Class I and II medical devices is accomplished through the 510(k) pre-market notification procedure by finding a device substantially equivalent to a legally marketed Class I or II device, unless the device is exempt. The majority of Class I medical devices are exempt from the 510(k) pre-market notification requirement. Devices deemed by the FDA to pose the greatest risk, such as life-sustaining, life-supporting, or implantable devices for which Class II controls are inadequate to assure safety or effectiveness, and novel devices, including devices deemed not substantially equivalent to a previously cleared 510(k) device, are placed in Class III. Class III devices generally require an approved PMA prior to marketing, unless classified into Class I or Class II through a de novo request.
A PMA must be supported by extensive data, including, but not limited to, technical, pre-clinical, clinical trials, manufacturing and labeling to demonstrate to the FDA’s satisfaction, and the safety and effectiveness of the device.
Investigational New Drug Application for Drugs and Biologics
Federal law requires that a new drug be the subject of an approved marketing application and that a biological product be properly licensed before each is introduced or delivered for introduction into interstate commerce. Because a sponsor often needs to ship an investigational drug or biological product to clinical investigators in many states, it must seek an exemption from that legal requirement. The Investigational New Drug (“IND”) application is the means through which the sponsor obtains this exemption from the FDA. It is additionally the request from a clinical study sponsor to obtain authorization from the FDA to administer an investigational drug or biological product to humans.
18
There are two IND categories: Commercial and Research (non-commercial). The IND application must contain information in three broad areas:
•Animal Pharmacology and Toxicology Studies - Preclinical data to permit an assessment as to whether the product is reasonably safe for initial testing in humans. Also included are any previous experience with the drug in humans (often foreign use).
•Manufacturing Information - Information pertaining to the composition, manufacturer, stability, and controls used for manufacturing the drug substance and the drug product. This information is assessed to ensure that the company can adequately produce and supply consistent batches of the drug.
•Clinical Protocols and Investigator Information - Detailed protocols for proposed clinical studies to assess whether the initial-phase trials will expose subjects to unnecessary risks. Also, information on the qualifications of clinical investigators (professionals (generally physicians) who oversee the administration of the experimental compound) to assess whether they are qualified to fulfill their clinical trial duties. Finally, commitments to obtain informed consent from the research subjects, to obtain review of the study by an independent institutional review board (“IRB”), and to adhere to the investigational new drug regulations.
Once the IND is submitted, the sponsor must wait 30 calendar days before initiating any clinical trials. During this time, the FDA has an opportunity to review the IND for safety to assure that research subjects will not be subjected to unreasonable risk. An IND will automatically become effective 30 days after receipt by the FDA, unless before that time the FDA raises concerns or questions about issues such as the conduct of the trials and or supporting pre-clinical data as outlined in the IND. In that case, the IND sponsor and the FDA must resolve any outstanding FDA concerns or questions before clinical trials can proceed. Therefore, submission of an IND may not result in the FDA allowing clinical trials to commence.
The following regulations apply to the IND application process:
•21 CFR Part 201 Drug Labeling
•21 CFR Part 312 Investigational New Drug Application
•21 CFR Part 314 IND and NDA Applications for FDA Approval to Market a New Drug (New Drug Approval)
•21 CFR Part 316 Orphan Drugs
•21 CFR Part 50 Protection of Human Subjects
•21 CFR Part 54 Financial Disclosure by Clinical Investigators
•21 CFR Part 56 Institutional Review Boards
•21 CFR Part 58 Good Lab Practice for Nonclinical Laboratory Studies
Biological Product License Application Pathway
The BLA is a request for permission to introduce, or deliver for introduction, a biological product into interstate commerce (21 CFR Part 601.2). Form 356h specifies the requirements for a BLA. Biological products require FDA approval of a BLA to be marketed. The application must demonstrate the safety, purity, and potency of the product candidate based on results of pre-clinical studies and clinical trials. A BLA must also contain extensive Chemistry, Manufacturing and Controls (“CMC”) and other manufacturing information, as well as labeling information. The applicant must pass an FDA pre-approval inspection of the manufacturing facility or facilities at which the biological product is produced to assess compliance with the FDA’s current cGMP requirements. Satisfaction of FDA approval requirements for biologics typically takes several years and the actual time required may vary substantially based on the type, complexity, and novelty of the product. We cannot be certain that any BLA approvals for our products will be granted on a timely basis, or at all.
The steps for obtaining FDA approval of a BLA to market a biological product in the U.S. include:
•Completion of pre-clinical laboratory tests, animal studies, and formulation studies under the FDA’s GLP regulations;
•Submission to the FDA of an IND for human clinical testing, which must become effective before human clinical trials may begin and which must include independent IRB approval at each clinical site before the trials may be initiated;
19
•Performance of an adequate and well-controlled clinical trial in accordance with Good Clinical Practices to establish the safety and efficacy of the product for each indication;
•Submission to the FDA of a BLA, which contains detailed information about the CMC for the product, reports of the outcomes and full data sets from the clinical trials, and proposed labeling and packaging for the product. With agreement from the FDA, sponsors can qualify to submit portions of an application as the information becomes available (“rolling submission”) as an alternative to providing all information in a single submission when it is available;
•Satisfactory review of the contents of the BLA by the FDA, including the satisfactory resolution of any questions raised during the review;
•Satisfactory completion of an FDA Advisory Committee review, if applicable;
•Satisfactory completion of an FDA inspection of the manufacturing facility or facilities at which the product is produced to assess compliance with cGMP regulations, to assure that the facilities, methods, and controls are adequate to ensure the product’s identity, strength, quality, and purity; and
•FDA approval of the BLA, including agreement on post-marketing commitments, if applicable.
Avance Nerve Graft Regulatory Classification and Regulatory Pathway
Avance Nerve Graft has been marketed domestically and internationally since 2007. In 2010, the FDA provided us with an enforcement discretion letter, regarding the marketing of Avance so long as we complied with certain terms that focused us on taking the necessary steps to support a BLA submission for the product. The FDA enforcement discretion letter states the FDA will end the period of enforcement discretion upon a final determination of our future BLA submission or if prior to the BLA submission, the FDA finds that we do not meet the conditions for the enforcement discretion terms or are not exercising due diligence in executing the transition plan.
In March of 2021, FDA provided written responses to a Type B Meeting Request that documented a transition plan for after BLA approval. Part of that transition plan included FDA agreement that Avance could be distributed as a tissue product following BLA approval until the depletion of Avance tissue product inventory and transition to distribution of Avance biologic product.
Axogen submitted the BLA for Avance on September 5, 2024. The FDA held the Mid-Cycle meeting with Axogen on March 7, 2025 and the Late-Cycle Meeting on May 21, 2025.
In April 2025, FDA performed a Pre-License Inspection (“PLI”) of the Avance Processing Center in Vandalia, Ohio in support of the Avance BLA regulatory submission. The manufacturing facility is now FDA registered and listed on the FDA Drug Establishment Registration site, and the PLI concluded with a positive outcome.
In December 2025, we received FDA approval of our BLA for Avance. The indications for sensory nerve discontinuities >25mm and for mixed and motor nerve discontinuities were approved under FDA’s Accelerated Approval pathway based on the effect on static two-point discrimination in sensory nerve gaps ≤25mm, which provided empirical evidence to reasonably predict clinical benefit given similarities in pathophysiology and anticipated therapeutic effects. Commercial availability of the licensed Avance product is expected early in the second quarter of 2026. In the meantime, Avance remains available under the current tissue framework.
As part of the FDA approval of Avance under the BLA, Axogen has agreed to a Post-Marketing Requirement (“PMR”). Under this PMR, Axogen will conduct a study comparing Avance to nerve autograft. Axogen will submit the final study protocol by February 5, 2026, complete the trial by December 5, 2030 and submit the final study report within six months of completion, as well as reports every 180 days, beginning May 31, 2026.
We believe that biologic licensing, which typically entails multiple clinical trials and takes many years, would be required for any future competitive peripheral nerve allograft. The FDA provided updated guidance, “Regulatory Considerations for Human Cells, Tissues, and Cellular and Tissue-Based Products: Minimal Manipulation and Homologous Use” in November 2017, which it revised in July 2020. The guidance clarified the FDA’s position that any processing that alters the biological characteristics of peripheral nerve tissue would be considered more than minimal manipulation, and therefore require a BLA prior to marketing.
20
Clinical Trials
Clinical trials are a category of clinical research designed to evaluate and test new interventions, medications, or procedures. Clinical trials are often conducted in four phases. The trials at each phase have a different purpose and help answer different questions.
•Phase I trials test an experimental drug or treatment in a small group of people for the first time. The researchers evaluate the treatment’s safety, determine a safe dosage range, and identify side effects.
•In Phase II trials, the experimental drug or treatment is given to a larger group of people to see if it is effective and to further evaluate its safety.
•In Phase III trials, the experimental study drug or treatment is given to large groups of people. Researchers aim to confirm its effectiveness, monitor side effects, compare it to commonly used treatments, and collect information that will allow the experimental drug or treatment to be used safely.
•Phase IV trials, also known as post-marketing studies, are conducted after a treatment is approved for use by the FDA and provide additional information including the treatment or drug’s risks, benefits, and best use.
Clinical trials are required to support a BLA or PMA and are sometimes required for 510(k) clearance or de novo classification. Clinical trials involve the administration of the investigational product to human subjects under the supervision of qualified investigators. Clinical trials are conducted under strict requirements to ensure the protection of human subjects participating in the trial and under protocols detailing, among other things, the objectives of the study, the parameters to be used in monitoring and safety, and the effectiveness criteria to be evaluated. Clinical trials for biological products require the submission and FDA acceptance of an IND and clinical trials for medical devices require the submission and FDA approval of an Investigational Device Exemption (“IDE”) application unless the device regulations provide for an exemption from the IDE requirement. Clinical trials for significant risk devices may not begin until the IDE is approved by the FDA and the IRB overseeing the particular clinical trial. If the product is considered a non-significant risk device under FDA regulations, the trial must only be approved by an IRB prior to its initiation. A protocol for each clinical trial and any subsequent protocol amendments must be submitted to the FDA as part of the IND or IDE, for significant risk devices. In addition, for these studies, an IRB at each site at which the study is conducted must approve the protocol, subject consent form and any amendments for each site at which the study is conducted. All research subjects must be informed, among other things, about the risks and benefits of the investigational product and provide their informed consent in writing.
Clinical trials under an IND typically are conducted in three sequential phases, but the phases may overlap or be combined. In our case, we believe that the Phase III clinical trial study for Avance Nerve Graft represents the only prospective clinical data that will be required to evaluate safety and effectiveness. Phase III clinical trials usually further evaluate clinical efficacy and test further for safety in an expanded patient population. Phase III clinical trials usually involve comparison with placebo, standard treatments, or other comparators. Usually, multiple well-controlled large Phase III or pivotal clinical trials demonstrating safety and efficacy are required to support a BLA. These trials are intended to establish the overall risk-benefit profile of the product and provide an adequate basis for physician labeling. Clinical testing may not be completed successfully within any specified period, if at all. Furthermore, we or the FDA may suspend or terminate a clinical trial at any time on various grounds, including a finding that the subjects are exposed to an unacceptable health risk, have experienced a serious and unexpected adverse event, or that continued use in an investigational setting may be unethical. Similarly, an IRB can suspend or terminate approval of research, for example, if the research is not being conducted in accordance with the IRB’s requirements or if the research has been associated with unexpected serious harm to patients. Additionally clinical data obtained from the observational study, RANGER, will be provided as supportive safety data and confirmatory data for Avance Nerve Graft.
Our Clinical Trials
We have an active clinical research program to gather data on our product portfolio. We have completed five clinical studies, are performing four ongoing clinical studies, and have plans to initiate further clinical studies. The ongoing studies are:
•“A Multicenter Retrospective Study of Avance Nerve Graft Utilization, Evaluations, and Outcomes in Peripheral Nerve Injury Repair” (“RANGER”) parent protocol and its Addendum 1 arm, “A Matched Autograft and Tube Conduit Case Control Cohort Arm of RANGER” (“MATCH”). Enrollment, follow-up, and analysis has been completed with reporting and close-out activities underway;
•“Breast Neurotization Outcomes for Women: A Registry Study of Recovery Outcomes, Quality of Life and Patient Satisfaction in Post-Mastectomy Autologous Breast Reconstruction” (“Sensation-NOW”). Enrollment is ongoing;
21
•“Nerve Protection Evaluation: Revision Cubital Tunnel Syndrome Decompression” (“COVERED”). Enrollment is ongoing; and
•“Tolerability and Feasibility Pilot Clinical Study of a Large-Diameter Nerve Cap for Protecting and Preserving Terminated Nerve Ends” (“REPOSE-XL”). Enrollment is ongoing.
Our completed studies are “A Multicenter, Prospective and Subject Blinded Comparative Study of Axoguard Nerve Cap and Neurectomy for the Treatment of Symptomatic Neuroma and Prevention of Recurrent End-Neuroma Pain” (“REPOSE”), “A Multicenter, Prospective, Randomized, Patient and Evaluator Blinded Comparative Study of Nerve Cuffs and Avance Nerve Graft Evaluating Recovery Outcomes for the Repair of Nerve Discontinuities” (“RECON”), “A Multicenter, Prospective, Randomized, Comparative Study of Hollow Nerve Conduit and Avance Nerve Graft Evaluation Recovery Outcomes of the Nerve Repair in the Hand” (“CHANGE”) published by Means et al, a pilot study to evaluate the use of Avance Nerve Graft in the reconstruction of nerves following prostatectomy, and “Registry of Avive Soft Tissue Membrane Utilization in Selected Applications of Acute Trauma of the Upper Extremity” (“ASSIST”). As Avive Soft Tissue Membrane is no longer on the market, the registry closed with no planned analysis.
In addition to these clinical research programs, we are developing additional clinical trials in peripheral nerve repair, including nipple areolar complex neurotization, urology, protection, and pain.
Clinical trials are subject to extensive recordkeeping and reporting requirements. Our clinical trials must be conducted under the oversight of an IRB for the relevant clinical trial sites and must comply with FDA regulations, including but not limited to, those relating to Good Clinical Practices. We are also required to obtain the patients’ written, informed consent in a form and substance that complies with both FDA requirements and state and federal privacy and human subject protection regulations. We, the FDA or the IRB may suspend a clinical trial at any time for various reasons, including a belief that the risks to study subjects outweigh the anticipated benefits. Even if a trial is completed, the results of clinical testing may not adequately demonstrate the safety and efficacy of the biological product or device, or may otherwise not be sufficient to obtain FDA approval to market the product in the U.S. Similarly, in the E.U., the clinical study for a medicine product must be authorized by the Competent Authority in each Member State where the clinical trial is to be conducted and must receive a favorable opinion from an ethics committee. See “Risk Factors - Clinical trials can be long and expensive, and results are ultimately uncertain.”, which could jeopardize our ability to obtain regulatory approval and continue to market our Avance Products.
RANGER
The RANGER study is an observational study and is a utilization registry of Avance Nerve Graft. As of December 31, 2025, 11 publications and more than 100 scientific conference presentations have been generated to date from the study. RANGER is designed to allow up to 2,500 subjects. An additional 500 subjects are allowed to be enrolled in Addendum 1, MATCH, and 2,000 enrolled in Addendum 2, Sensation-NOW. Sensation-NOW is a clinical study cohort, currently enrolling, designed to assess breast sensation following reconstruction with or without neurotization. We resumed enrollment in 2021 at select centers after pausing enrollment due to COVID-19 in 2020. The follow-up for the RANGER study is standard of care with a target of up to 36 months post peripheral nerve repair.
The RANGER study database is also utilized to monitor different nerve repair techniques. As part of this, we utilize the database to support additional regulatory submissions for the Axoguard products.
We have worked with leading institutions, researchers, and surgeons to support innovation in the field of surgical peripheral nerve repair. We believe that RANGER is currently the largest multi-center observational clinical study conducted in peripheral nerve gap repair. Various reviewers of the RANGER study have found Avance Nerve Graft nerve repairs resulted in meaningful motor and sensory recovery and reduced pain following neuroma excision and reconstruction with no safety concerns identified.
RECON
The RECON study is a prospective, randomized, controlled, patient and evaluator blinded, comparative study of Avance Nerve Graft and Collagen Nerve Cuffs (manufactured conduits) in the repair of peripheral nerve transections in digital nerves with gaps of 5 to 25mm. The study is designed to assess the outcomes of peripheral nerve repair in approximately 170 subjects in up to 20 centers. Subjects were intraoperatively randomized in a 1:1 ratio after stratification by length of the nerve injury by gap length into short gap (5-14mm) and long gap (15-25mm) categories. The primary objective of the study is to evaluate the safety and efficacy of Avance Nerve Graft for non-inferiority and if met, superiority, of static two-point discrimination, a measure of sensory function, at 12 months as compared to nerve cuffs. Given the pooled standard deviation assumptions and a
22
non-inferiority margin of 2mm, approximately 88 patients per treatment group are required to assess non-inferiority with at least 83% power. In addition to non-inferiority, a minimum treatment effect is required to be demonstrated. Based on an agreement with the FDA in the original protocol and an independent statistical analysis of the pooled standard deviation, the number of subjects was increased to 220 in up to 25 centers. Subjects were followed over the course of 12 months (based on the agreed-upon protocol, subjects have up to an additional three months to complete trial requirements) to assess safety and efficacy outcomes with assessments performed at various defined intervals up to 12 months. The study completed subject enrollment in July 2020. Subject follow-up was completed in August 2021 with topline study data read-out completed during the second quarter of 2022. Topline results showed that this pivotal study met its primary endpoint for the return of nerve function as measured by static two-point discrimination. It also demonstrated that the safety profile was consistent with previously published data. RECON results demonstrated statistical superiority for return of sensory function, as measured by static two-point discrimination, as compared to conduits in gaps greater than 12mm (p-value <0.05). Avance demonstrated statistical superiority for time to recover of static two-point discrimination over conduits in nerve gaps greater than 10mm (p-value <0.05). The data in this study supported our BLA approval and was defined by the FDA as an adequate and well-controlled study.
REPOSE
We are conducting a multicenter, prospective, randomized, and subject blinded study of Axoguard Nerve Cap as compared to neurectomy for the treatment of systematic neuroma. REPOSE is a two-phase study comparing standard neurectomy to Axoguard Nerve Cap, which leverages our chambered technology to aid in the management of symptomatic neuromas. The first phase, a non-randomized pilot, has completed enrollment and one-year follow-up. The second phase, a prospective, randomized controlled study, completed enrollment in 2022. Overall enrollment is designed to target 101 subjects with 15 in the first pilot phase followed by up to 86 in the randomized, comparative phase. The study assessed pain scores, quality of life, neuroma recurrence, and health outcomes over a 12-month follow-up period. Subject follow-up was completed in the third quarter of 2023 with topline analysis reported in January 2024.
REPOSE XL
REPOSE-XL is a prospective, multi-center clinical pilot study evaluating the tolerability and feasibility of the Axoguard Large-Diameter Nerve Cap (sizes 5-7mm) for protecting and preserving terminated nerve endings after limb trauma or amputation when immediate attention to the nerve injuries is not possible. Enrollment in REPOSE-XL started in 2022 and is underway.
COVERED
COVERED is a prospective, multi-center clinical case series evaluating Axoguard HA+ Nerve Protector in first revision cubital tunnel decompression. Enrollment in COVERED started in the fourth quarter of 2023.
Post-Market Regulatory Requirements
There are numerous regulatory requirements that apply after a product is cleared or approved. For medical devices, these include, but are not limited to the FDA’s regulations for device labeling (21 CFR Part 801), medical device reporting (21 CFR Part 803), reporting of corrections and removals (21 CFR Part 806), establishment of registration and device listing requirements (21 CFR Part 807), and compliance with the QSR per 21 CFR Part 820. Distribution of medical devices is also subject to license/registration requirements in some states. For tissue and biological products, the regulatory requirements include: the FDA’s registration and listing requirements, donor eligibility requirements and compliance with GTP in 21 CFR Part 1271 for human tissue products, compliance with the FDA’s cGMP in 21 CFR Parts 210, 211, and 600 for licensed biological products, and post-market BLA requirements (21 CFR Part 601), including The Drug Supply Chain Security Act, as well as any post marketing requirements required by FDA at the time of approval, such as post approval studies. Among other things, these regulations require manufacturers, including third party manufacturers to:
•Follow stringent design, testing, control, documentation, and other quality assurance procedures during all aspects of the manufacturing process;
•Comply with labeling regulations and FDA prohibitions against the false or misleading promotion or the promotion of products for uncleared, unapproved or off-label uses, or indications;
•Comply with requirements to obtain clearance or approval for certain changes affecting the product, including changes to the product’s manufacturing, labeling, or intended use;
23
•Report to the FDA certain adverse events, adverse reactions, and deviations;
•Comply with post-approval restrictions or conditions, including post-approval study commitments and post-market safety and annual reporting requirements;
•Follow post-market surveillance regulations that may apply when necessary to protect the public health or to provide additional safety and effectiveness data for the device; and
•Follow requirements to issue notices of correction or removal, or conduct market withdrawals, or recalls where quality or other issues arise.
Safety Reporting and Other Periodic Reporting
The Medical Device Reporting regulation 21 CFR Part 803 contains mandatory requirements for manufacturers, importers, and device user facilities to report certain device-related adverse events and product problems to the FDA.
In addition to the FDA, the advertising and promotion of medical products are also regulated by the Federal Trade Commission and in some instances by state regulatory and enforcement authorities. Recently, some promotional activities for FDA-regulated products have been the subject of enforcement action brought under healthcare reimbursement laws and consumer protection statutes. In addition, under the Federal Lanham Act and similar state laws, competitors, and others can initiate litigation relating to advertising claims.
Facilities Listing and Registrations
All of our facilities are properly registered with the FDA as drug, tissue or medical device establishments. The FDA has broad post-market and regulatory enforcement powers. We are subject to unannounced inspections by the FDA to determine compliance with the GTP, cGMP, and other regulations, and these inspections may also include suppliers’ manufacturing facilities.
Failure by us or our suppliers to comply with applicable regulatory requirements can result in enforcement action by the FDA or other federal or state authorities, which may include any of the following sanctions, among others:
•Warning letters, fines, injunctions, consent decrees and civil penalties;
•Customer notifications, repair, replacement, refunds, recall or seizure of our products;
•Operating restrictions, partial suspension, or total shutdown of production;
•Suspension or termination of our clinical trials;
•Refusing our 510(k), de novo classification request, PMA or BLA for new products, new intended uses, or modifications to existing products;
•Withdrawing or suspending pre-market approvals that have already been granted; and
•Criminal prosecution.
Education Grants, U.S. Anti-kickback, False Claims and Other Healthcare Fraud and Abuse Laws
Educational Grants
A medical product manufacturer may provide financial or in-kind support, including support by way of grants, to third parties for the purpose of conducting medical educational activities. If these supported activities are considered by the FDA to be independent of the manufacturer, then the activities fall outside the FDA restrictions on promotion to which the manufacturer is subject.
We seek to ensure that the educational activities we support through our grants program are in accordance with the appropriate criteria for independent educational activities. However, we cannot provide assurance that the FDA or other government authorities would view the programs supported as being independent.
24
Fraud, Abuse and False Claims
We are directly and indirectly subject to various federal and state laws pertaining to healthcare fraud and abuse, including anti-kickback false claims, and provider payment transparency laws, that govern our relationships with healthcare providers and facilities, distributors, third-party payors, charitable organizations, disease state associations, patients, and customers. In particular, the federal Anti-Kickback Statute (the “Anti-Kickback Statute”) prohibits persons from knowingly and willfully soliciting, offering, receiving, or providing remuneration, directly or indirectly, in exchange for or to induce either the referral of an individual, or the furnishing, arranging for or recommending a good or service for which payment may be made in whole or part under federal healthcare programs, such as the Medicare and Medicaid programs. In implementing the statute, the Office of Inspector General of the U.S. Department of Health and Human Services (“OIG”) has issued a series of regulations, known as “safe harbors.” These safe harbors set forth provisions that, if all their applicable requirements are met, will assure healthcare providers and other parties that they will not be prosecuted under the Anti-Kickback Statute for activities that fit within a safe harbor. Failure to meet all of the requirements of a particular applicable statutory exception or regulatory safe harbor does not make the conduct per se illegal under the Anti-Kickback Statute. Instead, the legality of the arrangement will be evaluated on a case-by-case basis based on a cumulative review of all of its facts and circumstances. Our practices may not in all cases meet all of the criteria for protection under a statutory exception or regulatory safe harbor.
The federal False Claims Act (“FCA”) imposes civil liability on any person or entity that submits, or causes the submission of, a false or fraudulent claim to the U.S. government. Damages under the FCA can be significant and consist of the imposition of fines and penalties. The FCA also allows a private individual or entity with knowledge of past or present fraud against the federal government to sue on behalf of the government to recover the civil penalties and treble damages. The U.S. Department of Justice (“DOJ”) has previously alleged that the marketing and promotional practices of pharmaceutical and medical device manufacturers including the off-label promotion of products or the payment of prohibited kickbacks to doctors violated the FCA resulting in the submission of improper claims to federal and state healthcare entitlement programs such as Medicaid.
Violations of federal and state healthcare laws, including but not limited to the Anti-Kickback Statute and FCA, may result in significant civil, criminal and/or administrative penalties, including but not limited to damages, fines, disgorgement, exclusion from participation in government programs, such as Medicare and Medicaid, injunctions, imprisonment, private “qui tam” actions brought by individual whistleblowers in the name of the government, additional oversight and reporting obligations, and the curtailment or restructuring of operations.
AdvaMed is one of the primary voluntary U.S. trade associations for medical device manufacturers. PhRMA is another trade association focused on the pharmaceutical industry. These associations have established guidelines and protocols for medical device and pharmaceutical manufacturers, respectively, in their relationships with healthcare professionals on matters, including research and development, product training and education, grants and charitable contributions, support of third-party educational conferences, and consulting arrangements. Adoption of the AdvaMed or PhRMA Codes by a medical device manufacturer is voluntary, and while the OIG and other federal and state healthcare regulatory agencies encourage its adoption, they do not view adoption of these codes as proof of compliance with applicable laws. Key to the underlying principles of the AdvaMed and PhRMA Codes is the need to focus the relationships between manufacturers and healthcare professionals on matters of training, education and scientific research, and limit payments between manufacturers and healthcare professionals to fair market value for legitimate services provided and payment of modest meal, travel, and other expenses for a healthcare professional under limited circumstances. We have incorporated these principles into our relationships with healthcare professionals under our consulting agreements, payment of travel and lodging expenses, research and educational grant procedures and sponsorship of third-party conferences. In addition, we have conducted and will continue to conduct training sessions on these principles. Finally, the Sunshine Act, as defined below, imposes additional reporting and disclosure requirements on us for any “transfer of value” made or distributed to physicians and teaching hospitals, as well as reporting of certain physician ownership interests. We cannot provide any assurance that regulatory, or enforcement authorities will view our relationships with physicians or policies as being in compliance with applicable regulations and laws.
Regulation Outside of the U.S.
Distribution and sales of medical products outside of the U.S. are subject to foreign governmental regulations that vary substantially from country to country.
There are restrictions under U.S. law on the export of medical devices and biological products that cannot be legally distributed in the U.S. The FDA has set forth certain requirements for the export of devices outside of the U.S. depending on the class of device and its FDA approval. We currently believe we comply with applicable regulations when exporting our products and we intend to continue such compliance in the event there are any regulatory changes regarding its products in the U.S.
25
The European Medicines Agency (“EMA” is the decentralized body of the E.U., located in Amsterdam in the Netherlands. It is responsible for the scientific evaluation, supervision, and safety monitoring of medicines for human and veterinary use in the E.U. The EMA serves the E.U. and three countries from the European Economic Area—Iceland, Norway, and Liechtenstein. The E.U. has adopted numerous directives, regulations, and promulgated voluntary standards regulating the design, manufacture and labeling of, and clinical trials and adverse event reporting for medicinal products including medical devices. Devices that comply with the requirements of a relevant regulation or directive will be entitled to bear CE marking, indicating that the device conforms to the essential requirements of the applicable regulation and directives and can be commercially distributed throughout the member states of the E.U. and other countries that comply. The method for assessing conformity varies depending on the type and class of the device, but normally involves an assessment by the manufacturer and a third-party assessment by a notified body, an independent and neutral institution appointed by a country to conduct the conformity assessment. This third-party assessment may consist of an audit of the manufacturer’s quality system and specific testing of the manufacturer’s device. Such an assessment is required for a manufacturer to commercially distribute the product throughout these countries. In the second quarter of 2014, Axogen’s Quality System became accredited to International Organization for Standardization (“ISO”) 13485 for Receipt, Handling, Storage and Distribution of Axoguard Nerve Connector and Axoguard Nerve Protector. We intend to maintain this accreditation on an ongoing basis.
Evergen is responsible for all regulatory filings for the Axoguard Nerve Connector and Axoguard Nerve Protector products, including international registrations. We provide the countries for Evergen to register with, and Evergen prepares and submits the product filing documentation to the Ministry of Health (“MOH”) for the country. Each country or region has its own regulations, and the documentation required for submission varies. It typically takes less than nine months from the initiation of the project to obtain clearance in a given country or region. To date, the Axoguard Nerve Connector and Axoguard Nerve Protector product lines were registered in May 2013 in Canada for distribution and in April 2013 the product lines were awarded the CE Mark allowing distribution into the E.U. and other countries that accept the CE Mark. Evergen received the renewal of the CE Mark for Axoguard Nerve Connector and Axoguard Nerve Protector in May 2021.
In addition, the new European Medical Device Regulation 2017/745 (“E.U. MDR”) passed in the European Parliament on April 5, 2017, and went into effect on May 25, 2017. The E.U. MDR is an extensive reform of the rules governing the medical device industry in Europe. Under this regulation, manufacturers had through May 2021 to comply with a broad set of new rules for almost every kind of medical device. The E.U. MDR requires changes in the clinical evidence required for medical devices, post-market clinical follow-up evidence, annual reporting of safety information for Class IIb and Class III products, and bi-annual reporting for Class IIa products, Unique Device Identification (“UDI”) for all products, submission of core data elements to a European UDI database prior to placement of a device on the market, reclassification of medical devices, and multiple other labeling changes.
While nine years have passed since the adoption of E.U. MDR, the E.U. MDR’s transitional provisions were amended in 2023 to give manufacturers and notified bodies more time to conduct the necessary conformity assessment procedures and to avert shortages of devices needed for the E.U. healthcare systems. European Regulation 2023/607 extended the transitional period according to the risk class of the legacy device until December 2027 or December 2028 if certain requirements are fulfilled (i.e., agreement with a notified body for conformity assessment in place, or a competent authority has granted a derogation from the conformity assessment procedure). European Regulation 2022/112 extended the transitional period for in vitro medical devices according to the risk class of the legacy device until May 2025 for devices already certified by a notified body under the Directive and class D devices, until May 2026 for class C devices and until May 2027 for class B and A sterile devices. Overall, medical device companies can continue to expect longer lead times to obtain product conformity assessments and registrations (i.e., CE Mark Certification) in the E.U. and a substantially costlier pathway to compliance in the E.U.
Evergen is responsible for registering and attaining MDR conformity for Axoguard Nerve Connector and Axoguard Nerve Protector in the E.U. As distributor of these two products in the E.U., we are not yet able to fully determine the costs of complying with these regulations, how the E.U. will continue interpreting and enforcing them, what the timelines for approvals of products will be and the overall effect of the E.U. MDR on the marketplace. Given the significant additional pre-market and post-market requirements imposed by the E.U. MDR, the overall impact of these new rules could have a material, adverse effect on our international revenue and expenses.
The U.K. left the E.U. in January 2020. We register our human tissue products in each individual E.U. country and our distributor in the U.K. has import authority for our human tissue product. It is expected that licensed U.K. establishments that import or export tissues or cells will need written agreements with the relevant E.U. licensed establishments to continue importing and exporting with the E.U. As we ship directly to the U.K. from the U.S., we did not experience and do not expect delays in shipment of human tissue products into the U.K.. Further, the RANGER clinical trial being performed at select hospitals in the U.K. was not affected by the U.K.’s departure from the E.U. (“Brexit”) as long as the products continue to come directly from the U.S. Beginning in January 2021, new changes became effective as the transition period for Brexit ended.
26
Specifically, all medical devices placed into the U.K. market had to be registered, subject to applicable grace periods, with the Medicines and Healthcare products Regulatory Agency (“MHRA”), will need to appoint a U.K. Responsible Person, and comply with additional product marking and conformity assessment requirements. Medical devices must be registered with the MHRA if they are being placed in the U.K. market after May 1, 2021. Evergen is responsible for appointing the U.K. Responsible Person and registering Axoguard Nerve Connector and Axoguard Nerve Protector in the U.K.
Tissue Products Are Not Currently Regulated Under the CE Mark
We are responsible for all regulatory filings for Avance Products. To obtain international approvals, we prepare the product filing documentation and submit this documentation to the MOH for a country.
Although some standards of harmonization exist, each country in which we conduct business has its own specific regulatory requirements, which are dynamic in nature and continually changing. We procure and process our tissue for the Avance Products in the U.S. and market the Avance Nerve Graft in Canada, the U.K., and certain other countries under compliance with the individual country regulations. We conduct a regulatory review at the time of submission of the product dossier. This involves reviewing the appropriate MOH regulations, discussion with in-country distributors and use of consultants. It typically takes less than nine months from the initiation of the product to develop a product dossier (specific for that country), submission of the documentation and MOH review of the product filing. While we believe that we are in compliance with all existing pertinent international and domestic laws and regulations, there can be no assurance that changes in governmental administrations and regulations will not negatively impact our operations. The FDA and international regulatory bodies conduct periodic compliance inspections of our U.S. processing facilities. Axogen’s processing and distribution locations are properly registered with CBER as tissue establishments. In 2023, AATB re-accredited Axogen for compliance to the AATB standards for tissue banking for all our facilities. Additionally, our facilities are appropriately licensed in the states of Florida, New York, California, Maryland, Delaware, Oregon, and Illinois as tissue establishments. We believe that worldwide regulation of tissue products is likely to intensify as the international regulatory community focuses on the growing demand for these implant products and the attendant safety and efficacy issues of recipients. Changes in governing laws and regulations could have a material adverse effect on our financial condition and results of operations. Our management further believes that it can help to mitigate this exposure by continuing to work closely with government and industry regulators.
Environmental
As a biotech company of our size, we believe our impact on the environment is modest. However, we are continuously evaluating how we can be the best possible stewards of the environment, and follow local, state, and federal environmental regulations. We are taking steps in our operations and facilities to positively impact the environment wherever possible.
Our products, as well as the chemicals used in processing these products, are handled and disposed of in accordance with country-specific, federal, state, and local environmental regulations. Since 2007, we have used outside third parties to perform all biohazard waste disposal.
We contract with independent, third parties to perform sterilization of our allografts. Because of the engagement of a third party to perform irradiation services, the requirements for compliance with radiation hazardous waste do not apply, and therefore we do not anticipate that this engagement will have any material adverse effect upon our capital expenditures, results of operations or financial condition. However, we are responsible for assuring that the service is performed in accordance with applicable regulations. Although we believe we are in compliance with all applicable environmental regulations, the failure to fully comply with any such regulations could result in the imposition of penalties, fines or sanctions that could have a material adverse effect on our business.
Human Capital
As of December 31, 2025, we had 622 employees, substantially all of whom were full-time. Of these employees, 284 work in operations, 196 work in sales and marketing, 120 work in corporate, and 22 work in research and development. As of December 31, 2025, we have not had a work stoppage, and no employees are represented by a labor union.
We believe in creating and maintaining a culture that encourages and rewards honesty, openness, accountability and passionate debate among our employees. We are committed to fostering a collaborative, respectful and high performing culture. Our corporate values guide us to lead by example, think like a customer, encourage innovation and risk-taking, embrace and cultivate collaboration and teamwork, and never forget that our business purpose is to restore health and improve quality of life by making restoration of peripheral nerve function an expected standard of care. Our Equal Employment Policy includes specific training on preventing discrimination and harassment. We are committed to advertising our opportunities on each
27
state’s job boards in order to reach an increasingly diverse population of candidates, and we conduct routine audits of our existing job postings, advertisements and candidate communications for gender coding, and update any gender specific language to gender neutral language. Additionally, we have a policy that supports employees who are veterans that participate in Honors Guards, who are selected from partnerships with veteran organizations and participating companies and attend, by invitation, military funerals. Further, some of our recruitment efforts are to engage with the next generation of scientists and engineers through targeted awareness and internship programs. We work with Women in Life Sciences, Society for Asian Scientists and Engineers, Society of Women Engineers, and BioFlorida to educate students and professionals about career opportunities available at our Company.
We strive to offer benefits plans that are viewed as attractive and beneficial to employees. In 2025, we maintained our offerings of multiple medical coverage plans that include a consumer driven health plan (“CDHP”) with a health savings account and the option of a limited flexible spending account for qualifying dental and vision expenses for those employees who select the CDHP. We continue to offer two options for dental (high or low preferred provider organization) plans and options for employees to select from two different vision plan providers, enabling employees to select the option that best fits their needs and includes their preferred physicians. The short-term disability offering pays 100% of the employee’s bi-weekly earnings for the first eight weeks of disability and 66.67% for the remaining disability period, up to 12 weeks. Axogen also offers supplemental benefits in the form of accidental insurance and critical illness insurance designed to help provide financial protection against expenses associated with accidents or illness not covered by medical insurance.
Employee safety is critical to our operations, and we follow Occupational Safety and Health Administration 29 CFR 1910, and use a series of company-wide policies, trainings, and procedures to protect all employees’ health and safety. We utilize an Environmental Health and Safety committee that meets monthly to analyze potential issues, review any incident data, and implement necessary process or procedural changes that can minimize the work-related injuries and occupational exposure to chemicals, biohazards, or illnesses, and eliminate any potential from serious injuries and fatalities.
The Compensation Committee of our Board of Directors (the “Board”) has oversight over human capital management.
Available Information
Our website address is http://www.axogeninc.com. We have included our website address as an inactive textual reference only. We make available, free of charge through our website, our annual reports on Form 10-K, our quarterly reports on Form 10-Q, our current reports on Form 8-K and amendments to those reports filed or furnished pursuant to Section 13(a) or 15(d) of the Exchange Act as soon as reasonably practicable after we electronically file, or furnish such material to the SEC. We also similarly make available, free of charge on our website, the reports filed with the SEC by our executive officers, directors and 10% shareholders pursuant to Section 16 under the Exchange Act as soon as reasonably practicable after copies of those filings are provided to us by those persons. Reference to our website, or any other website, does not constitute incorporation by reference of the information contained on the site and should not be considered part of this Annual Report on Form 10-K.
RISK FACTOR SUMMARY
Below is a summary of the principal risk factors that may affect our business, financial condition and results of operations. This summary does not address all of the risks that we face. Further discussion of the risks summarized in this risk factor summary, and other risks that we face, can be found below under the heading “Risk Factors” and should be carefully considered, together with other information in this Form 10-K and our other filings with the SEC before making an investment decision regarding our common stock.
Risks Related to Our Business and Strategy
•Approximately 60% of our total revenues are from sales of Avance Products and any adverse decision from the FDA would negatively impact our operations and financial condition.
•Portions of the FDA-approved indication for Avance were granted under the FDA’s accelerated approval program, and continued approval for these indications is contingent upon our timely completion of required post-marketing confirmatory studies.
•Our revenue growth depends on our ability to increase distribution and sales to existing customers and develop new customers, domestically and internationally, and there can be no assurance that these efforts will result in significant increases in sales.
•We are highly dependent on the continued availability of our facilities and could be harmed if we continue to experience operating challenges with our APC Facility or if any of our facilities are unavailable for any prolonged period of time.
28
•If our estimates of the TAM for peripheral nerve repair and reconstruction are inaccurate, our growth prospects and financial performance could be materially adversely affected.
•Our revenue depends upon prompt and adequate reimbursement from public and private insurers and national health systems.
•Our operating results could be adversely impacted if we are unable to effectively manage and sustain our future growth or scale our operations.
•Our success will be dependent on continued acceptance of our products by the medical community.
•If we do not manage product inventory in an effective and efficient manner, it could adversely affect profitability.
•There may be significant fluctuations in our operating results.
•We have a history of net losses and have not consistently experienced positive cash flow from operations, and our ability to achieve consistent positive cash flow will depend on increasing revenue from distribution of our products, which may not be achievable.
•Loss of key members of management, who we need to succeed, could adversely affect our business.
•Delays, interruptions, or the cessation of production by our third-party suppliers, including products supplied by single suppliers, of important materials may prevent or delay our ability to manufacture or process the final products.
•Technological change and competition for newly developed products could reduce demand for our products.
•We may not be successful in our efforts to build a pipeline of additional product candidates.
•We may be subject to future product liability litigation, which could be expensive, and our insurance coverage may not be adequate.
•We are currently operating in a period of economic uncertainty and capital markets volatility driven in part by growing geopolitical tensions and evolving global security risks, any of which could materially and adversely affect our business, financial condition and results of operations.
Risks Related to the Regulatory Environment in which We Operate
•If the FDA were to withdraw or suspend approval of, narrow the approved indication, or otherwise limit the use of our Avance Products, our revenues may be significantly impacted and would have a material adverse effect on us.
•Our business is subject to continuing regulatory compliance by the FDA and other authorities, which is costly and could result in negative effects on our business.
•The use, misuse or off-label use of our products may harm our reputation and the reputation of our products, which could result in injuries leading to product liability suits, and could be costly to our business, and/or result in FDA sanctions.
•BLA approval for Avance could result in different protocols for hospitals’ access to the product, as well as different coverage and reimbursement protocols, both of which may negatively affect surgeons’ access to, revenues derived from, and profitability of, Avance Products.
•Defective products could lead to recall or other negative business conditions.
•Clinical trials can be long and expensive, and results are ultimately uncertain.
•We rely on third parties to conduct our clinical trials, and they may not perform as contractually required or expected.
•Healthcare law and policy changes may have a material adverse effect on us.
•We could be subject to civil or criminal penalties if we are found to have violated laws protecting the confidentiality of health information, which could increase our liabilities and harm our reputation or our business.
Risks Related to Our Intellectual Property
•Failure to protect or maintain our IP rights could result in costly and time-consuming litigation and our loss of any potential competitive advantage.
•Others may claim an ownership interest in our IP or claim that we infringe on their IP rights, which could expose us to litigation and have a significant adverse effect on our prospects.
•Our trademarks are valuable, and our business may be adversely affected if trademarks are not adequately protected.
Risks Related to Financing Our Business
•We may need to raise additional funds to finance our future capital or operating needs, which could have adverse impacts on our business, results of operations and the interests of our shareholders.
29
ITEM 1A. RISK FACTORS
Our business involves a number of risks, some of which are beyond our control. The risk and uncertainties described below are not the only ones we face. Set forth below is a discussion of the risks and uncertainties that management believes to be material to us and could adversely affect our business, financial condition, results of operations, cash flows, growth prospects and the trading price of our common stock. Additional risks not currently known to us or that we currently deem to be immaterial also may materially adversely affect our business, financial condition or results of operations in future periods.
Risks Related to Our Business and Strategy
Approximately 60% of our total revenues are from sales of Avance Products and any adverse decision from the FDA would negatively impact our operations and financial condition.
Approximately 60% of our total revenues are from sales of Avance Products. The FDA approved Avance as a licensed biological product on December 3, 2025. Any action by the FDA following approval that withdraws or suspends the BLA, narrows the approved indication, imposes additional restrictions or limitations on use, or otherwise adversely affects the commercialization of Avance Products could have a material negative impact on our revenues and our operations. For additional information see: Risk Factors – If the FDA were to withdraw or suspend our BLA approval, narrow the approved indication, or otherwise limit the use of our Avance Products, our revenues would be significantly impacted and thus would have a material adverse effect on us. The FDA approved the BLA for Avance on December 3, 2025. However, if the FDA were to withdraw or suspend that approval, narrow the approved indication, or otherwise limit the use of our Avance Products, it would have a significant impact on our revenues and thus would have a material adverse effect on us.
Portions of the FDA-approved indication for Avance were granted under the FDA’s accelerated approval program, and continued approval for these indications is contingent upon our timely completion of required post-marketing confirmatory studies. Failure to verify clinical benefit or comply with accelerated approval requirements could result in withdrawal, restriction, or other adverse FDA action that would materially adversely affect our business, financial condition, and results of operations.
On December 3, 2025, the FDA approved the BLA for Avance, including for sensory nerve discontinuities greater than 25mm and for mixed and motor nerve discontinuities under the FDA’s accelerated approval pathway. As reflected in the prescribing information, continued approval of these indications is contingent upon verification and description of clinical benefit in confirmatory clinical trials. The FDA has required a post-marketing requirement study for sensory nerve discontinuities greater than 25 mm and for mixed and motor nerve discontinuities with a final protocol due February 5, 2026, study completion by December 5, 2030, and a final report due June 5, 2031, as well as progress reports every 180 days, beginning May 31, 2026.
Products approved under the accelerated approval program remain subject to heightened FDA oversight. If we fail to initiate, enroll, or complete the required confirmatory trial with due diligence, fail to meet specified milestones, fail to timely submit required progress reports, or fail to demonstrate clinical benefit in the confirmatory study, the FDA may withdraw approval of the affected indications on an expedited basis. In addition, the FDA retains authority to limit or modify these indications, impose additional post-marketing requirements, alter labeling, or require changes to our promotional materials. Even if we comply with all requirements, the confirmatory study may fail to verify the anticipated clinical benefit for sensory nerve gaps greater than 25mm or mixed or motor nerve discontinuities. The FDA could also determine that the results do not support continuation of the accelerated approval indications or that additional studies are necessary. Changes in FDA policies could also impact the design, timing, or acceptability of our confirmatory trial or otherwise affect our ability to maintain the accelerated approval indications.
If the FDA withdraws or restricts the accelerated approval portions of the Avance labeling, our ability to market the product for those uses would be limited or eliminated, which would materially adversely affect our commercialization efforts, reputation, competitive position, and revenue. Any such action also could result in significant operational, financial, and strategic consequences, and could negatively impact surgeon adoption or payer coverage. Delays or changes required to maintain the accelerated approval indications may require substantial additional resources and could divert management attention from other priorities.
The manufacturing process for Avance is complex and we or our third-party contractors may encounter difficulties in production. If we or our third-party contractors encounter such difficulties, our ability to supply Avance for commercial sale could be delayed or halted entirely, which would have a material adverse effect on our business.
30
The manufacture of Avance as a biological product is technically complex, highly regulated and subject to multiple risks. Production difficulties caused by unforeseen events may delay the availability of material used in the manufacture of Avance. The manufacturing process for Avance requires raw materials that are derived from biological sources, and as such each lot of starting material is inherently heterogenous in size and quality of material, which could materially harm our ability to produce Avance or supply finished product in sufficient quantity and sizes to meet market demand.
For example, our manufacturing process for Avance is dependent on the supply of donated cadaveric human tissue as a starting material. Cadaveric human tissue is a raw material that is susceptible to damage and contamination and may contain human pathogens, some of which may render a specific donor of tissue unsuitable as raw material for further manufacturing. If unsuitable tissue is not identified and discarded prior to the release of the tissue for further manufacturing, it may be necessary to discard intermediate or finished product made from that donor or to recall any finished product released to the market. Lot failures or product recalls or withdrawals could adversely affect supplies of Avance, which could be costly to us and otherwise harm our business, financial condition, results of operations and prospects. In addition, faulty or contaminated products that are unknowingly distributed could result in patient harm, threaten the reputation of our product and expose us to product liability claims and damages.
In addition, Avance is supplied commercially in an array of size combinations that vary by length and diameter of the tissue, and the available size combinations may not always match the commercial demand for our product. The source material of the product is donated human tissue, an inherently heterogenous starting material, and there is no assurance that specific diameter and length combinations will be generated with each manufacturing lot. This may cause temporary but unavoidable shortages in specific product sizes while generating an oversupply in others.
The production scale of Avance is dependent on the availability of a sufficient number of highly specialized manufacturing suites. We currently manufacture Avance for commercial distribution at our manufacturing facility in Vandalia, Ohio, where we have created a scalable manufacturing process that we believe will enable us to manufacture Avance in commercial quantities in compliance with applicable laws and regulations. Our ability to scale up Avance manufacturing beyond what was approved by FDA under the terms of our BLA for the product may require qualification of new manufacturing equipment and suites and may be subject to prior approval by FDA before implementation. Future efforts to scale up our manufacturing operations for Avance may not succeed. Scaling up a biologic manufacturing process is a difficult task, as there are risks including, among others, cost overruns, process reproducibility, stability issues, lot consistency and timely availability of raw materials. Additionally, our manufacturing process for Avance has evolved over time and we may not have the experience and resources to handle adoption of future changes or expansion of capacity. The forecasts of demand we plan to use to determine order quantities and lead times for Avance components from outside suppliers may be incorrect, and we may be unable to obtain such components when needed and at a reasonable cost. In addition, we currently rely on a third-party contractor to terminally sterilize our product. Any disruptions or planned shutdowns of our third-party contractor’s qualified site could cause temporary disruptions in our ability to supply Avance finished product, which could have an adverse effect on our business.
We also may encounter problems hiring and retaining the experienced scientific, quality control and manufacturing personnel needed to manage our manufacturing process for Avance, which could result in delays in our production or difficulties in maintaining compliance with applicable regulatory requirements.
31
Our revenue growth depends on our ability to increase distribution and sales to existing customers and develop new customers, domestically and internationally, and there can be no assurance that these efforts will result in significant increases in sales.
Beginning in 2020, we adjusted our commercial strategy to focus on deeper penetration of our existing surgeon customers through the development of long-term users of our algorithm of nerve repair in our largest market opportunity of extremity trauma. We believe that near-term growth can be supported first through expanded productivity of our existing sales force with existing accounts and second by adding additional customers. We expect the number of direct sales professionals to increase over time. Additionally, we believe that we have successfully utilized a hybrid commercial approach that includes the use of independent agencies in more remote geographies to provide appropriate local support for customers, without the travel time required of a direct sales representative. We may also need to establish a regional distribution center or centers at some point in the future to account for growth. The incurrence of these expenses may impact our operating results, and there can be no assurance of their effectiveness. If we are unable to increase sales to existing customers and attract new customers, and develop our sales force, there could be a material adverse impact on our business, results of operations, financial condition, and prospects. Additionally, our growth margin is dependent on maintaining a diversified demand mix. If demand only grows in one use application, it could negatively impact gross margin. We are focusing on creating balanced revenue growth and yield improvements in product processing, but we may be unable to do so.
We are highly dependent on the continued availability of our facilities and could be harmed if we continue to experience operating challenges with our APC Facility or if any of our facilities are unavailable for any prolonged period of time.
We transitioned the Avance Nerve Graft tissue processing and packaging to the APC Facility in November of 2023 but expect to continue to rely on the Dayton Facility for the processing of Avive+ Soft Tissue Matrix. We have experienced unanticipated operating challenges as we commenced processing operations at the APC Facility. Such challenges have negatively impacted our gross margins. Such challenges, if prolonged, could also cause a significant disruption in service to our customers if we were to lose, even temporarily, the availability of our production or distribution facilities. If we are not able to comply with the applicable regulatory requirements or produce products that meet our requirements and specifications, we will be subject to the same risks that would arise should third parties be unable to comply with the applicable regulatory requirements or produce products meeting our requirements or specifications, as described above. If we continue to experience operating challenges or fail to achieve the operating efficiencies that we anticipate, our business, results of operations, financial condition, and prospects could be adversely impacted.
In operating our APC facility, we have been forced to devote greater resources and management time than anticipated, particularly in areas relating to operations, quality control, raw material supply, regulatory compliance, facilities and information technology. We also have experienced unanticipated employee turnover at our APC Facility. If turnover at the APC Facility or any other facility is higher than anticipated, we may not be able to effectively manage our ongoing processing operations and we may not achieve the operating efficiencies that we anticipate from the APC Facility, which may negatively affect our business, results of operations, financial condition, and prospects.
Any failure in the physical infrastructure of our facilities, including the APC Facility, the Dayton Facility, and our distribution facility in Burleson, Texas could lead to significant costs and disruptions that could reduce our revenue and harm both our business reputation and financial results. Any natural or man-made event that impacts our ability to utilize our facilities could have a material impact on our business, results of operations, financial condition, and prospects. Although we have business interruption insurance that would cover certain costs in instances other than service agreement termination, it may not cover all costs nor help to regain our standing in the market. In addition, we may plan to expand the APC Facility or open additional office, lab or distributions space in the future, and our ability to license, renovate, rebuild, or find acceptable service facilities takes a considerable amount of time and expense.
If our estimates of the TAM for peripheral nerve repair and reconstruction are inaccurate, our growth prospects and financial performance could be materially adversely affected.
We estimate the market opportunity for our products based on assumptions regarding the number of peripheral nerve injuries and procedures, surgeon adoption of advanced nerve repair technologies, utilization across approved indications, reimbursement levels, and expected growth in clinical practice patterns. These estimates are inherently uncertain and may prove to be overstated. The actual number of procedures in which our products are used may be lower than we expect due to changes in clinical practice, competition from alternative repair techniques, reimbursement limitations, or shifts in surgeon or patient preferences. If our TAM is smaller than anticipated or does not grow as expected, our ability to achieve our projected revenue, expand adoption of our products, or execute our long-term strategy could be limited and could have a material adverse effect on our business, financial condition, and results of operations.
32
Our revenue depends on a limited number of products.
Substantially all of our revenue is currently derived from six products, Avance Products, Axoguard Nerve Protector, Axoguard Nerve Connector, Axoguard HA+ Nerve Protector, Axoguard Nerve Cap, and Avive+ Soft Tissue Matrix for the treatment of peripheral nerve damage. Of these six products, Avance Products represent approximately 60% of our total revenue. Any disruption in our ability to generate revenue from the processing, distribution, and sale of products, especially Avance Products, will have a material adverse impact on our business, results of operations, financial condition, and prospects.
Axoguard Nerve Connector and Axoguard Nerve Protector are only available through a distribution agreement with Evergen. The Distribution Agreement terminates on December 31, 2030. However, there are conditions for continuation of the agreement, including payment terms and minimum purchase requirements, that if breached could result in an earlier termination of the agreement. Through mutual agreement, the parties have not established such minimums and to date have not enforced such minimum purchase provision. Additionally, in the event that Evergen were to enforce minimum purchase quantities and we fail to reach an agreement as to such minimums, Evergen could terminate the agreement if we fail to generate commercially reasonable sales of Axoguard Nerve Connector and Axogen HA+ Nerve Protector as measured by sales similar to a competitive product at the same stage in its commercial launch as verified by a mutually acceptable third party. We distribute Axoguard Nerve Connector and Axoguard Nerve Protector for Evergen, and Evergen is the contract manufacturer for our Axoguard HA+ Nerve Protector and Axoguard Nerve Cap products. Although we believe we could develop or obtain products that would replace the Axoguard products obtained through the Evergen agreements, the loss of the ability to sell the Axoguard products could have a material adverse effect on our business, results of operations, financial condition, and prospects.
Our revenue depends upon prompt and adequate reimbursement from public and private insurers and national health systems.
Political, societal, economic, and regulatory influences are fundamentally changing the U.S. healthcare industry. The ability of a hospital or an ambulatory surgery center to pay fees for our products depends in part on the availability of adequate coverage and reimbursement from third-party payors for our products specifically, the procedures associated with the use of our products, or both. Providers that purchase our products generally rely on third-party payors to reimburse all or part of the costs and fees associated with the procedures performed with our products or the products themselves. Therefore, adequate coverage and reimbursement from third-party payors, including government payors such as Medicare and Medicaid, are important for obtaining product acceptance and widespread adoption in the marketplace.
When our products are used in the operating room of a hospital, they are currently commonly treated as general supplies utilized in surgery, and the cost is currently included in payment to the facility for the procedure. When Avance Nerve Graft and Axoguard Connector are used in an outpatient setting where the nerve repair is the primary reason for the procedure, facilities currently may use a Category I CPT code to facilitate payment.
In January 2018, the American Medical Association created a Category I CPT code (64912) specific to nerve repair with nerve allograft (Avance Nerve Graft) and a separate code (+64913) for each additional strand of allograft used in a procedure. Category I CPT codes are used by providers to facilitate payment to the provider (either hospital or ambulatory surgery center) for outpatient procedures. Additionally, Category I CPT codes are used to facilitate payment to the surgeon, for both time spent in outpatient and inpatient procedures. Prior to January 2018, there was no designated Category I CPT code for nerve repair cases that included nerve allograft. The Category I CPT code specific to nerve repair with nerve allograft, has allowed for nerve allograft repair cases to be uniquely identified in the Medicare claims data. This in turn allowed the Centers for Medicare & Medicaid Services (“CMS”) to have visibility to nerve allograft nerve procedure costs, and thereby confirm that nerve allograft qualified as a device intensive procedure.
Another important change in nerve repair reimbursement occurred in January 2020, when most direct repair procedures were moved from the higher paying level 2 nerve repair Ambulatory Payment Category 5432 to the lower paying level 1 Ambulatory Payment Category 5431, thus aligning payment rates more consistently with the lesser costs of a direct repair.
Effective January 1, 2026, CMS created a new Ambulatory Payment Category 5433 for level 3 nerve repair. CPT 64912 was moved into this new code group, which further improves payment adequacy for advanced nerve repair.
As a result of the allograft device intensive status and direct repair Ambulatory Payment Category realignment, CMS reimbursement rates for nerve repair in the outpatient setting have changed significantly during the last three years. With the new 2026 CMS reimbursement rates for nerve repair in the outpatient setting that became effective January 1st, reimbursement for procedures using Avance Nerve Graft have increased 96% in hospital outpatient centers and 221% in ambulatory surgery centers since 2019. During this same timeframe, reimbursement rates for procedures involving conduits and connectors also
33
increased 96% in hospital outpatient centers and 126% in ambulatory surgery centers. While Medicare patients represent a relatively small percentage of trauma cases, CMS’ direction often influences commercial payor policies and payments.
The process for securing coding for a product or procedure is separate from the process of securing coverage and establishing a reimbursement payment rate. In the U.S., coverage and reimbursement for medical devices varies among payors. In addition, payors review coverage policies on an ongoing basis and can change or deny coverage for these new products and procedures without notice. We estimate that commercial payors covering a significant number of U.S. covered lives have legacy non-coverage policies relating to our Avance Nerve Graft and our Axoguard Product Line, designating these products investigational or experimental. Some commercial payors do not currently cover or reimburse our products because they have determined insufficient evidence of favorable clinical outcomes is available. Although some payors consider Avance Nerve Graft and our Axoguard Product Line investigational or experimental at this time, these payors may in the future determine sufficient evidence has been developed to cover and reimburse our products and related procedures. In partnership with healthcare providers, we are working actively to reverse these non-coverage decisions and have been successful with several regional plans. However, we cannot provide assurance that we will continue to be successful in these efforts. If we are not successful in reversing existing non-coverage policies, or if other third-party payors issue similar policies, this could have a material adverse effect on our business and operations. Further, third-party payors who currently cover and reimburse customers for procedures using our products may in the future choose to decrease current levels of reimbursement or eliminate reimbursement altogether, which would cause our business to suffer.
The amount of reimbursement received by our customers from third-party payors is dependent generally on fee schedules established by these payors for the existing CPT codes. For governmental payors, such as Medicare and Medicaid, the fee schedule amount is determined by statutory and regulatory formulas as previously discussed. For commercial payors, the reimbursement amount generally is dependent upon the specific contract terms between the provider and payor. We cannot provide assurance that government or commercial payors will continue to reimburse for procedures with our products using the existing codes, nor can we provide assurance that the payment rates will be adequate. If providers and physicians are unable to obtain reimbursement for the procedure at adequate levels when use of our products is included, this could have a material adverse effect on our business and operations. Hospitals and ambulatory surgery centers may not purchase our products if they do not receive payment sufficient to cover the cost of our products and related procedures. In addition, in the event that the current coding and/or payment methodology for these procedures changes, this could have a material effect on our business, results of operations, financial condition, and prospects.
Additionally, healthcare law and policy changes may have a material adverse effect on our revenues. See Risk Factors – Healthcare law and policy changes may have a material adverse effect on us.
Our operating results could be adversely impacted if we are unable to effectively manage and sustain our future growth or scale our operations.
There can be no assurance that we will be able to manage our future growth efficiently or profitably. Our business is unproven on a large scale, and actual revenue and operating margins, or revenue and margin growth, may be less than expected. If we are unable to scale our production capabilities efficiently or maintain pricing without significant discounting, we may fail to achieve expected operating margins, which would have a material and adverse effect on our operating results. Growth may also stress our ability to adequately manage our operations, quality of products, safety, and regulatory compliance. Failure to implement necessary procedures, equipment, or processes or to hire the necessary personnel in a timely and effective manner could result in higher costs or an inability to meet market demand and could have a material adverse impact on our business, results of operations, financial condition, and prospects. Additionally, our future growth will increase the demands placed on our third-party suppliers, and there is no guarantee that our suppliers will be able to support our anticipated growth. If growth significantly decreases, it will negatively impact our cash reserves, and we may be required to obtain additional financing, which may increase indebtedness or result in dilution to shareholders. Further, there can be no assurance that we would be able to obtain additional financing on acceptable terms, if at all.
Our success will be dependent on continued acceptance of our products by the medical community.
Our success is dependent on continued acceptance of our products by the medical community, which will depend on our ability to demonstrate that our products are an attractive alternative to existing or new nerve reconstruction treatment options, including both surgical techniques and products. Our ability to do so will depend on surgeons’ evaluations of clinical safety, efficacy, ease of use, reliability, and cost-effectiveness, including insurance reimbursement, of our nerve repair products. For example, although our Avance Products follow stringent safety standards, including sterilization by gamma irradiation, we believe that a small portion of the medical community has lingering concerns over the risk of disease transmission through the use of allografts in general. If the medical community and patients do not ultimately accept our products as safe and effective or
34
we are unable to raise awareness of our products and processes, our ability to sell the products may be materially and adversely affected, and our business, results of operations, financial condition, and prospects may be adversely affected.
If we do not manage product inventory in an effective and efficient manner, it could adversely affect profitability.
Many factors affect the efficient use and planning of product inventory, such as our ability to predict demand for donor tissue, prepare manufacturing to meet that demand and product mix and handle product expiration. We may be unable to manage our inventory efficiently, keep inventory within expected budget goals, keep our work-in-process inventory on hand or manage it efficiently, control expired product or keep sufficient product on hand to meet demand. Finally, we can provide no assurance that we can keep inventory costs within our target levels, particularly in light of overall cost increases due to global inflation. Failure to do so may materially and adversely impact our business, results of operations, financial condition, and prospects.
There may be significant fluctuations in our operating results.
Significant quarterly fluctuations in our results of operations may be caused by, among other factors, our volume of revenue, seasonal changes in nerve repair activity, timing of sales force expansion, unforeseen restrictions on our ability to access healthcare providers such as inflationary pressures, competitive factors and general economic conditions. There can be no assurance that the level of revenue and profit, if any, we achieve in any particular fiscal period, will not be significantly lower than in other comparable fiscal periods. Our expense levels are based, in part, on our expectations as to future revenue. As a result, if future revenue is below expectations, net income or loss may be disproportionately affected by a reduction in revenue, as any corresponding reduction in expenses may not be proportionate to the reduction in revenue.
We have a history of net losses and have not consistently experienced positive cash flow from operations, and our ability to achieve consistent positive cash flow will depend on increasing revenue from distribution of our products, which may not be achievable.
We have historically incurred net losses and operated with negative cash flow from our operations and may continue to incur losses and operate with negative cash flow from operations for the foreseeable future. We have incurred net losses of $15.7 million, $10.0 million, and $21.7 million for the years ended December 31, 2025, 2024 and 2023, respectively. As of December 31, 2025, we had an accumulated deficit of approximately $307.0 million. If revenue does not increase as anticipated, we will continue to incur net losses and experience negative cash flows and adverse operating conditions. If we raise funds by selling additional equity, such sale would result in dilution to our shareholders. There is no assurance that if we are required to secure funding, we would be able to do so on terms acceptable to us, or at all.
Loss of key members of management, who we need to succeed, could adversely affect our business.
Our future success depends on the continued efforts of the members of our executive management team. Competition for experienced management personnel in the healthcare industry is intense. If one or more of our executives or other key personnel are unable or unwilling to continue in their present positions, or if we are unable to attract and retain high quality executives or key personnel in the future, our business, results of operations, financial conditions, and prospects may be adversely affected.
Delays, interruptions, or the cessation of production by our third-party suppliers, including products supplied by single suppliers, of important materials may prevent or delay our ability to manufacture or process the final products.
Most of the raw materials used in the process for Avance Products are available from more than one supplier. However, there are materials within the manufacturing and production process that come from single suppliers, some of which are outside of the U.S., or certain supplies which may be difficult to procure due to supply chain shortages or changes in global trade regulations. Macroeconomic factors could cause disruptions in the supply chain and impair our ability to obtain the materials needed for our product line.
We do not have written contracts that guarantee supply with our suppliers, and at any time they could stop supplying our orders. FDA review of a new supplier may be required if these materials become unavailable from our current suppliers. Although there may be other suppliers that have equivalent materials that would be available to us, if FDA review is required, it could take several months or years to obtain, if approval is able to be obtained at all. Any delay, interruption, or cessation of production by our third-party suppliers of important materials, or any delay in qualifying new materials, if necessary, would prevent or delay our ability to manufacture products. We are working on identifying and contracting with additional suppliers to reduce our dependence on single source suppliers and service providers.
35
In addition, an uncorrected impurity, a supplier’s variation in a raw material or testing, either unknown to us or incompatible with our manufacturing process, or any other problem with our materials, testing or components, would prevent or delay our ability to process tissue. These delays may limit our ability to meet demand for our products and delay our clinical trials, which would have a material adverse impact on our business, results of operations, financial condition, and prospects.
Technological change and competition for newly developed products could reduce demand for our products.
The medical technology industry is intensely competitive. We compete with both U.S. and international entities that engage in the development and production of medical technologies and processes, including:
•biotechnology, orthopedic, pharmaceutical, biomaterial, chemical, and other companies;
•academic and scientific institutions; and
•public and private research organizations.
Our products compete with autograft, hollow-tube conduits, commercially available wraps, and amnion products, as well as with alternative medical procedures. For the foreseeable future, we believe a significant number of surgeons will continue to choose to perform autograft procedures when feasible, despite the necessity of performing a second operation and its drawbacks. In addition, many members of the medical community will continue to prefer the use of hollow-tube conduits due in part to their familiarity with these products and the procedures required for their use. Also, steady improvements have been made in synthetic human tissue substitutes, which could compete with our products in the future. Unlike allografts, synthetic tissue technologies are not dependent on the availability of human or animal tissue. Although our growth strategy contemplates the introduction of new technologies, the development of these technologies is a complex and uncertain process, which require a high level of innovation, as well as the ability to accurately predict future technology and market trends. We may not be able to respond effectively to technological changes and emerging industry standards, or to successfully identify, develop or support new technologies or enhancements to existing products in a timely and cost-effective manner, if at all. There can be no assurance that in the future our competitors will not develop products that have superior performance or are less expensive relative to our products, rendering our products obsolete or noncompetitive. Due to our resource allocation, size, and relatively early stage, we may face competitive challenges from new products or existing products and barriers that are difficult to overcome and could negatively impact our growth.
We may not be successful in our efforts to build a pipeline of additional product candidates.
We may not be able to continue to identify and develop new product candidates in addition to our current pipeline. Even if we are successful in continuing to build our pipeline, the potential product candidates that we identify may require extensive resources to develop, may not be suitable for clinical development or may not achieve market acceptance. Failure or delay in development of new products could damage the reputation of our R&D capabilities and our reputation in the market as being a leader in the tissue nerve repair space. Furthermore, if we do not successfully develop and commercialize product candidates based upon our approach, we will not be able to obtain product revenue in future periods, which likely would result in significant harm to our financial position and adversely affect our stock price.
Negative publicity concerning methods of donating human tissue and screening of donated tissue may reduce demand for our products and negatively impact the supply of available donor tissue.
We are highly dependent on our ability to recover human peripheral nerve tissue from tissue donors for our Avance Products. The availability of acceptable donors is relatively limited, and this availability is impacted by regulatory changes, general public opinion of the donation process, and our reputation for handling the donation process. Media reports or other negative publicity concerning both improper methods of tissue recovery from donors and disease transmission from donated tissue, including bones and tendons, may limit widespread acceptance of our Avance Products. Unfavorable reports of improper or illegal tissue recovery practices, both in the U.S. and internationally, as well as incidents of improperly processed tissue leading to transmission of disease, may broadly affect the rate of future tissue donation and market acceptance of allograft technologies and donated tissue use. Potential patients may not be able to distinguish our products, technologies, and tissue recovery and processing procedures from others engaged in tissue recovery. In addition, unfavorable reports could make families of our potential donors or donors themselves from whom we are required to obtain consent before processing tissue reluctant to agree to donate tissue to for-profit tissue processors. Any disruption in the supply caused by these publicity issues could have a material impact for our business, results of operations, financial condition, and prospects.
36
The failure of third parties to perform many necessary services for the commercialization of our products, including services related to recovery/acquisition, sterilization, distribution, and transportation, would impair our ability to meet commercial demand.
We rely upon third parties for certain recovery/acquisition, sterilization, distribution, and transportation services for our products. For example, the Avance Nerve Graft processing consists of several steps, and we use a number of recovery and/or acquisition agencies to supply the human tissue needed for these products. While we believe our current contracts and the ability to enter into future contracts will provide us with the tissues required for the products, we cannot be sure that we will be able to obtain the tissue that we need in the future. Disruptions in the tissue supply may adversely impact both tissue products and our overall business. If any of the third parties that we rely upon in our recovery/acquisition, distribution or transportation process fail to comply with applicable laws and regulations, fail to meet expected deadlines, or otherwise do not carry out their contractual duties, experience delays due to macroeconomic factors, or encounter physical damage or natural disaster at their facilities, our ability to deliver product to meet commercial demand may be significantly impaired, which could have a material adverse impact on our business, results of operations, financial condition or prospects.
We are dependent on our relationships with independent agencies to generate a material portion of our revenue.
We derive material revenue through our relationships with independent agencies. In 2025, approximately 8% of global product revenue was generated through independent agencies. If certain agency relationships were terminated or discontinued for any reason, it could adversely affect our ability to generate revenue and profit. If we require additional agencies, we may not be able to find additional agencies who will agree to market and distribute our products on commercially reasonable terms, if at all. If we are unable to establish new agency relationships or renew certain current distribution agreements on commercially acceptable terms, our business, results of operations, financial condition, and prospects could be materially and adversely impacted.
We may be unsuccessful in commercializing our products outside the U.S.
To date, we have focused our commercialization efforts in the U.S., except for minor revenue in certain international countries. We intend to expand distribution and sales outside the U.S. and will need to comply with applicable foreign regulatory requirements, including obtaining the requisite approvals to do so. The regulatory environment for our portfolio of products is complex. Avance Nerve Graft is distributed in Canada, the U.K., and certain other countries. We originally received approval to distribute Avance Nerve Graft in Germany in December 2019. The original approval was for product manufactured at our Dayton Facility. We are in the process of updating our registration status with the German Health Authorities for product manufactured at our APC Facility and will be unable to ship the Avance Nerve Graft to Germany until we receive approval from The German Health Authorities. The Axoguard Nerve Connector and Axoguard Nerve Protector CE Mark has been renewed as of May 2021 by Evergen.
Further, we will need to either enter into distribution agreements with third parties or develop a direct sales force in international markets. If we do not obtain adequate levels of reimbursement from third-party payers outside of the U.S., we may be unable to develop and grow our revenue internationally. Outside of the U.S., reimbursement systems vary significantly by country. Many ex-U.S. markets have government-managed healthcare systems that govern reimbursement for medical devices, implants, and procedures. Some ex-U.S. reimbursement systems provide for limited payments in a given period and therefore result in extended payment periods. If we are unable to successfully commercialize our products internationally, our long-term growth prospects may be limited.
We may seek to expand our business in ways that could result in diversion of resources and extra expenses.
We may in the future pursue acquisitions of businesses, products and technologies, establish joint venture arrangements, or make minority equity investments to expand our business. We are unable to predict whether or when any prospective acquisition, equity investment or joint venture will be completed. The process of negotiating potential acquisitions, joint ventures or equity investments, as well as the integration of acquired or jointly developed businesses, technologies or products may be prolonged due to unforeseen difficulties and may require a disproportionate amount of our resources and management’s attention. We cannot assure you that we will be able to successfully identify suitable acquisition or investment candidates, complete acquisitions or investments, or integrate acquired businesses or joint ventures with our operations. If we were to make any acquisition or investment or enter into a joint venture, we may not receive the intended benefits of the acquisition, investment or joint venture or such an acquisition, investment or joint venture may not achieve comparable levels of revenues, profitability or productivity as our existing business or otherwise perform as expected. The occurrence of any of these events could harm our business, financial condition or results of operations. Future acquisitions, investments or joint ventures may require substantial capital resources, which may require us to seek additional debt or equity financing.
37
Future acquisitions, joint ventures or minority equity investments by us could result in the following, any of which could seriously harm our results of operations or the price of our stock:
•issuance of equity securities that would dilute our current shareholders’ percentages of ownership;
•large one-time write-offs or equity investment impairment write-offs;
•incurrence of debt and contingent liabilities;
•difficulties in the assimilation and integration of operations, personnel, technologies, products and information systems of the acquired companies;
•inability to realize cost efficiencies or synergies, thereby incurring higher operating expenditures as a result of the acquisition;
•diversion of management’s attention from other business concerns;
•contractual disputes;
•risks of entering geographic and business markets in which we have no or only limited prior experience; and
•potential loss of key employees of acquired organizations.
We may be subject to future product liability litigation, which could be expensive, and our insurance coverage may not be adequate.
Although we are not currently subject to any product liability proceedings and have no provision for product liability disbursements, we may incur material liabilities relating to product liability claims in the future, including product liability claims arising out of the usage of our products. Although we currently carry product liability insurance in an amount, we believe is consistent with industry averages, our insurance coverage and any provision we may maintain in the future for product related liabilities may not be adequate and our business, results of operations, financial conditions, and prospects could suffer material adverse consequences.
We are currently operating in a period of economic uncertainty and capital markets volatility driven in part by growing geopolitical tensions and evolving global security risks, any of which could materially and adversely affect our business, financial condition and results of operations.
We are exposed to the risk of changes in social, geopolitical, legal, and economic conditions. The global economy has been, and may continue to be, negatively impacted by regional conflicts and associated sanctions and trade restrictions, including as a result of reduced consumer demand, supply chain disruptions, increased cybersecurity risks, and increased costs for transportation, energy, and raw materials. Additionally, further escalation of trade tensions between the U.S. and China, heightened geopolitical competition in the Indo-pacific region, periodic flare-ups of instability in the Middle East, as well as other localized or emerging geopolitical disruptions, could result in a global economic slowdown and long-term changes to global trade. Although we do not have material operations in regions experiencing such conflicts or instability, further escalation of geopolitical tensions could have a broader impact that expands into other markets where we have material operations, which may adversely affect our business, financial condition and results of operations.
Further, changes in domestic and global economic conditions, supply chain disruptions, tight labor markets, as well as other stimulus and spending programs, have led to persistently high inflation, which is likely to lead to increased costs and may cause changes in fiscal and monetary policy. Additionally, our ability to access capital markets and other funding sources in the future may not be available on commercially reasonable terms, if at all. Impacts from inflationary pressures, such as increasing costs for research and development of our products, administrative and other costs of doing business, could adversely affect our business, financial condition and results of operations.
Additionally, our customers could experience financial and operational pressures as a result of labor shortages, supply chain disruptions, and increased inflation, which could impact their ability to access capital markets and other funding sources, increase cost of funding, or impede their ability to comply with debt covenants, which in turn could impede their ability to provide patient care, conduct further research and development, marketing and commercialization efforts, or impact their profitability. To the extent that our customers continue to face such financial pressures, it could impact their willingness to spend on our products and services, which could adversely affect our business, financial condition and results of operations.
Changes in U.S. trade policy, threats of international tariffs, and changes to the U.S. political landscape may adversely affect our business, results of operations, financial condition, and prospects.
Rising threats of international tariffs, including tariffs applied to goods traded between the U.S. and China, could materially and adversely affect our business, results of operations, financial condition, and prospects. Over the past several years, legislative and executive action from U.S. and foreign leaders has led to both threats of and the imposition of tariffs on certain
38
materials and products. The U.S. and China imposed tariffs or announced proposed tariffs to be applied in the future to certain of each other’s exports. Changes in political conditions in China and changes in the state of China-U.S. relations, including ongoing trade tensions, are difficult to predict and could adversely affect our operations or financial condition. We cannot predict the extent to which the U.S. or other countries will impose quotas, duties, tariffs, taxes or other similar restrictions upon the import or export of our products in the future, nor can we predict future trade policy or the terms of any renegotiated trade agreements and their impact on our business. The adoption and expansion of trade restrictions, the occurrence of a trade war, or other governmental action related to tariffs or trade agreements or policies has the potential to adversely impact demand for our products, our costs, our customers, our suppliers, and the U.S. economy, which in turn could have a material adverse effect on our business, results of operations, financial condition, and prospects.
Our failure to protect our technology systems and comply with data protection laws and regulations could lead to government enforcement actions and significant penalties against us, and adversely impact our business, results of operations, financial condition, and prospects.
We rely on information technology systems, including technology from third-party vendors, to process, transmit and store electronic information in our day-to-day operations. Similar to other companies, the size and complexity of our information technology systems makes them vulnerable to a cyberattack, malicious intrusion, breakdown, destruction, loss of data privacy, or other significant disruption. Our information systems require an ongoing commitment of resources to maintain, protect and enhance existing systems and develop new systems to keep pace with continuing changes in information processing technology, evolving systems and regulatory standards and the increasing need to protect patient and customer information. We expend significant resources to comply with applicable data privacy and security laws and regulations (together with applicable industry standards) and minimize the risk of security breaches, including deploying additional personnel and protection technologies, training employees annually, and engaging third-party experts and contractors. Significant and increasing investments of time and resources by management and the Board have been, and will continue to be, required to anticipate and address cybersecurity risks and incidents. However, given that the techniques used to obtain unauthorized access or to sabotage systems change frequently, and often are not identified until they are launched against a target, we may be unable to anticipate these techniques or implement adequate preventative measures in time to stop a cyber incident. Any failure by us to maintain or protect our information technology systems and data integrity could result in the unauthorized access to patient data and personally identifiable information, theft of IP or other misappropriation of assets, or otherwise compromise our confidential or proprietary information and disrupt our operations. Cyberattacks, intrusions, or other breaches could adversely impact our business, results of operations, financial condition, and prospects and potentially subject us to fines and penalties. See Item 1C. Cybersecurity for additional information related to cybersecurity risks.
In the U.S., federal and state privacy and security laws require certain of our operations to protect the confidentiality of personal information, including patient medical records and other health information. Limiting and/or restricting the use of certain personal data and information, as well as added transparency obligations to data subjects is becoming an increasing focus as evidenced by the implementation of the California Consumer Privacy Act which became effective on January 1, 2020. In Europe, E.U. member states and other foreign jurisdictions, including Switzerland, have adopted data protection laws and regulations which impose significant compliance obligations. Moreover, the collection and use of personal health data in the E.U. is governed by the E.U. General Data Protection Regulation (“GDPR”). The GDPR imposes several requirements relating to the consent of the individuals to whom the personal data relates, the information provided to the individuals, the security and confidentiality of the personal data, data breach notification and the use of third-party processors in connection with the processing of personal data. The GDPR also imposes strict rules on the transfer of personal data out of the E.U. to the U.S., provides an enforcement authority and imposes large penalties for noncompliance, including the potential for fines of up to 4% of the annual global revenue of the noncompliant company. The implementation of the GDPR has increased our responsibility and liability in relation to personal data that we process, including in clinical trials, and we may in the future be required to put in place additional mechanisms to ensure compliance with the GDPR, which could divert management’s attention and increase our cost of doing business.
Additionally, we expect that there will be other proposed laws, regulations and industry standards relating to privacy and data protection in the U.S., the E.U. and other jurisdictions, and we cannot determine the impact such future laws, regulations and standards may have on our business results of operations, financial condition, and prospects.
We are dependent on internal information and telecommunications systems, and any failure of these systems, including system security breaches, data protection breaches or other cybersecurity attacks, may negatively impact our business and results of operations.
Cyberattacks and other tactics designed to gain access to and exploit sensitive information by breaching mission critical systems of large organizations are constantly evolving and have been increasing in sophistication in recent years. High profile
39
security breaches leading to unauthorized release of sensitive information have occurred with increasing frequency at a number of major U.S. companies, despite widespread recognition of the cyberattack threat and improved data protection methods. While to date we have not experienced a significant data loss, significant compromise or any material financial losses related to cybersecurity attacks, our systems, those of our customers, and those of our third-party service providers are under constant threat. Cybercrime, including phishing, social engineering, attempts to overload our servers with denial-of-service attacks, or similar disruptions from unauthorized access to our systems, could cause us critical data loss or the disclosure or use of personal or other confidential information. Outside parties may attempt to fraudulently induce employees to disclose personally identifiable information or other confidential information which could expose us to a risk of loss or misuse of this information. Although we incur significant expenses to minimize the risk of security breaches, given that the techniques used to obtain unauthorized access or to sabotage systems change frequently, and often are not identified until they are launched against a target, we may be unable to anticipate these techniques of implement adequate preventive measures in time to stop or effectively mitigate a cyber incident. See Item 1C. Cybersecurity for additional information related to cybersecurity risks.
We are dependent on internal information and telecommunications systems, and we are vulnerable to failure of these systems, including through system security breaches, data protection breaches or other cybersecurity attacks. If these events occur, the unauthorized disclosure, loss or unavailability of data and disruption to our business may have a material adverse effect on our reputation and harm our relationships with vendors and customers. Additionally, these events may lead to financial losses from remedial actions, or potential liability from fines, including in relation to noncompliance with the GDPR, as well as possible litigation and punitive damages. Failures of our internal information or telecommunications systems may prevent us from taking customer orders, shipping products and billing customers. Sales may also be impacted if our customers are unable to access our pricing and product availability information. The occurrence of any of these events could have a material adverse impact on our business and results of operations.
Our business and financial performance could be adversely affected, directly or indirectly, by natural or man-made disasters or other similar events.
Neither the occurrence nor the potential impact of risks such as earthquakes, hurricanes, floods and other natural disasters, fire, power shortages, geopolitical unrest, war, terrorist attacks and other hostile acts, epidemics or pandemics, international hostilities or other criminal activities and other events beyond our control and the control of the third parties on which we depend can be predicted. However, these occurrences could impact us directly as a result of damage to our facilities or by preventing us from conducting our business in the ordinary course, or indirectly as a result of their impact on our customers, suppliers, or other counterparties. We could also suffer adverse consequences to the extent that these disasters affect the financial markets or the economy in general or in any particular region.
Additionally, climate change could present immediate and long-term risks to our industry and our customers. The potential for increased severe weather events could have a material adverse effect on our operations and infrastructure or the operations and infrastructure of our suppliers. In addition, the effects of climate change could include long-term changes in temperature levels and water availability, increased energy costs, and increased supply costs impacted by those increasing energy costs.
Our ability to mitigate the adverse consequences of such occurrences is in part dependent on the quality of our resiliency planning, and our ability, if any, to anticipate the nature of any such event that occurs. The adverse impact of natural or man-made disasters also could be increased to the extent that there is a lack of preparedness on the part of national or regional emergency responders or on the part of other organizations and businesses that we deal with, particularly those that we depend upon but have no control over.
Changes in the tax code could have a material adverse effect on our results of operations, financial condition, liquidity, and capital investments.
In recent years, political discourse has centered on potential changes in tax laws or tax rulings. Certain of these changes could negatively affect our financial condition. For example, on July 4, 2025, President Trump signed into law the One Big Beautiful Bill Act (“OBBBA”), which makes permanent key elements of the Tax Cuts and Jobs Act of 2017, including 100% bonus depreciation, domestic research and development cost expensing, and the business interest expense limitation. The OBBBA includes multiple effective dates, with certain provisions effective in 2025 and others implemented through 2027. While the new legislation did not have a material impact on our effective tax rate in 2025, future interpretations, regulations, or amendments to the OBBBA could impact our tax position in subsequent periods. In addition, our ability to use net operating loss and tax credit carryforwards and certain built-in losses to reduce future tax payments may be limited by provisions of the Internal Revenue Code, and it is possible that certain transactions or a combination of certain transactions may result in material additional limitations on our ability to use our net operating loss and tax credit carryforwards.
40
We may have exposure to additional tax liabilities as a result of our foreign operations.
We are subject to income taxes in the U.S. and various foreign jurisdictions. We have operations in Canada, Germany, the U.K., Spain and several other countries. Significant judgment is required in determining our worldwide provision for income taxes and other tax liabilities. In the ordinary course of a global business, there are many intercompany transactions and calculations where the ultimate tax determination is uncertain. We are regularly under audit by tax authorities. Our intercompany transfer pricing may be reviewed by the U.S. Internal Revenue Service and by foreign tax jurisdictions. Although we believe that our tax estimates are reasonable, due to the complexity of our corporate structure, the multiple intercompany transactions and the various tax regimes, we cannot assure you that a tax audit or tax dispute to which we may be subject will result in a favorable outcome for us. If taxing authorities do not accept our tax positions and impose higher tax rates on our foreign operations, our overall tax expenses could increase.
Risks Related to the Regulatory Environment in which We Operate
We obtained regulatory approval for certain uses of Avance through the FDA’s accelerated approval pathway and traditional approval will be contingent on successful completion of a confirmatory post-approval trial. Failure to successfully complete our confirmatory post-approval trial or obtain traditional approval would have a material adverse effect on our business.
Although the FDA granted traditional approval to Avance as an acellular nerve scaffold for the treatment of adult and pediatric patients aged one month or older with sensory nerve discontinuity (≤25mm), the FDA granted accelerated approval to Avance for the treatment of sensory nerve discontinuity (>25mm) and mixed and motor nerve discontinuity. For products granted accelerated approval, sponsors are required to verify and describe the product’s anticipated clinical benefit generally in the form of confirmatory trials. These confirmatory trials must be completed with due diligence and, pursuant to the Food and Drug Omnibus Reform Act (“FDORA”), the FDA is authorized to require a post-approval trial to be underway prior to approval or within a specified time period following approval. FDORA also requires the FDA to specify conditions of any required post-approval trial and requires sponsors to submit progress reports for required post-approval studies and any conditions required by the FDA. FDORA enables the FDA to initiate enforcement action for the failure to conduct with due diligence a required post-approval trial, including a failure to meet any required conditions, which may include enrollment targets, the study protocol and study milestones, specified by the FDA, or to submit timely reports. Failure to conduct our required post-approval confirmatory trial with due diligence, to meet the timelines and conditions agreed to by FDA for the completion of the confirmatory trial, or to verify and describe the anticipated clinical benefit of Avance during our post-approval confirmatory trial would allow the FDA to withdraw approval on an expedited basis as to the portions of the indication providing for use of Avance to treat sensory nerve discontinuity (>25mm), mixed and motor nerve discontinuity, or both, which would have a material adverse effect on our business.
In addition, all promotional materials for products approved under the accelerated approval pathway are subject to prior review by the FDA. If the FDA were to object to our promotional pieces, we may be required to revise our materials, or be subject to untitled or warning letters.
If the FDA were to withdraw or suspend our BLA approval, narrow the approved indication, or otherwise limit the use of our Avance Products, our revenues would be significantly impacted and thus would have a material adverse effect on us.
The FDA approved the BLA for Avance to treat sensory nerve discontinuity (<25mm) on December 3, 2025. Following approval, Avance is subject to all post-approval regulatory requirements applicable to licensed biologics, including manufacturing, labeling, packaging, storage, advertising, promotion, sampling, record-keeping, conduct of post-marketing clinical trials and submission of safety, efficacy and other post-approval information, including both federal and state requirements in the U.S. and requirements of comparable foreign regulatory authorities. If we fail to comply with any post-approval requirements, the FDA could take regulatory action, including issuing warning letter, imposing fines, mandating product recalls, restricting the approved indication, or withdrawing or suspending the BLA. Any such action would have a material adverse effect on our operations and financial viability.
Because approximately 60% of our total revenues are from sales of Avance Products, any narrowing of the approved indication, additional FDA-imposed restrictions, or other adverse action relating to the BLA could have a material negative impact on our revenues and our operations. For additional information see: Risk Factors - Approximately 60% of our total revenues are from sales of Avance Products and any adverse decision from the FDA would negatively impact our operations and financial condition.
41
Manufacturers and manufacturers’ facilities are required to continuously comply with FDA and comparable foreign regulatory authority requirements, including ensuring that quality control and manufacturing procedures conform to cGMP regulations and corresponding foreign regulatory manufacturing requirements. As such, we and our third-party suppliers will be subject to continual review and inspections to assess compliance with cGMP and adherence to commitments made in our BLA. Any failure by us or our third-party suppliers to adhere to or document compliance with such regulatory requirements could lead to a delay or interruption in the availability of materials for the manufacture of Avance, among other consequences. For example, Avance is supplied by a single Axogen owned and operated manufacturing facility and our ability to continue manufacturing Avance at this facility under the terms of our BLA is dependent on the successful implementation of certain updates to our previous environmental monitoring program. The FDA requires sponsors to conduct environmental monitoring for biologics manufacturing to control microbial and particulate contamination and ensure product safety and efficacy. We have an ongoing Environmental Monitoring Performance Qualification (“EMPQ”) study intended to demonstrate the adequacy of the
updates to our environmental monitoring program to the FDA and that they have been successfully implemented. We will need to demonstrate that our environmental monitoring program continues to be adequate post-licensure, and the FDA will verify the results of our EMPQ study during follow-up inspections of our facility. If we fail to properly qualify our environmental monitoring program or the FDA finds that the results of the EMPQ study require changes to our environmental monitoring program, we may be forced to implement additional monitoring procedures, obtain and qualify new equipment, or hire and train additional staff, and we may be forced to run a new EMPQ study, any of which could impact our ability to manufacture product, which would have a material effect on our business.
If we or our third-party contractors fail to comply with the requirements of the FDA or other regulatory authorities or fail to comply with other post-approval commitments, it could result in sanctions being imposed on us, including fines, injunctions, civil penalties, delays, suspension or withdrawal of approvals, clinical holds or termination of clinical trials. Form 483s, warning or untitled letters, regulatory communications warning the public about safety issues with a product, import or export refusals, license revocation, seizures, detentions, or recalls of product candidates or product, operating restrictions, criminal prosecutions or debarment, suits under the civil False Claims Act, corporate integrity agreements, or consent decrees any of which could significantly and adversely affect supplies of Avance and our business, financial conditions and results of operations could be materially adversely affected.
Our business is subject to continuing regulatory compliance by the FDA and other authorities, which is costly and could result in negative effects on our business.
We are subject to extensive regulation by foreign and domestic government entities, including compliance with regulations governing appropriate relationships with healthcare professionals, such as physicians, hospitals, distributors, third-party payors, patients, charitable organizations, and those to whom and through whom we may market our products. We are subject to various federal, state, and territorial laws in the U.S. and other jurisdictions in which we conduct business. These include, for example, anti-kickback laws, false claims laws, healthcare fraud, waste, and abuse laws, and anti-bribery laws such as the U.S. Foreign Corrupt Practices Act. Violations of these laws can be punishable by criminal and/or civil sanctions, including, in some instances, fines, imprisonment and, within the U.S., exclusion from participation in government healthcare programs, including Medicare, Medicaid, and Veterans Administration health programs. These laws are administered and enforced by, among others, the DOJ, which issued new compliance guidance in 2020, the Office of Inspector General of the Department of Health and Human Services, state attorneys general, and their respective counterparts in the applicable foreign jurisdictions in which we conduct business. Many of these agencies have increased their enforcement activities with respect to drug and medical device manufacturers in recent years. There can also be changes to the regulations by foreign and domestic government entities that require us to update or upgrade business processes or to perform additional validation activities for product or processes. Compliance with such changes can be costly to implement or result in non-compliance, thus restricting the ability to distribute tissue or sell products, which could have a material adverse effect on our business, results of operations, financial condition, and prospects.
Our products are also subject to regulation by the FDA in the U.S. The FDA regulates the development, pre-clinical and clinical testing, requirements for commercial marketing and distribution, manufacturing and quality, safety, labeling, and promotion of medical products including human cells, tissues and cellular and tissue-based products (“HCT/Ps”), medical devices, and biological products. See Business — Government Regulations — U.S. Government Regulation Overview.
The FDA also regulates medical devices, for example the Axoguard products, and generally requires them to be cleared through the 510(k) pre-market notification process prior to marketing or through other pre-market approval processes. The FDA’s pre-market review process for new and modified existing devices that precedes product marketing can be time consuming and expensive. Some of the future products and enhancements to such products that we expect to develop, and market may require marketing clearance or approval from the FDA.
42
There can be no assurance, however, that clearance or approval will be granted with respect to any of our medical device products or enhancements of marketed products or that we will continue to meet the FDA’s requirements applicable to the approved BLA for Avance Nerve Graft. FDA review of our devices or biological products may encounter significant delays during pre-market review or supplemental review processes, which could adversely affect our ability to market our products or enhancements. Any products regulated solely under Section 361 of the PHS Act are subject to close scrutiny by the FDA and potentially subject to regulatory change. Failure to comply with applicable regulatory requirements could expose us to potential compliance actions by FDA or other regulators and could risk the commercial availability of the product.
It is possible that if regulatory clearances or approvals to market a product are obtained from the FDA, the clearances or approvals may contain limitations on the indicated uses of such product and other uses may be prohibited, as well as require continuing regulatory review. Product approvals by the FDA can also be withdrawn due to failure to comply with regulatory standards or the occurrence of unforeseen problems following initial approval. The FDA may require post marketing clinical studies or other activities that may add cost or limit marketing of the product. Furthermore, the FDA could limit or prevent the distribution of our products, and the FDA has the authority to require the recall of such products. FDA regulations depend heavily on administrative interpretation, and there can be no assurance that future interpretations made by the FDA or other regulatory bodies will not adversely affect our business, results of operations, financial condition, and prospects. We, and our facilities, may be inspected by the FDA from time to time to determine compliance with various regulations relating to specifications, development, documentation, validation, testing, manufacturing, quality control and product labeling. A determination that we are in violation of such regulations could lead to imposition of civil penalties, including fines, product recalls or product seizures and, in certain cases, criminal sanctions.
Failure to obtain regulatory and pricing approvals in foreign jurisdictions after BLA approval for Avance or our other products could delay or prevent commercialization of our products abroad.
Distribution of our human tissue products outside the U.S. are subject to foreign regulatory requirements that vary from country to country. In the E.U., human tissue regulations, if applicable, differ from one E.U. member state to the next. Because of the absence of a fully harmonized regulatory framework and the proposed regulation for advanced therapy medicinal products in the E.U., as well as for other countries, the approval process for human derived cell or tissue based medical products may be extensive, lengthy, expensive, and unpredictable. Our products are subject to E.U. member states’ regulations that govern the donation, procurement, testing, coding, traceability, processing, preservation, storage, and distribution of human tissues and cells and cellular or tissue-based products. In addition, some E.U. member states have their own tissue banking regulations. The inability to meet foreign regulatory requirements could materially affect our future growth and compliance with such requirements could place a significant financial burden on us. As a result of Brexit, we cannot be sure what changes could occur or what the cost of regulatory compliance with the U.K. would be. Accordingly, the cost of regulatory compliance for sales outside the U.S. can be significant and time consuming.
As a result of the product’s BLA approval, some foreign regulators may request updated notifications or documentation regarding Avance’s U.S. regulatory status and this may require a Certificate of Foreign Government which we would obtain from the FDA. The approval procedure varies among countries and can involve additional testing. The time required to obtain approval abroad may differ from that required to obtain FDA approval. The foreign regulatory approval process may include all of the risks associated with obtaining FDA approval, as well as obtaining Certificates of Foreign Government from the FDA, and additional risks associated with requirements particular to those foreign jurisdictions where we will seek regulatory approval of our products. We may not obtain foreign regulatory approvals on a timely basis, if at all. Approval by the FDA does not ensure approval by regulatory authorities in other countries, and approval by one foreign regulatory authority does not ensure approval by regulatory authorities in other foreign countries. We and our collaborators may not be able to file for regulatory approvals and may not receive necessary approvals to commercialize our products in any market outside the U.S. The failure to obtain these approvals could materially adversely affect our business, financial condition and results of operations.
Finally, regulatory expectations in foreign jurisdictions are subject to constant change. There can be no assurance that we can meet the requirements of future regulations and guidance or that compliance with current regulations and guidance assures future capability to distribute and sell our products.
The use, misuse or off-label use of our products may harm our reputation and the reputation of our products, which could result in injuries leading to product liability suits, and could be costly to our business, and/or result in FDA sanctions.
If our products are misused or used for off-label purposes, our reputation and our product’s reputation may suffer, injuries could occur, which may lead to product liability litigation, or we may be subject to FDA sanctions if we are deemed to have engaged in off-label promotion. Following FDA approval of the BLA for Avance, including the accelerated approval pathway,
43
our promotional materials and training methods must comply with FDA requirements and other applicable laws and regulations, including the prohibition against off-label promotion. Our promotion of the Axoguard products, which are regulated as medical devices, also must comply with FDA’s requirements, and must only use labeling that is consistent with the specific indication(s) for use included in the FDA substantial equivalence order that results in marketing the devices. The FDA does not restrict or regulate a physician’s use of a medical product within the practice of medicine, and we cannot prevent a physician from using our products for an off-label use. However, the FD&C Act and the FDA’s regulations restrict the kind of promotional communications that may be made about our products, and if the FDA determines that our promotional or training materials constitute the unlawful promotion of an off-label use, it could request that we modify training or promotional materials and/or subject us to regulatory or enforcement actions, including the issuance of an untitled letter, a warning letter, civil money penalties, seizure, injunction or criminal fines, and penalties. Other federal, state, or foreign governmental authorities might also take action if they consider our promotion or training materials to constitute promotion of an uncleared or unapproved use, which could result in significant fines or penalties under other statutory authorities, such as laws prohibiting false claims for reimbursement, or exclusion from participation in federal health programs. In that event, our reputation could be damaged, and our products’ use in the marketplace could be impaired.
There may be increased risk of injury if physicians or others attempt to use our products off-label. Furthermore, the use of our product for indications other than those for which our products have been approved, cleared, or licensed by the FDA may not effectively treat the conditions not referenced in product indications, which could harm our reputation in the marketplace among physicians and patients. Physicians may also misuse our products or use improper techniques if they are not adequately trained in the particular use, potentially leading to injury and an increased risk of product liability litigation. Product liability claims are expensive to defend and could divert management’s attention from our primary business and result in substantial damage awards against us. Any of these events could harm our business, results of operations, financial condition, and prospects.
U.S. governmental regulation could restrict the use of our Avance Products, restrict our procurement of tissue or increase costs.
In addition to the FDA requirements for biological products, our Avance Products will continue to be subject to various requirements for human tissue under 21 CFR Part 1271. Human tissues intended for transplantation have been regulated by the FDA since 1993. In May 2005, three new comprehensive regulations went into effect that address manufacturing activities associated with HCT/P. The first regulation requires companies that produce and distribute HCT/Ps register with the FDA. The second regulation provides criteria that must be met for donors to be eligible to donate tissues and is referred to as the “Donor Eligibility” rule. The third regulation governs the processing and distribution of the tissues and is often referred to as the “Current Good Tissue Practices” rule. The Current Good Tissue Practices rule covers all stages of allograft processing, from procurement of tissue to distribution of final allografts. Together, the three basic requirements of 21 CFR Part 1271 are designed to ensure that sound, high quality practices are followed to reduce the risk of tissue contamination and of communicable disease transmission to recipients. These regulations increased regulatory scrutiny within the industry in which we operate and have led to increased enforcement actions, which affects the conduct of our business. In addition, guidance was issued by the FDA in November 2017 and revised in July 2020 on Regulatory Considerations for Human Cells, Tissues, and Cellular and Tissue-Based Products: Minimal Manipulation and Homologous Use, which could have potential implications on future HCT/P products being evaluated by us.
Additional regulations or guidance documents may be implemented by the FDA in the future. These changes may impose new documentation requirements, process changes or testing that could increase costs, and regulatory burden. See Business — Government Regulations. These regulations can also increase the cost of tissue recovery activities. Finally, our Avance Products are subject to certain state and local regulations, as well as compliance with the standards of the tissue bank industry’s accrediting organization, the AATB.
The procurement and transplantation of allograft nerve tissue is also subject to federal law pursuant to the National Organ Transplant Act (“NOTA”), a criminal statute that prohibits the purchase and sale of human organs used in human transplantation, including nerve and related tissue, for “valuable consideration.” NOTA only permits reasonable payments associated with the removal, transportation, processing, preservation, quality control, implantation, and storage of human nerve tissue. We make payments to certain of our clients and tissue banks for their services related to recovering allograft nerve and umbilical cord tissue on our behalf. If NOTA is interpreted or enforced in a manner that prevents us from receiving payment for services we render or prevents us from paying tissue banks or certain of our clients for the services they render for us, our business, results of operations, financial condition, and prospects could be materially and adversely affected.
We have engaged, through marketing employees, independent sales agents and sales representatives, in ongoing efforts designed to educate the medical community as to our products’ benefits, and we intend to continue our educational activities.
44
Although we believe that NOTA permits payments in connection with these educational efforts as reasonable payments associated with the processing, transportation and implantation of our products, payments in connection with such education efforts are not exempt from NOTA’s restrictions and our inability to make such payments in connection with these education efforts may prevent us from paying our sales representatives and could adversely affect our business, results of operations, financial condition, and prospects. No federal agency or court has determined whether NOTA is, or will be, applicable to every allograft nerve tissue-based material that our processing technologies may generate. Assuming that NOTA applies to our processing of allograft nerve and umbilical cord tissue, we believe that we comply with NOTA, but there can be no assurance that more restrictive interpretations of, or amendments to, NOTA will not be adopted in the future, which would call into question one or more aspects of our method of operations.
Failure to obtain regulatory or other approvals from certain states in which we operate after BLA approval for Avance could delay, hinder, or prevent commercialization of our products.
We are subject to regulations of state agencies which have statutes covering tissue banking. Regulations issued by Florida, New York, California, and Maryland, among other states, are particularly relevant to our business. Most states do not currently have tissue banking regulations. However, incidents of allograft related issues in the industry may stimulate the development of regulation in other states. It is possible that third parties may make allegations against us or against donor recovery groups or tissue banks about non-compliance with applicable FDA regulations or other relevant statutes or regulations. Allegations like these could cause regulators or other authorities to take investigative or other action or could cause negative publicity for our business and the industry in which we operate.
As a result of our BLA approval for Avance, we must obtain separate regulatory approvals and comply with numerous and varying state regulatory and licensing requirements. The approval procedures vary among states and can involve additional testing, supplying additional information, or filing state specific applications. The time required to obtain state approval may differ from that required to obtain FDA approval. The state approval processes may include all of the risks associated with obtaining FDA approval and additional risks associated with requirements particular to those states where we will seek approval to manufacture and/or distribute our products. We may not obtain state approvals on a timely basis, if at all. Approval by the FDA does not ensure approval by state authorities, and approval by one state authority does not ensure approval by authorities in other states. The failure to obtain these approvals could materially adversely affect our business, financial condition and results of operations.
Finally, state regulatory expectations are subject to constant change. There can be no assurance that we can meet the requirements of future regulations and guidance or that compliance with current regulations and guidance assures future capability to distribute and sell our products.
BLA approval for Avance could result in different protocols for hospitals’ access to the product, as well as different coverage and reimbursement protocols, both of which may negatively affect surgeons’ access to, revenues derived from, and profitability of, Avance Products.
BLA approval for Avance could result in a different characterization of the product which could result in different protocols for hospital access to the product and coverage and reimbursement changes. We historically entered into contracts to sell and distribute our products to hospitals and surgical facilities for use in caring for patients with peripheral nerve damage or transection. The BLA approval for Avance may require new processes for supplying our products to customers. While we have experience providing hospitals and surgical facilities with access to our products through existing processes, the BLA approval may require our Avance Products to be subject to new or additional protocols of hospitals and surgical facilities. If this occurs, the availability of Avance Products may be subject to new or differing review and approval processes that are increasingly impacted by pricing pressures and other cost-containment measures, each of which may adversely impact our ability to sell Avance Products profitably. In addition, third party payors may limit coverage to specific products on an approved list, or formulary, which might not include all of the FDA-approved products for a particular indication. As a result, a different characterization of Avance after BLA approval may also result in different coverage and reimbursement protocols which could reduce the amount of approved reimbursement for our products, deny coverage altogether, or impose new requirements to justify our prices. Any such result could reduce a surgeon’s access to Avance Products which would have an adverse impact on the revenues and profitability derived from Avance Products.
Our Axoguard products are subject to FDA and international regulatory requirements.
Our Axoguard Product Line is regulated as a medical device in the U.S. and international countries where we market Axoguard products. In the U.S., the Axoguard Product Line is regulated under the FD&C Act and subject to pre-market notification and clearance requirements under section 510(k) of the FD&C Act, 21 CFR Part 820 QSR and other FDA
45
regulations. In the rest of the world, each region (such as the E.U.) or country has their independent international regulations such as the Medical Device Regulations (“CE Mark”) in Europe, U.K. MHRA, and Taiwan Pharmaceutical Affairs Act.
We distribute Axoguard Nerve Connector and Axoguard Nerve Protector products for Evergen, and Evergen is responsible for the regulatory compliance of these products. In the U.S., Evergen has obtained a 510(k) pre-market clearance for Axoguard Nerve Connector from the FDA for porcine (pig) small intestine submucosa for the repair of peripheral nerve transections where gap closure can be achieved by flexion of the extremity. Evergen has also obtained a 510(k) pre-market clearance for Axoguard Nerve Protector for the repair of peripheral nerve damage in which there is no gap or where a gap closure is achieved by flexion of the extremity. In countries where Axoguard is marketed, Evergen has obtained regulatory clearance with the same indications except for Europe and the U.K. For the CE Mark, the Axoguard Nerve Protector indication is the same; however, for Axoguard Nerve Connector, the indication is more specific, as follows: “The Axoguard Nerve Connector is indicated for the repair of peripheral nerve discontinuities with gaps up to 5mm.”
We are the authorization holder of the Axoguard Nerve Cap and Axoguard HA+ Nerve Protector. We have obtained 510(k) pre-market clearance for Axoguard Nerve Cap, indicated to protect a peripheral nerve end and to separate the nerve from surrounding environment to reduce the development of symptomatic or painful neuroma. We have obtained two 510(k) pre-market clearances for Axoguard HA+ Nerve Protector. The first 510(k) K223640 was cleared on April 7, 2023, indicated for the management and protection of peripheral nerve injuries where there is no gap. The second 510(k) K231708 was cleared on October 12, 2023, expanding the indication to the management and protection of peripheral nerve injuries where there is no gap, or following closure of the gap.
If we or Evergen fail to comply with applicable regulatory requirements, the regulatory bodies in each country could deny or withdraw regulatory clearance/approval for the Axoguard products, or impose civil penalties, including fines, product seizures or product recalls and, in certain cases, criminal sanctions.
Our operations must comply with FDA and other governmental requirements.
Our operations require us to comply with the FDA’s and other governmental authorities’ laws and regulations on topics including the manufacture and production and sales and marketing of medical products, and compliance efforts related to such laws is costly, and failure to comply could subject us to enforcement action. See Business — Government Regulations — Education Grants, U.S. Anti-kickback, False Claims and Other Healthcare Fraud and Abuse Laws — Fraud, Abuse and False Claims. Enforcement actions could impair our ability to produce products in a cost-effective and timely manner to meet customer demands. We may also be required to bear other costs or take other actions that may have an adverse impact on our future revenue and our ability to generate profits. Furthermore, our key material suppliers, licensors, distributors, and our other contractors or partners may not continue to be in compliance with all applicable regulatory requirements, which could result in our failure to produce products on a timely basis and in the required quantities, if at all.
Healthcare providers and facilities, and third-party payors, often play a primary role in the recommendation and prescription of any currently marketed products and product candidates for which we may obtain marketing approval. Our current and future arrangements with healthcare providers and facilities, distributors, third-party payors, charitable organizations, disease state associations, patients, and customers, and our sales, marketing, and educational activities, may expose us to broadly applicable fraud and abuse and other healthcare laws and regulations (at the federal, state, and foreign level) that may constrain our business or financial arrangements and relationships through which we market, sell, and distribute our products for which we obtain marketing approval. In addition, our operations are also subject to various federal, state and foreign fraud and abuse, and payment transparency laws and regulations.
Payments made to physicians and other healthcare providers, and other financial interests, have been the subject of a range of federal and state laws. The federal physician payment transparency requirements, sometimes referred to as the Physician Payments Sunshine Act (the “Sunshine Act”), was created under the Affordable Care Act. The Sunshine Act, among other things, imposes reporting requirements on drug manufacturers for payments or other transfers of value made by them to physicians and teaching hospitals, as well as ownership and investment interests held by physicians, other healthcare providers, including physician assistants, nurse practitioners, and other mid-level healthcare practitioners, and their immediate family members. Reporting relative to these mid-level practitioners began in 2022 for payments or other transfers of value in 2021, which could increase the likelihood of a mistake in submission or failure to submit the required information by that group. Failure to submit required information may result in civil monetary penalties of up to an aggregate of $150 thousand per year and up to an additional aggregate of $1 million per year for “knowing failures,” for all payments, transfers of value or ownership or investment interests that are not timely, accurately, and completely reported in an annual submission. Additionally, certain states also mandate implementation of compliance programs, impose restrictions on marketing practices and/or require the tracking and reporting of gifts, compensation and other remuneration to physicians and other HCPs.
46
In addition to the federal fraud, waste, and abuse laws noted, there are analogous state laws and regulations, such as state anti-kickback and false claims laws, and other state laws addressing the medical product and healthcare industries, which may apply to items or services reimbursed by any third-party payor, including commercial insurers, and in some cases may apply regardless of payor, i.e., even if reimbursement is not available. Some state laws require pharmaceutical or device companies to comply with the industry’s voluntary compliance guidelines (the PhRMA Code and AdvaMed Code) and the relevant compliance program guidance promulgated by the OIG in addition to other requirements, many of which differ from each other in significant ways and may not have the same effect, thus complicating compliance efforts.
Finally, regulatory expectations in the U.S. are subject to constant change. There can be no assurance that we can meet the requirements of future regulations and guidance or that compliance with current regulations and guidance assures future capability to distribute and sell our products. It is possible that governmental authorities will conclude that our business practices do not comply with current or future statutes, regulations, agency guidance or case law involving applicable fraud and abuse or other healthcare laws and regulations. If any such actions are instituted against us, those actions could have a significant impact on our business, including the imposition of significant civil, criminal and administrative penalties, damages, disgorgement, monetary fines, imprisonment, possible exclusion from participation in Medicare, Medicaid and other federal healthcare programs or comparable foreign programs, contractual damages, reputational harm, diminished profits and future earnings, additional reporting requirements and/or oversight if we become subject to a corporate integrity agreement or similar agreement to resolve allegations of non-compliance with these laws, and curtailment of our operations, any of which could adversely affect our ability to operate our business and our results of operations. Although effective compliance programs can mitigate the risk of investigation and prosecution for violations of these laws, these risks cannot be entirely eliminated. Any action against us for an alleged or suspected violation could cause us to incur significant legal expenses and could divert our management’s attention from the operation of our business, even if our defense is successful.
Our business is subject to continuing compliance with standards set by various accreditation and registration bodies, which is costly, and loss of accreditation or registration could result in negative effects on our business.
We are subject to accreditation such as that by the AATB and as a National Association of Boards of Pharmacy accredited drug distributor. We have registration requirements such as that with ISO 13485 registration bodies. These accreditations and registration standards can affect distribution and sale of our products on a state-by-state basis, within the U.S. and also affects distribution and sale of our products outside of the U.S. The loss of accreditation or registration could keep us from selling and distributing our products, which may have negative effects on our business, results of operations, financial condition, and prospects.
Defective products could lead to recall or other negative business conditions.
If our products are defective or otherwise pose safety risks, the FDA could require their recall, or we may initiate a voluntary recall of our products. The risk of recall applies to both our medical device products and our FDA-approved biologic products, including those we market under a BLA, which are subject to FDA lot-level oversight and recall authority if any quantity of the product is determined to present an imminent or substantial hazard to public health. The FDA may require recall of a marketed medical device product, such as the Axoguard products, in the event that it determines the medical device presents a reasonable probability of serious adverse health consequences or death. However, most device recalls do not rise to this level of health significance and result from voluntary action. The FDA has authority to recall biological products when a batch, lot or other quantity of the product presents an imminent or substantial hazard to the public health. However, in such circumstances, the FDA usually initially requests voluntary recalls of biological products, such as the Avance Products. If a company does not comply with an FDA request for a recall, the FDA can order one under the above-referenced circumstances or take other enforcement actions, such as product seizure. In addition, manufacturers may, on their own initiative, recall a product to remove or correct a deficiency or to remedy a violation of the FD&C Act that may pose a risk to health. A government-mandated, government-requested, or voluntary recall could occur as a result of an unacceptable risk to health, reports of safety issues, failures, manufacturing errors, design or labeling defects or other deficiencies, and issues. Recalls and other field corrections for any of our products would divert managerial and financial resources and have an adverse effect on our business, results of operations, financial condition, and prospects. A recall could adversely impact our reputation with customers and our sales. If the FDA were to disagree with our internal determinations and decision making relative to potential recalls (including corrections and removal), we could be subject to further regulatory or enforcement action against.
If our products cause or contribute to a death, a serious injury, or any adverse reaction involving a communicable disease, or malfunction in certain ways, we will be subject to reporting regulations, which can result in voluntary corrective actions or agency enforcement actions. See Business — Government Regulations — Education Grants, U.S. Anti-kickback, False Claims and Other Healthcare Fraud and Abuse Laws. If we fail to report these events to the FDA within the required timeframes, or at all, the FDA could take regulatory or enforcement action against us. Any adverse event involving our products could result in
47
future voluntary corrective actions, such as recalls or customer notifications, or agency action, such as inspection, mandatory recall, or other enforcement action. Any corrective action, whether voluntary or involuntary, as well as defending ourselves in a lawsuit, would require the dedication of time and capital, distract management from operating our business, and may adversely impact our reputation, business, results of operations, financial condition, and prospects.
Clinical trials can be long and expensive, and results are ultimately uncertain.
The FDA’s approval of the BLA for Avance is conditioned on the completion of additional clinical or post-marketing studies for sensory nerve discontinuities greater than 25mm and for mixed and motor nerve discontinuities, and the FDA may determine that existing or future data do not adequately verify the clinical benefit of for sensory nerve discontinuities greater than 25mm and for mixed and motor nerve discontinuities. Under this post-marketing requirement, the Company will conduct a study comparing Avance to nerve autograft, with a final protocol due February 5, 2026, study completion by December 5, 2030, and a final report due June 5, 2031, as well as progress reports every 180 days, beginning on May 31, 2026.
The results of pre-clinical studies do not necessarily predict future clinical trial results and predecessor clinical trial results may not be repeated in subsequent clinical trials. Additionally, the FDA may disagree with our interpretation of the data from our pre-clinical studies and clinical trials and may require us to conduct additional clinical trials, modify our labeling, or comply with new post-marketing commitments. If we fail to satisfy any post-approval requirements, the FDA could restrict the approved indication, impose additional limitations on commercialization, or withdraw the approval entirely.
Even after approval, the FDA may determine that the totality of evidence does not continue to support the current labeling and could require changes that limit the approved indication, restrict marketing, or otherwise adversely affect commercialization of our Avance Products which could materially adversely affect our operations, financial condition, results of operations and prospects.
We rely on third parties to conduct our clinical trials, and they may not perform as contractually required or expected.
We rely on third parties, such as contract research organizations (“CROs”), medical institutions, clinical investigators, and contract laboratories to conduct our clinical trials and certain nonclinical studies. We and our CROs are required to comply with all applicable regulations governing clinical research, including Good Clinical Practices. The FDA enforces these regulations through periodic inspections of trial sponsors, principal investigators, CROs and trial sites. If we or our CROs fail to comply with applicable FDA regulations, the data generated in our clinical trials may be deemed unreliable and the FDA may require us to perform additional clinical trials before approving our applications. We cannot be certain that, upon inspection, the FDA and similar foreign regulatory authorities will determine that our clinical trial complies or complied with clinical trial regulations, including GCP. In addition, our clinical trial must be conducted with product produced under applicable GCP regulations. Failure to comply with the clinical trial regulations, including GCP, may require us to repeat clinical trials, which would delay the regulatory approval process. Further, if these third parties do not successfully carry out their contractual duties or regulatory obligations or meet expected deadlines, need to be replaced, or the quality or accuracy of the data they obtain is compromised due to the failure to adhere to our clinical protocols or regulatory requirements or for other reasons, our non-clinical development activities or clinical trials may be extended, delayed, suspended or terminated, and we would not be able to obtain regulatory approval for our products on a timely basis, if at all, and our business, results of operations, financial condition, and prospects would be adversely affected. Furthermore, our third-party clinical trial investigators may be delayed in conducting our clinical trials for reasons outside of their control.
Healthcare law and policy changes may have a material adverse effect on us.
In the U.S. there have been a number of legislative and regulatory changes and proposed changes regarding the healthcare system that could prevent or delay marketing approval of our product candidates, restrict or regulate post-approval activities, and affect our ability, or the ability of our collaborators, to profitably sell any products for which we obtain marketing approval. We expect that current laws, as well as other healthcare reform measures that may be adopted in the future, may result in more rigorous coverage criteria and in additional downward pressure on the price that we, or our collaborators, may receive for any approved products.
In the future, there may continue to be additional proposals relating to the reform of the U.S. healthcare system. Future legislation, federal agency regulations and Presidential Executive Orders may impact the healthcare system in ways important to our business. Adoption of certain proposals could limit the prices we are able to charge for our products or the amounts of reimbursement available for our products and could also limit the acceptance and availability of our products. The adoption of some or all of these proposals could have a material adverse effect on our business, results of operations, financial condition, and prospects.
48
Additionally, initiatives sponsored by government agencies, legislative bodies, and the private sector in the U.S. and elsewhere to limit the growth of healthcare costs, especially for drugs and biologics, including price regulation and policies regarding generic drugs and biosimilars, are ongoing in markets where we do business. For example, on August 16, 2022, the Inflation Reduction Act of 2022 (“IRA”) was signed into law. The IRA includes several provisions to lower prescription drug costs for people with Medicare and reduce drug spending by the federal government, including allowing Medicare to negotiate prices for certain prescription drugs, requiring drug manufacturers to pay a rebate to the federal government if prices for single-source drugs and biologics covered under Medicare Part B and nearly all covered drugs under Part D increase faster than the rate of inflation based on the consumer price index for all urban consumers, and limiting out of pocket spending for Medicare Part D enrollees. Implementation of the drug price negotiation provisions of the IRA began in 2023 and will continue to be implemented over the next several years. Multiple pharmaceutical manufacturers have challenged the law in court, largely on constitutional grounds. These suits remain ongoing, including at the appellate level, and the timing and ultimate outcome of such legal challenges are uncertain. At this time, we continue to evaluate the effect of the IRA on our business operations and financial condition and results as the full impact of the IRA remains uncertain. Additionally, on October 14, 2022, President Biden signed Executive Order 14087 on “Lowering Prescription Drug Costs for Americans.” The Executive Order specifically requests that the Center for Medicare and Medicaid Innovation consider “models that may lead to lower cost sharing for commonly used drugs and support value-based payment that supports high-quality care.” Continued government efforts to lower healthcare costs would affect our market materially. We could experience an adverse impact on operating results due to increased pricing pressure in the U.S. and in other markets. Governments, hospitals, pharmacy benefit managers, and other third-party payors could reduce the amount of approved reimbursement for our products, deny coverage altogether, or impose new requirements on manufacturers to justify their prices. Reductions in reimbursement levels or coverage or other cost-containment measures could unfavorably affect our future operating results.
We could be subject to civil or criminal penalties if we are found to have violated laws protecting the confidentiality of health information, which could increase our liabilities and harm our reputation or our business.
There are a number of federal and state laws protecting the confidentiality of certain health information and restricting the use and disclosure of that protected information. In particular, the U.S. Department of Health and Human Services promulgated privacy rules under the Health Insurance Portability and Accountability Act (“HIPAA”). These privacy rules protect medical records and other personal health information by limiting their use and disclosure, giving individuals the right to access, amend and seek accounting of their own health information and limiting most use and disclosures of health information to the minimum amount reasonably necessary to accomplish the intended purpose. If we are found to be in violation of the privacy rules under HIPAA, we could be subject to civil or criminal penalties, which could increase our liabilities, harm our reputation, and have a material adverse effect on our business, results of operations, financial condition, and prospects.
Risks Related to Our Intellectual Property
Failure to protect or maintain our IP rights could result in costly and time-consuming litigation and our loss of any potential competitive advantage.
Our success will depend, to a large extent, on our ability to successfully obtain and maintain patents, prevent misappropriation or infringement of IP, maintain trade secret protection, and conduct operations without violating or infringing on the IP rights of third parties. See Business — Intellectual Property. There can be no assurance that our patented and patent-pending technologies will provide us with a competitive advantage, that we will be able to develop or acquire additional technology that is patentable, or that third parties will not develop and offer technologies which are similar to ours. Moreover, we can provide no assurance that confidentiality agreements with our employees, consultants and other parties, agreements to protect trade secrets or similar agreements intended to protect unpatented technology or prevent unauthorized use, disclosure, or misappropriation will not be breached by those third parties. IP litigation is extremely expensive and time-consuming, and it is often difficult to predict the outcome of such litigation. A failure by us to protect our IP, or a breach by third parties of agreements aimed at protecting our IP, could have a materially adverse effect on our business, results of operations, financial condition, and prospects.
Future protection for our proprietary rights is uncertain and may impact our ability to successfully compete in our industry.
The degree of future protection for our proprietary rights is uncertain. We cannot ensure that:
•We, or our licensors, were the first to make the inventions covered by each of our patents;
•We, or our licensors, were the first to file patent applications for these inventions;
•Others will not independently develop similar or alternative technologies or duplicate any of our technologies;
49
•Any of our pending patent applications will result in issued patents;
•Any of our issued patents or those of our licensors are valid and enforceable;
•Any patents issued to us, or our collaborators will provide any competitive advantages or will not be challenged by third parties;
•We will develop additional proprietary technologies that are patentable;
•The patents of others will not have a material adverse effect on our business rights; or
•The measures we rely on to protect our IP underlying our products are adequate to prevent third parties from using, disclosing, or misappropriating that IP, all of which could harm our ability to compete in the market.
Our commercial success depends in part on our ability and the ability of our collaborators and licensors to avoid infringing patents and proprietary rights of third parties, which could expose us or our collaborators and licensors to litigation or commercially unfavorable licensing arrangements. Third parties may accuse us or collaborators and licensors of employing their proprietary technology without authorization in our products, or in the materials or processes used to make our products. Any legal action against our collaborators, licensors or those claiming damages and/or seeking to enjoin our commercial activities relating to the affected products, materials and processes could, in addition to subjecting us to potential liability for damages, require us or our collaborators and licensors to obtain a license to continue to utilize the affected materials or processes or to manufacture or market the affected products. We cannot predict whether we or our collaborators and licensors would prevail in any of these actions or whether any license required under any of these patents would be made available on commercially reasonable terms, if at all. If we were unable to obtain such a license, we and our collaborators and licensors may be unable to continue to utilize the affected materials or processes, or manufacture or market the affected products, or we may be obligated by a court to pay substantial royalties and/or other damages to the patent holder. Even if we were able to obtain such a license, the terms of such a license could substantially reduce the commercial value of the affected product or products and impair our prospects for profitability. Accordingly, we cannot predict whether, or to what extent, the commercial value of the affected product or products or our prospects for profitability may be harmed as a result of any of the liabilities discussed above. Furthermore, infringement and other IP claims, with or without merit, can be expensive and time-consuming to litigate and can divert management’s attention from our core business. We and our collaborators and licensors may be unable to obtain and enforce IP rights to adequately protect our products and related IP, which could materially and adversely impact our business, results of operations, financial condition, or prospects.
The patent protection for our products may expire before we are able to maximize their commercial value, which may subject us to increased competition and reduce or eliminate our opportunity to generate product revenue.
The patents for our commercialized products and products in development have varying expiration dates and, when these patents expire, we may be subject to increased competition, and we may not be able to recover our development costs. For example, the material U.S. patents covering the formulations used in our Axoguard Product Line, which are held by Evergen, have expired. Expiration of these patents could adversely affect our ability to successfully execute our business strategy to maximize the value of Axoguard products and could materially and adversely impact our business, results of operations, financial condition, and prospects.
Others may claim an ownership interest in our IP or claim that we infringe on their IP rights, which could expose us to litigation and have a significant adverse effect on our prospects.
A third party may claim an ownership interest in one or more of our patents or other IP. While we believe we own the right, title, and interest in the patents for which we or our licensors have applied and our other IP (including that which is licensed from third parties) and is presently unaware of any claims or assertions by third parties with respect to our patents or IP, we cannot guarantee that a third party will not assert a claim or an interest in any of such patents or IP.
Also, a third party may bring legal actions against us claiming we infringed their IP rights and seek monetary damages and/or enjoin clinical testing, manufacturing, and marketing of the affected product or products. There are many issued patents and pending patent applications in the U.S. and in other jurisdictions, owned by third parties, potentially covering various medical devices and biological products. There may be patents owned by third parties that we are currently unaware of, with issued claims that cover one or more of our current or future products or use or manufacture of those products. Since patents may take many years to issue, there may be pending patent applications owned by third parties that may lead to issued claims that cover one or more of our current or future products or use or manufacture of those products.
If we become involved in any litigation, it could consume a substantial portion of our resources and cause a significant diversion of effort by our technical and management personnel. If any of these actions were successful, in addition to any potential liability for damages, we could be required to obtain a license to continue to manufacture or market the affected
50
product, in which case we may be required to pay substantial royalties or grant cross-licenses to our patents. We cannot, however, assure that any such license will be available on acceptable terms, if at all. Ultimately, we could be prevented from commercializing a product or be forced to cease some aspect of our business operations as a result of claims of patent infringement or violation of other IP rights, which could have a material and adverse effect on our business, results of operations, financial condition, and prospects. Further, the outcome of IP litigation is subject to uncertainties that cannot be adequately quantified in advance, including the demeanor and credibility of witnesses and the identity of the adverse party. This is especially true in IP cases that may turn on the testimony of experts as to technical facts or the scope or meaning of patent claims upon which experts may reasonably disagree.
Our trademarks are valuable, and our business may be adversely affected if trademarks are not adequately protected.
In the U.S. and other countries, we currently hold trademark registrations and have trademark applications pending, any of which may be the subject of a governmental or third-party objection, which could prevent the maintenance or issuance of the same. As our products mature, our reliance on our trademarks to protect our brand, increase our name recognition and, in part, differentiate us from our competitors increases. As a result, if our trademark applications are not successful and if we are unable to prevent third parties from adopting, registering, or using trademarks, including trade dress, that infringe, dilute, or otherwise violate our trademark rights, our business, results of operations, financial condition, and prospects could be materially adversely affected.
Patent reform legislation could increase the uncertainties and costs surrounding the prosecution of our patent applications and the enforcement or defense of our issued patents.
New legislation or court precedent on patent law in the U.S. and in other jurisdictions may increase the uncertainties and costs for us to obtain and enforce patent claims broad enough to exclude others from making, using, or selling our current and future products. These changes in the patent law may also increase the uncertainties associated with the potential third party patent infringement claims against our current and future products. Depending on decisions by the U.S. Congress, the U.S. federal courts, the USPTO, or similar authorities in foreign jurisdictions, the laws and regulations governing patents could change in unpredictable ways to weaken our ability to obtain and enforce patent rights relevant to our products, and/or our ability to defend our business against third party infringement claims in the future.
Risks Related to Financing Our Business
We may need to raise additional funds to finance our future capital or operating needs, which could have adverse impacts on our business, results of operations and the interests of our shareholders.
We may need to seek to raise funds through the issuance of public or private debt or the sale of equity to achieve our business strategy. If we raise funds, this could dilute the interests of our shareholders. Moreover, the availability of additional capital, whether debt or equity from private capital sources (including banks) or the public capital markets, fluctuates as our financial condition and industry or market conditions in general change. There may be times when the private capital markets and the public debt or equity markets lack sufficient liquidity or when our securities cannot be sold at attractive prices, in which case we would not be able to access capital from these sources on favorable terms, if at all. We can give no assurance as to the terms or availability of additional capital.
Risks Related to Our Common Stock
An active trading market in our common stock may not be maintained.
The trading market in our common stock has been volatile. The quotation of our common stock on The Nasdaq Capital Market does not assure that a meaningful, consistent, and liquid trading market will exist. We cannot predict whether an active market for our common stock will be maintained in the future. An absence of an active trading market could adversely affect our shareholders’ ability to sell our common stock at current market prices in short time periods, or possibly at all. Additionally, market visibility for our common stock may be limited and such lack of visibility may have a depressive effect on the market price for our common stock. As of December 31, 2025, approximately 27.5% of our outstanding shares of common stock were held by our officers, directors, beneficial owners of 5% or more of our securities and their respective affiliates, which adversely affects the liquidity of the trading market for our common stock, in as much as federal securities laws restrict sales of our shares by these shareholders. If our affiliates continue to hold their shares of common stock, there will be limited trading volume in our common stock, which may make it more difficult for investors to sell their shares or increase the volatility of our stock price.
51
The price of our common stock could be volatile due to a number of factors, which could lead to losses by investors and costly securities litigation.
Our common stock is listed on The Nasdaq Capital Market under the symbol “AXGN.” The stock market in general, and the market for medical technology companies in particular, have experienced and could in the future experience volatility that has often been unrelated to the operating performance of particular companies. The trading price of our common stock has experienced volatility and is likely to continue to be volatile in response to a number of factors including, without limitation, the following:
•Fluctuations in price and volume due to investor speculation, including short sales, social media speculation and other factors that may not be tied to our financial performance;
•Our performance in the execution of our business plan;
•Financial viability;
•Actual or anticipated variations in our operating results;
•Announcements of developments by us or our competitors;
•Market conditions in our industry;
•Announcements by us or our competitors of significant acquisitions, strategic partnerships, joint ventures or capital commitments;
•Adoption of new accounting standards affecting our industry;
•Additions or departures of key personnel;
•Introduction of new products by us or our competitors;
•Sales of our common stock or other securities in the open market;
•Regulatory developments in both the U.S. and foreign countries;
•Performance of products sold and advertised by licensees in the marketplace;
•Economic and other external factors;
•Period-to-period fluctuations in financial results; and
•Other events or factors, including the other factors described in this “Risk Factors” section, many of which are beyond our control.
The stock market is subject to significant price and volume fluctuations. Such fluctuations have and could expose us to securities class action litigation, which could adversely impact our business, results of operations, financial condition, and prospects.
We may fail to meet our publicly announced guidance or other expectations about our business and future operating results, which could cause a decline in our stock price.
We provide financial guidance about our business and future operating results. In developing this guidance, our management makes certain assumptions and judgments about our future operating performance, including projected hiring of sales professionals, continued increase of our market share, and continued stability of the macro-economic environment in our key markets. Furthermore, analysts and investors may develop and publish their own projections of our business, which may form a consensus about our future performance. Our business results may vary significantly from such guidance or that consensus due to a number of factors, many of which are outside of our control, and which could adversely affect our operations and operating results. Furthermore, if we make downward revisions of our previously announced guidance, or if our publicly announced guidance of future operating results fails to meet expectations of securities analysts, investors, or other interested parties, the market price of our common stock could decline.
We do not anticipate paying any cash dividends in the foreseeable future.
The operation and expansion of our business will continue to require funding. We do not anticipate that we will pay any cash dividends on our common stock for the foreseeable future. Any determination to pay dividends in the future will be at the discretion of our Board and will depend upon results of operations, financial condition, contractual restrictions, restrictions imposed by applicable law, and other factors our Board deems relevant. Accordingly, if any investor purchases shares of common stock, realization of a gain on such investment will depend on the appreciation of the price of our common stock, which may never occur.
52
Anti-takeover provisions in Minnesota law may deter acquisition bids for us that you might consider favorable.
We are governed by the provisions of Sections 302A.671, 302A.673 and 302A.675 of the Minnesota Business Corporation Act (the “MBCA”). These provisions may discourage a negotiated acquisition or unsolicited takeover of us and deprive our shareholders of an opportunity to sell their common stock at a premium over the market price.
In general, Section 302A.671 of the MBCA provides that a corporation’s shares acquired in a control share acquisition have no voting rights unless voting rights are approved in a prescribed manner. A “control share acquisition” is a direct or indirect acquisition of beneficial ownership of shares that would, when added to all other shares beneficially owned by the acquiring person, entitle the acquiring person to have voting power of 20% or more in the election of directors.
In general, Section 302A.673 of the MBCA prohibits a public Minnesota corporation from engaging in a business combination with an interested shareholder for a period of four years after the date of the transaction in which the person became an interested shareholder, unless the business combination is approved in a prescribed manner. The term “business combination” includes mergers, asset sales, and other transactions resulting in a financial benefit to the interested shareholder. An “interested shareholder” is a person who is the beneficial owner, directly or indirectly, of 10% or more of a corporation’s voting stock or who is an affiliate or associate of the corporation, and who, at any time within four years before the date in question, was the beneficial owner, directly or indirectly, of 10% or more of the corporation’s voting stock. Section 302A.673 does not apply if a committee of our Board consisting of all of its disinterested directors (excluding current and former officers) approves the proposed transaction or the interested shareholder’s acquisition of shares before the interested shareholder becomes an interested shareholder.
If a tender offer is made for our common stock, Section 302A.675 of the MBCA precludes the offeror from acquiring additional shares of stock (including in acquisitions pursuant to mergers, consolidations, or statutory share exchanges) within two years following the completion of the tender offer, unless shareholders selling their shares in the later acquisition are given the opportunity to sell their shares on terms that are substantially the same as those contained in the earlier tender offer. Section 302A.675 does not apply if a committee of our Board consisting of all of its disinterested directors (excluding its current and former officers) approves the proposed acquisition before any shares are acquired pursuant to the earlier tender offer.
We are subject to legal proceedings from time to time. Legal proceedings, if decided adversely to or settled by us, and not covered by insurance, could result in liability material to our financial condition, results of operations or cash flows. Likewise, regardless of outcome, legal proceedings could result in substantial costs and expenses, affect the availability or cost of some of our insurance coverage and significantly divert the attention of our management. There can be no assurance that we will be able to prevail in, or achieve a favorable settlement of, any pending or future legal proceedings to which we become subject. Even claims without merit could subject us to adverse publicity and require us to incur significant legal fees.
Our management has broad discretion in the use and placement of our cash and cash equivalents and, despite management’s efforts, cash and cash equivalents may be used in a manner that does not increase the value of shareholders’ investments or placed in otherwise reputable financial institutions that fail.
Our management has broad discretion in the use and placement of our cash and cash equivalents, and investors must rely on the judgment of management regarding the use and placement of such cash and cash equivalents. Management may invest our cash and cash equivalents in short-term or long-term, investment-grade, interest-bearing securities. These investments may not yield favorable returns to shareholders. If we do not invest or apply our cash and cash equivalents in ways that enhance shareholder value, we may fail to achieve expected financial results, which could cause our stock price to decline. Furthermore, the most reputable financial institutions may fail. Despite the judgment of management regarding the placement of cash and cash equivalents in deemed reputable financial institutions, events outside of our control could occur, the result of which could result in us not having access to our cash and cash equivalents.
General Risks
Our business and stock price may be adversely affected if our internal controls are not effective.
Section 404 of the Sarbanes-Oxley Act of 2002 requires that public companies conduct a comprehensive evaluation of their internal control over financial reporting. To comply with this statute, each year we are required to document and test our internal control over financial reporting and our management is required to assess and issue a report concerning it.
Although we have systems in place to strengthen our internal control over financial reporting, we cannot assure you that we will not discover material weaknesses in the future or that no material weakness will result from any difficulties, errors, delays, or disruptions while we implement and transition to new internal systems. The existence of one or more material weaknesses
53
could result in errors in our financial statements, and substantial costs and resources may be required to rectify these or other internal control deficiencies. If we cannot produce reliable financial reports, investors could lose confidence in our reported financial information, the market price of our common stock could decline significantly, we may be unable to obtain additional financing to operate and expand our business and our business, results of operations, financial condition, and prospects could be adversely impacted.
We incur costs as a result of operating as a public company, and our management is required to devote substantial time to compliance initiatives.
As a public company, we incur legal, accounting, and other expenses to comply with relevant securities laws and regulations, including without limitation, the requirement of establishment and maintenance of effective disclosure and financial controls and corporate governance practices. Our management devotes substantial time and financial resources to these compliance initiatives. Failure to comply with public company requirements could have a material adverse effect on our business. In addition, activity by shareholders or others that bring into question aspects of our business, financial reporting, or management’s integrity, whether based on facts, beliefs or baseless and contrived for individual economic gain, can have a negative impact on the price of our stock and can result in substantial time and financial resources being expended to address the situation.
ITEM 1B. UNRESOLVED STAFF COMMENTS
None.
ITEM 1C. CYBERSECURITY
Cybersecurity represents an important component of our overall approach to risk management. Our cybersecurity policies, standards and practices are integrated into our enterprise risk management approach, and cybersecurity risks are one of the enterprise risks that are subject to oversight by our Board. Our cybersecurity standards and practices follow industry trends, which align with frameworks established by the Center for Internet Security. We approach cybersecurity threats through a cross-functional strategy which endeavors to: (i) identify, prevent and mitigate cybersecurity threats to us; (ii) preserve the confidentiality, security and availability of the information that we collect and store to use in our business; (iii) protect our IP; (iv) maintain the confidence of our customers, clients and business partners; and (v) provide appropriate public disclosure of cybersecurity risks and incidents when required.
Risk Management and Strategy
Our cybersecurity program focuses on the following areas:
a.Vigilance: We maintain cybersecurity threat operations with the goal of identifying, preventing and mitigating cybersecurity threats and responding to cybersecurity incidents in accordance with our established incident response and recovery plans.
b.Systems Safeguards: We deploy system safeguards that are designed to protect our information systems from cybersecurity threats, including firewalls, intrusion prevention and detection systems, anti-malware functionality and access controls, which are evaluated and improved through ongoing vulnerability assessments and cybersecurity threat intelligence.
c.Collaboration: We utilize collaboration mechanisms established with public and private entities, including intelligence and enforcement agencies, industry groups and third-party service providers, to identify, assess and respond to cybersecurity risks.
d.Third-Party Risk Management : We endeavor to identify and oversee cybersecurity risks presented by third parties, as well as the systems of third parties that could adversely impact our business in the event of a cybersecurity incident affecting those third-party systems.
e.Training: We provide periodic training for personnel regarding cybersecurity threats, which reinforces our information security policies, standards and practices.
f.Incident Response and Recovery Planning: We have established and maintain incident response and recovery plans that address our response to a cybersecurity incident and the recovery from a cybersecurity incident, and such plans are tested and evaluated periodically.
54
g.Communication, Coordination and Disclosure: We utilize a cross-functional strategy to address the risk from cybersecurity threats, involving management personnel from our technology, operations, legal, risk management and other key business functions, as well as the members of the Board and the Audit Committee of the Board in an ongoing dialogue regarding cybersecurity threats and incidents, while also implementing controls and procedures for the escalation of cybersecurity incidents pursuant to established thresholds so that decisions regarding the disclosure and reporting of such incidents can be made by management in a timely manner.
h.Governance: The Board’s oversight over the effectiveness of our cybersecurity risk management is supported by the Audit Committee, which regularly interacts with our Vice President of Information Technology and other members of the cyber team and management.
Governance
The Board, in coordination with the Audit Committee, oversees the effectiveness of management of risks from cybersecurity threats, including the policies, standards, processes and practices that our management implements to address risks from cybersecurity threats. The Board and the Audit Committee each receive regular presentations and reports on cybersecurity risks, which address a wide range of topics including, for example, recent developments, evolving standards, vulnerability assessments, third-party reviews, the threat environment, technological trends and information security considerations arising with respect to our peers. The Board and the Audit Committee also receive prompt and timely information regarding any cybersecurity incident that meets established reporting thresholds, as well as ongoing updates regarding such incident until it has been addressed. On a regular basis, the Board and the Audit Committee discuss our approach to cybersecurity risk management with our cyber team and senior leadership team.
Our Vice President of Information Technology in coordination with senior leadership, works collaboratively across the Company to implement a program designed to protect our information systems from cybersecurity threats and to promptly respond to any cybersecurity incidents. To facilitate the success of this program, multidisciplinary teams throughout the Company are deployed to address cybersecurity threats and to respond to cybersecurity incidents in accordance with our policy as it relates to the incident, management response and recovery plan. Through the ongoing communications from these teams, the Vice President of Information Technology and senior leadership monitor the prevention, detection, mitigation and remediation of cybersecurity incidents in real time, and report such incidents to the Audit Committee when appropriate.
55
ITEM 2. PROPERTIES
Our material physical properties consisted of the following as of December 31, 2025:
| Location | General Character | Total Square Feet | Square Feet Utilized | Expiration | ||||||||||||||||||||||
Alachua, Florida (1) | Headquarters - General office, warehousing and distribution | 19,000 | 19,000 | October 31, 2026 | ||||||||||||||||||||||
Tampa, Florida (1)(2) | Headquarters - General office, medical laboratory and meeting space | 75,000 | 50,000 | October 31, 2034 | ||||||||||||||||||||||
Burleson, Texas (1) | Facility - Raw material and finished goods warehousing and distribution | 15,000 | 15,000 | April 30, 2027 | ||||||||||||||||||||||
| 10,000 | 5,000 | September 30, 2027 | ||||||||||||||||||||||||
| Vandalia, Ohio | Facility - Clean-room, manufacturing, warehousing and office space | 107,000 | 84,000 | N/A | ||||||||||||||||||||||
Dayton, Ohio (3) | Facility - Clean-room, manufacturing, warehousing and office space | Varies | Varies | December 31, 2026 | ||||||||||||||||||||||
___________
(1)Property is encumbered by a lease agreement.
(2)Portions of this property are subleased under two sublease agreements with different sublessees.
(3)Property is encumbered by our License and Services Agreement as an embedded lease. Total square feet and utilization varies each month for the use of clean room, manufacturing, warehousing, and office space in accordance with the License and Services Agreement. See Note 15 - Commitments and Contingencies in the Notes to the Consolidated Financial Statements in Part II, Item 8 of this Form 10-K for details regarding the License and Services Agreement.
We believe that our facilities will be sufficient to operate our business for the next 12 months and that current lease obligations will not change materially.
ITEM 3. LEGAL PROCEEDINGS
Information required by this item is set forth in Note 15 - Commitments and Contingencies of the Notes to Consolidated Financial Statements in this Annual Report on Form 10-K and is incorporated herein by reference.
ITEM 4. MINE SAFETY DISCLOSURES
None.
56
PART II
ITEM 5. MARKET FOR REGISTRANT’S COMMON EQUITY, RELATED STOCKHOLDER MATTERS AND ISSUER PURCHASES OF EQUITY SECURITIES
Our common stock is traded on the Nasdaq Capital Market under the symbol “AXGN.” On February 20, 2026, the last reported closing sale price of our common stock on the Nasdaq Capital Market was $35.30 per share.
Shareholders
As of February 20, 2026, we had 51,897,682 shares of common stock outstanding, and approximately 178 common shareholders of record, based upon information received from our stock transfer agent. However, this number does not include beneficial owners whose shares were held of record by nominees or broker dealers. Additional information called for by this item is incorporated herein by reference to the following sections of this Report: Note 11 - Stock-Based Compensation of the Notes to Consolidated Financial Statements included in Item 8; and Part III, Item 12 Security Ownership of Certain Beneficial Owners and Management and Related Stockholder Matters – Equity Compensation Plan Information.
Purchases of Equity Securities by the Issuer and Affiliated Purchasers
We did not repurchase any of our securities in the fourth quarter of 2025.
Recent Sales of Unregistered Securities
We had no sales of unregistered securities in 2025.
Securities Authorized for Issuance Under Equity Compensation Plans
See Part III, Item 12 Security Ownership of Certain Beneficial Owners and Management and Related Stockholder Matters.
Dividends
We have never declared or paid and do not anticipate paying or declaring a cash dividend on our common stock. We intend to retain any earnings to finance the growth and development of our business. Our Board may declare dividends at its discretion.
57
Performance Graph
The following graph compares the cumulative total shareholder return on our common stock for the period from December 31, 2020 to December 31, 2025 with (i) the Nasdaq Stock Market Biotechnology Index and (ii) the Nasdaq Stock Market Composite Index. The graph assumes an investment of $100 in our common stock and the respective indices for the period of December 31, 2020 to December 31, 2025. The comparisons set forth in the graph are provided pursuant to SEC rules and are not intended to forecast or be indicative of the future performance of our common stock or either of the included indices. The performance graph shall not be deemed incorporated by reference by any general statement incorporating by reference this annual report into any filing under the Securities Act of 1933, as amended, or the Exchange Act of 1934, as amended, except to the extent we specifically incorporate this information by reference and shall not otherwise be deemed filed under such acts.
| December 31, | |||||||||||||||||||||||||||||||||||
| 2020 | 2021 | 2022 | 2023 | 2024 | 2025 | ||||||||||||||||||||||||||||||
| Axogen, Inc. | $ | 100.00 | $ | 52.35 | $ | 55.20 | $ | 38.60 | $ | 92.07 | $ | 182.85 | |||||||||||||||||||||||
| Nasdaq Stock Market Biotechnology Index | $ | 100.00 | $ | 100.02 | $ | 89.07 | $ | 95.89 | $ | 93.49 | $ | 124.75 | |||||||||||||||||||||||
| Nasdaq Stock Market Composite Index | $ | 100.00 | $ | 122.18 | $ | 81.81 | $ | 117.28 | $ | 154.48 | $ | 187.14 | |||||||||||||||||||||||
ITEM 6. [RESERVED]
Not applicable.
58
Axogen, Inc.
Management’s Discussion and Analysis of Financial Condition and Results of Operations
(in thousands, except share and per share amounts)
ITEM 7. MANAGEMENT’S DISCUSSION AND ANALYSIS OF FINANCIAL CONDITION AND RESULTS OF OPERATIONS
The following information should be read in conjunction with our consolidated financial statements and the notes thereto contained in Item 8 of Part II in this Form 10-K, “Forward-Looking Statements” contained in Part I of this Form 10-K, “Risk Factors” contained in Item 1A of this Form 10-K, and the other information appearing elsewhere in, or incorporated by reference into, this Form 10-K. Dollar amounts referenced in this Item 7 are in thousands, except per share amounts.
Unless the context otherwise requires, all references in this report to “Axogen,” the “Company,” “we,” “us” and “our” refer to Axogen, Inc., and its wholly owned subsidiaries Axogen Corporation (“AC”), Axogen Processing Corporation, Axogen Europe GmbH and Axogen Germany GmbH.
Overview
We are the leading company focused specifically on the science, development, and commercialization of technologies for peripheral nerve regeneration and repair. We are passionate about providing the opportunity to restore nerve function and quality of life for patients with peripheral nerve injuries. We provide innovative, clinically proven, and economically effective repair solutions for surgeons and healthcare providers. Peripheral nerves provide the pathways for both motor and sensory signals throughout the body. Every day, people suffer traumatic injuries or undergo surgical procedures that impact the function of their peripheral nerves. Physical damage to a peripheral nerve or the inability to properly reconnect peripheral nerves can result in the loss of muscle or organ function, the loss of sensory feeling, or the initiation of pain.
Product Portfolio
Our platform for peripheral nerve repair features a comprehensive portfolio of products, including:
•Avance® (acellular nerve allograft-arwx) an FDA-approved acellular nerve scaffold for the treatment of adult and pediatric patients aged one month or older with sensory, mixed, and motor peripheral nerve discontinuities (“Avance”).
•Avance® Nerve Graft, a biologically active off-the-shelf processed human nerve allograft for bridging severed peripheral nerves without the comorbidities associated with a second surgical site (“Avance Nerve Graft” and together with Avance, the “Avance Products”).
•Axoguard Nerve Connector®, a porcine (pig) submucosa extracellular matrix (“ECM”) coaptation aid for tensionless repair of severed peripheral nerves.
•Axoguard Nerve Protector®, a porcine submucosa ECM product used to wrap and protect damaged peripheral nerves and reinforce the nerve reconstruction while minimizing soft tissue attachments.
•Axoguard HA+ Nerve Protector™, a porcine submucosa ECM base layer coated with a proprietary hyaluronate-alginate gel, a next-generation technology designed to enhance nerve gliding and provide short- and long-term protection for peripheral nerve injuries.
•Axoguard Nerve Cap®, a porcine submucosa ECM product used to protect a peripheral nerve end and separate the nerve from the surrounding environment to reduce the development of symptomatic or painful neuroma.
•Avive+ Soft Tissue Matrix™, a multi-layer amniotic membrane allograft used to protect and separate tissues in the surgical bed during the critical phase of tissue healing.
Our portfolio of products is currently available in the United States (“U.S.”), Canada, Germany, the United Kingdom, Spain and several other countries.
We derive substantially all of our revenues from sales of our nerve repair products to customers in the U.S.
On December 3, 2025, the FDA approved the Biologics License Application (“BLA”) for Avance (acellular nerve allograft-arwx). Continued approval depends on verification and description of clinical benefits in confirmatory studies.
59
Axogen, Inc.
Management’s Discussion and Analysis of Financial Condition and Results of Operations - Continued
(in thousands, except share and per share amounts)
Our strategy remains focused on deepening our presence in high-potential accounts, specifically Level 1 trauma centers and academic-affiliated hospitals with a high number of trained microsurgeons. We will drive growth in these accounts through targeted expansion of nerve repair indications and driving deeper adoption of our nerve repair algorithm across multiple surgical specialties.
Summary of Operational and Business Highlights
•Revenue was $225,208 for the year ended December 31, 2025, an increase of $37,870, or 20.2%, compared to the year ended December 31, 2024.
•Gross profit was $167,353 for the year ended December 31, 2025, an increase of $25,376, or 17.9%, compared to the year ended December 31, 2024.
•Gross margin reflects one-time costs of approximately $1.9 million, or 1% for full-year 2025, respectively, related to the FDA BLA approval of Avance®. 67% of the one-time costs are non-cash and relate to the vesting of certain stock compensation awards containing FDA BLA approval of Avance® milestones.
•Net loss for the full-year 2025 was $15.7 million, or $0.34 per share, compared to $10.0 million, or $0.23 per share for 2024.
•As of December 31, 2025, cash and cash equivalents, restricted cash, and investments was $45.5 million, as compared to $39.5 million as of December 31, 2024, an increase of $6.0 million.
•Expanded coverage and reimbursement for nerve repair for peripheral nerve injuries using synthetic conduits or allografts, increasing the total number of new lives covered in 2025 to approximately 19.8 million and bringing coverage amongst commercial payers to more than 65%.
•Effective January 1, 2026, CMS created a new Level 3 Nerve Procedure Code, increasing Avance facility reimbursement 40% year-over-year to $9 for hospital outpatient and 35% to $6 for ASC-based procedures.
•On January 23, 2026, we closed an upsized public offering with the sale of 4,600,000 shares of common stock and receipt of $133,338 of net proceeds. $69,707 of the net proceeds were used to fully repay and terminate our term loan facility on January 28, 2026 with the remaining funds available for working capital, capital expenditures, and other general corporate purposes.
60
Axogen, Inc.
Management’s Discussion and Analysis of Financial Condition and Results of Operations - Continued
(in thousands, except share and per share amounts)
Results of Operations
Comparison of the Years Ended December 31, 2025 and 2024
The following table sets forth our results of operations expressed as dollar amounts and as percentages of total revenue for the periods presented:
| Years Ended December 31, | |||||||||||||||||||||||
| 2025 | 2024 | ||||||||||||||||||||||
| (dollars in thousands) | Amount | % of Revenue | Amount | % of Revenue | |||||||||||||||||||
| Revenues | $ | 225,208 | 100.0 | % | $ | 187,338 | 100.0 | % | |||||||||||||||
| Cost of goods sold | 57,855 | 25.7 | 45,361 | 24.2 | |||||||||||||||||||
| Gross profit | 167,353 | 74.3 | 141,977 | 75.8 | |||||||||||||||||||
| Cost and expenses | |||||||||||||||||||||||
| Sales and marketing | 97,740 | 43.4 | 78,461 | 41.9 | |||||||||||||||||||
| Research and development | 32,885 | 14.6 | 27,767 | 14.8 | |||||||||||||||||||
| General and administrative | 44,577 | 19.8 | 39,036 | 20.8 | |||||||||||||||||||
| Total costs and expenses | 175,202 | 77.8 | 145,264 | 77.5 | |||||||||||||||||||
| Loss from operations | (7,849) | (3.5) | (3,287) | (1.8) | |||||||||||||||||||
| Other income (expense): | |||||||||||||||||||||||
| Investment income | 1,168 | 0.5 | 1,141 | 0.6 | |||||||||||||||||||
| Interest expense | (7,702) | (3.4) | (8,206) | (4.4) | |||||||||||||||||||
| Change in fair value of derivatives | (1,487) | (0.7) | 587 | 0.3 | |||||||||||||||||||
| Other expense | 167 | 0.1 | (199) | (0.1) | |||||||||||||||||||
| Total other expense, net | (7,854) | (3.5) | (6,677) | (3.7) | |||||||||||||||||||
| Net loss | $ | (15,703) | (7.0) | % | $ | (9,964) | (5.3) | % | |||||||||||||||
Revenues
Revenues for the year ended December 31, 2025 increased $37,870, or 20.2%, to $225,208, as compared to $187,338 for the year ended December 31, 2024. Revenue growth was driven by an increase in unit volume, as well as the impact of changes in price and product mix.
Gross Profit
Gross profit for the year ended December 31, 2025 increased $25,376, or 17.9%, to $167,353, as compared to $141,977 for the year ended December 31, 2024. Gross margin as a percentage of revenue decreased to 74.3% for the year ended December 31, 2025, as compared to 75.8% for the year ended December 31, 2024. Gross margin reflects one-time costs of approximately $1,900, or 1%, related to the FDA BLA approval of Avance, with approximately 67% of such costs related to the vesting of stock compensation awards containing FDA BLA approval of Avance milestones. The decrease in gross margin was due to higher product costs and BLA-related stock-based compensation costs, partially offset by lower inventory write-offs.
61
Axogen, Inc.
Management’s Discussion and Analysis of Financial Condition and Results of Operations - Continued
(in thousands, except share and per share amounts)
Costs and Expenses
Following is a summary of the change in costs and expenses for the year ended December 31, 2025:
| (dollars in thousands) | Total costs and expenses | Sales and marketing | Research and development | General and administrative | |||||||||||||||||||
| For the year ended December 31, 2024 | $ | 145,264 | $ | 78,461 | $ | 27,767 | $ | 39,036 | |||||||||||||||
| Change from: | |||||||||||||||||||||||
Compensation costs (1) | 22,230 | 13,312 | 5,481 | 3,437 | |||||||||||||||||||
| Marketing program costs | 3,676 | 3,676 | — | — | |||||||||||||||||||
| Travel costs | 2,446 | 2,063 | 213 | 170 | |||||||||||||||||||
| Occupancy related costs | (1,145) | (269) | (316) | (560) | |||||||||||||||||||
Research and development project costs (2) | (69) | — | (69) | — | |||||||||||||||||||
| Professional services fees and expenses | (12) | 209 | (207) | (14) | |||||||||||||||||||
| Other | 2,812 | 288 | 16 | 2,508 | |||||||||||||||||||
| Total change | 29,938 | 19,279 | 5,118 | 5,541 | |||||||||||||||||||
| For the year ended December 31, 2025 | $ | 175,202 | $ | 97,740 | $ | 32,885 | $ | 44,577 | |||||||||||||||
| Percentage change | 20.6 | % | 24.6 | % | 18.4 | % | 14.2 | % | |||||||||||||||
__________
(1)The increase in compensation costs is primarily due to higher: (i) stock-based compensation, primarily due to $7,236 from PSU awards vesting in connection with Avance BLA approval by the FDA, and (ii) salaries and sales commissions, due to higher headcount and sales volume. BLA approval-related stock compensation expenses included $749 in sales and marketing, $4,600 in research and development, and $1,887 in general and administrative.
(2)The decrease in research and development costs and expenses was primarily due to product development and clinical expenses. Product development costs include spending for a number of specific programs, including the non-clinical expenses related to the BLA for Avance. Product development costs and expenses represented approximately 50% and 53% of total research and development costs and expenses for the years ended December 31, 2025 and 2024, respectively. Clinical trial costs and expenses represented approximately 50% and 47% of total research and development costs and expenses for the years ended December 31, 2025 and 2024, respectively.
Other Expense
Total other expense, net increased $1,177, or 17.6%, to $7,854 for the year ended December 31, 2025, as compared to $6,677 for the year ended December 31, 2024. The increase was primarily due to the change in the fair value of debt derivative liabilities. See Note 6 - Fair Value Measurement in the Notes to the Consolidated Financial Statements in Part II, Item 8 of this Form 10-K for details regarding the valuation of the debt derivative liabilities.
Income Taxes
We had no federal income tax expense or benefit for the years ended December 31, 2025 and 2024 due to the incurrence of net operating losses in both years, the benefits of which have a full valuation allowance. From time to time, we receive notices of examination of prior tax filings from federal and state authorities. The Internal Revenue Service is currently examining the Company’s 2021 federal income tax return.
Liquidity and Capital Resources
As of December 31, 2025, our principal sources of liquidity were our cash and cash equivalents and investments totaling $41,528. Our cash equivalent is comprised of a money market mutual fund and our investments are comprised of U.S. Treasuries. Our cash and cash equivalents and investments increased $8,046 to $41,528 from $33,482 at December 31, 2024, primarily due to proceeds from the exercise of stock options and an increase in proceeds from the sale of investments, net of investment purchases, partially offset by a reduction in cash generated from general operating activities. On December 31, 2025 and 2024, our current assets exceeded our current liabilities by $96,866 and $68,607, respectively. Based on current estimates, we believe that our existing cash and cash equivalents and investments, as well as cash provided by sales of our products will allow us to fund our operations through at least the next twelve months from the date of issuance of the accompanying financial statements.
62
Axogen, Inc.
Management’s Discussion and Analysis of Financial Condition and Results of Operations - Continued
(in thousands, except share and per share amounts)
Cash Flow Information
The following table presents a summary of our cash flows from operating, investing and financing activities for the periods presented:
| Years Ended December 31, | |||||||||||
| (in thousands) | 2025 | 2024 | |||||||||
| Net cash provided (used in) by: | |||||||||||
| Operating activities | $ | 812 | $ | 4,535 | |||||||
| Investing activities | (5,317) | (10,297) | |||||||||
| Financing activities | 10,499 | 2,290 | |||||||||
| Net increase (decrease) in cash and cash equivalents | $ | 5,994 | $ | (3,472) | |||||||
Net Cash Provided By Operating Activities
Net cash provided by operating activities was $812 compared to $4,535 for the years ended December 31, 2025 and 2024, respectively. The unfavorable change in net cash provided by operating activities of $3,723 was due to an unfavorable change in working capital of $13,855 and the increase in net loss of $5,739, partially offset by the favorable change in noncash accounts of $16,589.
Net Cash Used In Investing Activities
Net cash used in investing activities was $5,317 compared to $10,297 for the year ended December 31, 2024. The favorable change in net cash used in investing activities of $4,980 was primarily due to the increase in proceeds from the sale of investments, net of investment purchases, totaling $6,050, partially offset by a net increase in capital expenditures of $644.
Net Cash Provided by Financing Activities
Net cash provided by financing activities was $10,499 as compared to $2,290 for the year ended December 31, 2024, an increase of $8,209, or 358%. The increase in net cash provided by financing activities was primarily due to an increase of $8,231 in proceeds from the exercise of stock options and Employee Stock Purchase Plan (“ESPP”) purchases year-over-year.
Sources of Capital
Our expected future capital requirements may depend on many factors including expanding our customer base and sales force, the timing and extent of spending in obtaining regulatory approval, strategic opportunities and introduction of new products. Additional sources of liquidity available to us include issuance of additional equity securities through public or private equity offerings, debt financings or from other sources. The sale of additional equity may result in dilution to our shareholders. There is no assurance that we will be able to secure funding on terms acceptable to us, or at all. Should additional capital not become available to us as needed, we may be required to take certain actions, such as slowing sales and marketing expansion, delaying regulatory approvals, or reducing headcount.
Credit Facilities
As of December 31, 2025, we had $50,000 outstanding in indebtedness under the Term Loan Agreement, dated June 30, 2020, with Oberland Capital and its affiliates, TPC Investments II LP and Argo LLC (as amended, the “Credit Facility”); $35,000 maturing on June 30, 2027 and $15,000 maturing on June 30, 2028. Quarterly interest only and revenue participation payments are due through each of the maturity dates. Interest is calculated as 7.5% plus the greater of the forward-looking term rate based on the secured overnight financing rate as set by the Federal Reserve Bank of New York plus 0.10% (“Adjusted SOFR”) or 2.0% (11.59% as of December 31, 2025). Revenue participation payments are calculated as a percentage of our net revenues, up to $70,000 in any given year, adding approximately 1.5% per year of additional interest payments on the outstanding indebtedness. Upon such date early repayment occurs, we are required repay the principal balance and provide a make-whole payment calculated to generate an internal rate of return to the lender equal to 11.5%, less the total of all quarterly interest and revenue participation payments previously paid (the “Make-Whole Payment”). See Note 9 - Long-Term Debt, Net of Debt Discount and Financing Fees and Note 15 - Commitments and Contingencies in the Notes to the Consolidated Financial Statements Part II, Item 8 of this Form 10-K.
63
Axogen, Inc.
Management’s Discussion and Analysis of Financial Condition and Results of Operations - Continued
(in thousands, except share and per share amounts)
Contractual Obligations and Commitments
The following table presents current and long-term material cash requirements as of December 31, 2025:
| (in thousands) | 2026 | 2027-2028 | 2029-2030 | Thereafter | Total | ||||||||||||||||||||||||
Credit Facility principal (1) | $ | — | $ | 50,000 | $ | — | $ | — | $ | 50,000 | |||||||||||||||||||
Credit Facility interest (1)(2) | 5,793 | 4,634 | — | — | 10,427 | ||||||||||||||||||||||||
Credit Facility revenue participation payments (1) | 756 | 987 | — | — | 1,743 | ||||||||||||||||||||||||
Operating and finance lease obligations (3) | 4,294 | 6,252 | 6,287 | 12,285 | 29,118 | ||||||||||||||||||||||||
| $ | 10,843 | $ | 61,873 | $ | 6,287 | $ | 12,285 | $ | 91,288 | ||||||||||||||||||||
__________
(1)On January 28, 2026, all obligations under the Credit Facility, including the Make-Whole Payment, were paid in full with net proceeds from the Offering. See Note 9 - Long-Term Debt, Net of Debt Discount and Financing Fees, Note 15 - Commitments and Contingencies and Note 16 - Subsequent Events in the Notes to the Consolidated Financial Statements in Part II, Item 8 of this Form 10-K.
(2)Calculated at 11.59%; the interest rate as of December 31, 2025.
(3)See Note 8 - Leases in the Notes to the Consolidated Financial Statements in Part II, Item 8 of this Form 10-K.
Critical Accounting Estimates
In preparing our financial statements in accordance with generally accepted accounting principles, there are certain accounting policies, which may require substantial judgment or estimation in their application. We believe these accounting policies and the others set forth in Note 2 - Summary of Significant Accounting Policies in the Notes to the Consolidated Financial Statements in Part II, Item 8 of this Form 10-K are critical to understanding our results of operations and financial condition. Actual results could differ from our estimates and assumptions, and any such differences could be material to our results of operations and financial condition.
Inventories
Description
Inventories consist of purchased materials, direct labor and manufacturing overhead, and are stated at the lower of cost or net realizable value, as determined by the first-in, first-out method.
Judgments and Uncertainties
We maintain reserves for excess and obsolete inventory resulting from the potential inability to sell certain products in excess of current carrying cost. We make estimates regarding the future recoverability of the costs of these products and record provisions based on historical experience, expiration dates and expected future trends.
Sensitivity of Estimate to Change
As of December 31, 2025, we have reserved $2,202 for potential losses relating to inventory. If our actual product life cycles, product demand or acceptance of new product introductions are less favorable than projected by management, additional inventory write downs may be required, which could unfavorably affect future operating results.
Derivative Instruments
Description
We review debt instruments to determine whether there are embedded derivative instruments, which are required to be bifurcated and accounted for separately as a derivative financial instrument. Embedded derivatives that are not clearly and closely related to the debt host are bifurcated and recognized at fair value on the Consolidated Balance Sheets with changes in fair value recognized as either a gain or loss on the Consolidated Statements of Operations for each reporting period.
64
Axogen, Inc.
Management’s Discussion and Analysis of Financial Condition and Results of Operations - Continued
(in thousands, except share and per share amounts)
Judgments and Uncertainties
The fair value of embedded derivatives is measured based on equity markets and interest rates, as well as an estimate of our nonperformance risk adjustment. This estimate includes an option adjusted spread and an estimate of our discount rate.
Sensitivity of Estimate to Change
As of December 31, 2025, we recorded a derivative liability of $3,886. However, if the discount rate were to change by 1%, it would have an approximately $600 effect on our derivative liability fair value.
On January 28, 2026, all obligations under the Credit Facility were paid in full. See Note 9 - Long-Term Debt, Net of Debt Discount and Financing Fees in the Notes to the Consolidated Financial Statements Part II, Item 8 of this Form 10-K for details regarding the termination of the Credit Facility.
Stock-Based Compensation
Description
Stock-based compensation is in the form of stock options, restricted stock units (“RSU”) and performance stock units (“PSU”) granted to employees and directors. Stock-based compensation expense is based on the fair value of the stock options, RSUs and PSUs.
Judgments and Uncertainties
We estimate the grant date fair value of each stock option award on the date of grant using a multiple-point Black-Scholes option-pricing model. In addition, we estimate the grant date fair value of stock options granted to employees at a premium price based on market conditions, such as the trading price of our common stock, using a Monte Carlo simulation option-pricing model. The value of the portion of the award that is ultimately expected to vest is recognized as expense over the requisite service periods.
The fair value of the PSU grants tied to revenue goals (the “Revenue PSUs”) is determined based on the fair value of our common stock on the date of grant and our estimate of achieving the applicable revenue goals. Compensation expense for the Revenue PSUs is recorded ratably over the performance period. The number of shares delivered to recipients and the related compensation cost recognized as an expense will be based on the actual performance metrics as set forth in the applicable Revenue PSU award agreement. The amount actually awarded will be based upon achievement of the performance measures. Expectations related to the achievement of the revenue goals associated with the Revenue PSU grants is assessed at each reporting period and is used to determine whether any of the Revenue PSU grants are expected to vest. If the performance-based milestones related to the Revenue PSU grants are not met or not expected to be met, any compensation expense recognized associated with such grants is reversed.
The grant date fair value of the Total Shareholder Return (“TSR”) PSUs (“TSR PSUs”) is calculated using a Monte Carlo simulation. The number of TSR PSUs that will be earned is based upon the achievement of stock price targets (“Price Targets”) over the measurement period. Compensation expense is recognized regardless if the Price Targets are satisfied. Compensation expense will be reversed if an employee’s employment is terminated prior to satisfying the requisite service period.
The fair value of the PSU grants tied to revenue compounded annual growth rate (“CAGR”) goals, subject to modification based on our TSR relative to the TSR of companies within our industry (“Relative TSR”) over the performance period (the “CAGR TSR PSUs”) is determined based on the fair value of our common stock on the date of grant and our estimate of achieving the applicable revenue CAGR goals. The estimated grant date fair value of the CAGR TSR PSUs is calculated using a Monte Carlo simulation. Compensation expense for the CAGR TSR PSUs is recorded ratably over the performance period. The number of shares delivered to recipients and the related compensation cost recognized as an expense will be based on the actual performance metrics as set forth in the applicable CAGR TSR PSU award agreement. The amount actually awarded will be based upon achievement of the performance measures. Expectations related to the achievement of the revenue CAGR goals associated with the CAGR TSR PSU grants is assessed at each reporting period and is used to determine whether any of the CAGR TSR PSU grants are expected to vest. If the performance-based milestones related to the CAGR TSR PSU grants are not met or not expected to be met, any compensation expense recognized associated with such grants is reversed.
65
Axogen, Inc.
Management’s Discussion and Analysis of Financial Condition and Results of Operations - Continued
(in thousands, except share and per share amounts)
We recognize compensation expense related to the ESPP based on the estimated fair value of the options on the date of grant. We estimate the grant date fair value using a Black-Scholes option pricing model for each purchase period. The grant date fair value is expensed on a straight-line basis over the offering period.
The determination of fair value using option-pricing models, as indicated above, is affected by our stock price, as well as assumptions regarding several subjective variables. These variables include, but are not limited to, our expected stock price volatility over the expected term of the awards. We determine the expected term of each award giving consideration to the contractual terms, vesting schedules, and post-vesting forfeitures. We use the risk-free interest rate on the implied yield available on U.S. Treasury issues with an equivalent remaining term approximately equal to the expected term of the award. The fair value of the Revenue PSUs and CAGR TSR PSUs also includes our estimates of achieving the applicable revenue and revenue CAGR goals. The value of the portion of the award that is ultimately expected to vest is recognized as expense over the requisite service periods in our Consolidated Statements of Operations. If factors change and different assumptions are used, stock-based compensation expense could be materially different in the future.
Sensitivity of Estimate to Change
If we determine that the pay-out range of unvested PSUs outstanding were to change by 50 basis points from expected payout as of December 31, 2025, our stock-based compensation expense would change by $541 for the fiscal year ended December 31, 2025.
Recent Accounting Pronouncements
See Note 2 - Summary of Significant Accounting Policies in the Notes to the Consolidated Financial Statements, Part II Item 8 of this Form 10-K for further information.
Subsequent Events
On January 20, 2026, we entered into a payoff letter (the “Payoff Letter”) with Oberland Capital and its affiliates, TPC Investments II LP and Argo LLC (collectively, the “Lender”). Pursuant to the Payoff Letter, the final payoff amount was approximately $69,707 (the “Payoff Amount”), so long as the payoff date was on or before February 15, 2026. Upon payment of the Payoff Amount on January 28, 2026 and satisfaction of the other conditions specified in the Payoff Letter, all obligations under the Credit Facility, including the Make-Whole Payment, were paid in full, all liens and security interests securing such obligations were released, and the Credit Facility and related loan documents were terminated, subject to certain customary surviving provisions. Payment of the Payoff Amount was funded by proceeds from an equity offering completed by the Company on January 23, 2026. See Note 9 - Long-Term Debt, Net of Debt Discount and Financing Fees and Note 16 - Subsequent Events in the Notes to the Consolidated Financial Statements Part II, Item 8 of this Form 10-K.
On January 21, 2026, we entered into an underwriting agreement (the “Underwriting Agreement”) with Wells Fargo Securities, LLC and Mizuho Securities USA LLC, as representatives of the underwriters (the “Underwriters”). Pursuant to the terms and conditions of the Underwriting Agreement, we agreed to sell 4,000,000 shares of our common stock, $0.01 par value per share, at a public offering price of $31.00 per share, plus an additional 600,000 shares sold pursuant to the Underwriters’ option to purchase additional shares (the “Offering”). The Offering closed on January 23, 2026, with a sale of 4,600,000 shares.
We received net proceeds from the Offering of $133,338, after deducting the underwriting discounts and commissions and expenses in connection with the Offering. Net proceeds from the Offering were used for the early payoff and termination of the Credit Facility, as discussed above. The remaining net proceeds will be used for working capital, capital expenditures and other general corporate purposes. See Note 16 - Subsequent Events in the Notes to the Consolidated Financial Statements Part II, Item 8 of this Form 10-K for additional details regarding the Offering.
66
Item 7A. QUANTITATIVE AND QUALITATIVE DISCLOSURES ABOUT MARKET RISK
Interest Rate Risk
We are subject to interest rate risk from exposure to changes in interest rates based upon our investing and cash management activities. For our cash equivalents and investments, a change in interest rates affects the amount of interest income that can be earned.
We have not entered into derivative transactions related to cash and cash equivalents. We do not expect changes in interest rates to have a material adverse effect on our income or our cash flows in 2026. However, we give no assurance that interest rates will not significantly change in the future.
We also have interest rate exposure as a result of the Credit Facility. As of December 31, 2025, the outstanding principal amount of our loans under the Credit Facility was $50,000. Interest on our loans under the Credit Facility is payable quarterly during the term of the loans and is calculated as 7.5% plus the greater of Adjusted SOFR or 2.0% (11.59% as of December 31, 2025); provided that the interest rate shall never be less than 9.5%.
On January 28, 2026, all obligations under the Credit Facility were paid in full. See Note 9 - Long-Term Debt, Net of Debt Discount and Financing Fees in the Notes to the Consolidated Financial Statements Part II, Item 8 of this Form 10-K for details regarding the termination of the Credit Facility.
Credit Risk
Financial instruments that potentially subject us to credit risk consist of cash and cash equivalent balances, investments in U.S. Treasuries and accounts receivable. Certain of our cash and cash equivalents balances exceed Federal Deposit Insurance Corporation (“FDIC”) insured limits or are invested in money market accounts with investment banks that are not FDIC-insured. As of December 31, 2025, $35,048 of the cash and cash equivalents balance was in excess of FDIC limits or not FDIC-insured.
We invest our cash primarily in money market accounts and in U.S. Treasuries. We believe our cash is invested in a conservative manner, with cash preservation being the primary investment objective.
With respect to accounts receivable, we perform credit evaluations of our customers and do not require collateral. There have been no material losses on accounts receivable. Concentrations of credit risk with respect to accounts receivable are limited because a large number of geographically diverse customers make up our customer base, thus spreading the trade credit risk. We also control credit risk through credit approvals and monitoring procedures.
Foreign Currency Exchange Risk
The value of the U.S. dollar compared to the foreign currencies of the countries where we distribute our products has little to no effect on our financial results. In our international markets, we distribute our products and services to independent distributors who, in turn, distribute and market to medical clinics. The revenue from the distribution of our products in our international markets through independent distributors is denominated in U.S. dollars. As a result, we have minimal exposure related to foreign exchange rate fluctuations. Our portfolio of products is currently available in the U.S., Canada, Germany, the U.K., Spain and several other countries.
67
ITEM 8. FINANCIAL STATEMENTS AND SUPPLEMENTARY DATA
| Table of Contents | |||||
| Page | |||||
REPORT OF INDEPENDENT REGISTERED PUBLIC ACCOUNTING FIRM (PCAOB ID No. | |||||
68
REPORT OF INDEPENDENT REGISTERED PUBLIC ACCOUNTING FIRM
To the shareholders and the Board of Directors of Axogen, Inc.
Opinions on the Financial Statements and Internal Control over Financial Reporting
We have audited the accompanying consolidated balance sheets of Axogen, Inc. and subsidiaries (the “Company”) as of December 31, 2025 and 2024, the related consolidated statements of operations, shareholders’ equity, and cash flows, for each of the three years in the period ended December 31, 2025, and the related notes and the schedule listed in the Index at Item 15a (collectively referred to as the “financial statements”). We also have audited the Company’s internal control over financial reporting as of December 31, 2025, based on criteria established in Internal Control — Integrated Framework (2013) issued by the Committee of Sponsoring Organizations of the Treadway Commission (COSO).
In our opinion, the financial statements referred to above present fairly, in all material respects, the financial position of the Company as of December 31, 2025 and 2024 and the results of its operations and its cash flows for each of the three years in the period ended December 31, 2025, in conformity with accounting principles generally accepted in the United States of America. Also, in our opinion, the Company maintained, in all material respects, effective internal control over financial reporting as of December 31, 2025, based on criteria established in Internal Control — Integrated Framework (2013) issued by COSO.
Basis for Opinions
The Company’s management is responsible for these financial statements, for maintaining effective internal control over financial reporting, and for its assessment of the effectiveness of internal control over financial reporting, included in the accompanying Management’s Annual Report on Internal Control Over Financial Reporting. Our responsibility is to express an opinion on these financial statements and an opinion on the Company’s internal control over financial reporting based on our audits. We are a public accounting firm registered with the Public Company Accounting Oversight Board (United States) (PCAOB) and are required to be independent with respect to the Company in accordance with the U.S. federal securities laws and the applicable rules and regulations of the Securities and Exchange Commission and the PCAOB.
We conducted our audits in accordance with the standards of the PCAOB. Those standards require that we plan and perform the audits to obtain reasonable assurance about whether the financial statements are free of material misstatement, whether due to error or fraud, and whether effective internal control over financial reporting was maintained in all material respects.
Our audits of the financial statements included performing procedures to assess the risks of material misstatement of the financial statements, whether due to error or fraud, and performing procedures to respond to those risks. Such procedures included examining, on a test basis, evidence regarding the amounts and disclosures in the financial statements. Our audits also included evaluating the accounting principles used and significant estimates made by management, as well as evaluating the overall presentation of the financial statements. Our audit of internal control over financial reporting included obtaining an understanding of internal control over financial reporting, assessing the risk that a material weakness exists, and testing and evaluating the design and operating effectiveness of internal control based on the assessed risk. Our audits also included performing such other procedures as we considered necessary in the circumstances. We believe that our audits provide a reasonable basis for our opinions.
Definition and Limitations of Internal Control over Financial Reporting
A company’s internal control over financial reporting is a process designed to provide reasonable assurance regarding the reliability of financial reporting and the preparation of financial statements for external purposes in accordance with generally accepted accounting principles. A company’s internal control over financial reporting includes those policies and procedures that (1) pertain to the maintenance of records that, in reasonable detail, accurately and fairly reflect the transactions and dispositions of the assets of the company; (2) provide reasonable assurance that transactions are recorded as necessary to permit preparation of financial statements in accordance with generally accepted accounting principles, and that receipts and expenditures of the company are being made only in accordance with authorizations of management and directors of the company; and (3) provide reasonable assurance regarding prevention or timely detection of unauthorized acquisition, use, or disposition of the company’s assets that could have a material effect on the financial statements.
Because of its inherent limitations, internal control over financial reporting may not prevent or detect misstatements. Also, projections of any evaluation of effectiveness to future periods are subject to the risk that controls may become inadequate because of changes in conditions, or that the degree of compliance with the policies or procedures may deteriorate.
69
Critical Audit Matter
The critical audit matter communicated below is a matter arising from the current-period audit of the financial statements that was communicated or required to be communicated to the audit committee and that (1) relates to accounts or disclosures that are material to the financial statements and (2) involved our especially challenging, subjective, or complex judgments. The communication of critical audit matters does not alter in any way our opinion on the financial statements, taken as a whole, and we are not, by communicating the critical audit matter below, providing a separate opinion on the critical audit matter or on the accounts or disclosures to which it relates.
Inventory – Excess and Obsolete (E&O) Inventory — Refer to Notes 2 and 3 to the financial statements
Critical Audit Matter Description
Inventories, consisting of materials, direct labor and manufacturing overhead, are stated at the lower of cost or net realizable value. At each balance sheet date, the Company evaluates inventories for excess quantities, obsolescence or shelf-life.
The Company monitors the shelf life of its products and historical expiration and spoilage trends, and reserves for inventory based on the estimated amount of inventory that will not be distributed before expiration or spoilage. To estimate the amount of inventory that will expire or spoil prior to being distributed, the Company reviews inventory quantities on hand, historical and projected distribution levels, historical expiration trends, and historical spoilage trends. The Company’s calculation of the amount of inventory that will expire prior to distribution has three components: 1) a spoilage-based component that compares historical spoilage rates to inventory quantities on hand 2) a demand or consumption-based component that compares projected distribution to inventory quantities on hand; and 3) an expiring inventory component that assesses the risk related to inventory that is near expiration.
Given the significant judgments associated with evaluating the valuation of E&O inventory, auditing the reasonableness of management’s estimates and assumptions involved especially subjective judgment and an increased extent of effort.
How the Critical Audit Matter Was Addressed in the Audit
Our audit procedures related to the Company’s valuation of E&O inventory included the following, among others:
•We tested the design, implementation and operating effectiveness of controls over the E&O inventory valuation.
•We evaluated management’s ability to accurately forecast E&O inventory by comparing the historical inventory reserve estimates to subsequent inventory destructions and expirations.
•We evaluated the reasonableness of the Company’s E&O valuation methodology and tested the mathematical accuracy of the calculation.
•We tested the accuracy and completeness of the underlying data used in the calculation of the Company’s expiring inventory and spoilage models.
•We made inquiries of the Company’s employees outside of the accounting department and evaluated other areas of the audit to identify business, product, or industry changes that may impact the inputs in the inventory valuation calculation.
/s/ Deloitte & Touche LLP
February 24, 2026
We have served as the Company’s auditor since 2018.
70
Axogen, Inc.
Consolidated Balance Sheets
December 31, 2025 and 2024
(in thousands, except share and per share amounts)
| December 31, 2025 | December 31, 2024 | ||||||||||
| Assets | |||||||||||
| Current assets: | |||||||||||
| Cash and cash equivalents | $ | $ | |||||||||
| Restricted cash | |||||||||||
| Investments | |||||||||||
Accounts receivable, net of allowance for doubtful accounts of $ | |||||||||||
| Inventory | |||||||||||
| Prepaid expenses and other assets | |||||||||||
| Total current assets | |||||||||||
| Property and equipment, net | |||||||||||
| Operating lease right-of-use assets | |||||||||||
| Intangible assets, net | |||||||||||
| Total assets | $ | $ | |||||||||
| Liabilities and shareholders’ equity | |||||||||||
| Current liabilities: | |||||||||||
| Accounts payable and accrued expenses | $ | $ | |||||||||
| Current maturities of long-term lease obligations | |||||||||||
| Total current liabilities | |||||||||||
| Long-term debt, net of debt discount and financing fees | |||||||||||
| Long-term lease obligations | |||||||||||
| Debt derivative liabilities | |||||||||||
| Other long-term liabilities | |||||||||||
| Total liabilities | |||||||||||
| Commitments and contingencies - see Note 15 | |||||||||||
| Shareholders’ equity: | |||||||||||
Common stock, $ | |||||||||||
| Additional paid-in capital | |||||||||||
| Accumulated deficit | ( | ( | |||||||||
| Total shareholders’ equity | |||||||||||
| Total liabilities and shareholders’ equity | $ | $ | |||||||||
The accompanying notes are an integral part of these consolidated financial statements.
71
Axogen, Inc.
Consolidated Statements of Operations
Years ended December 31, 2025, 2024 and 2023
(in thousands, except share and per share amounts)
| Years Ended December 31, | |||||||||||||||||
| 2025 | 2024 | 2023 | |||||||||||||||
| Revenues | $ | $ | $ | ||||||||||||||
| Cost of goods sold | |||||||||||||||||
| Gross profit | |||||||||||||||||
| Costs and expenses: | |||||||||||||||||
| Sales and marketing | |||||||||||||||||
| Research and development | |||||||||||||||||
| General and administrative | |||||||||||||||||
| Total costs and expenses | |||||||||||||||||
| Loss from operations | ( | ( | ( | ||||||||||||||
| Other income (expense): | |||||||||||||||||
| Investment income | |||||||||||||||||
| Interest expense | ( | ( | ( | ||||||||||||||
| Change in fair value of debt derivative liabilities | ( | ||||||||||||||||
| Other income (expense), net | ( | ( | |||||||||||||||
| Total other expense, net | ( | ( | ( | ||||||||||||||
| Net loss | $ | ( | $ | ( | $ | ( | |||||||||||
| Weighted average common shares outstanding — basic and diluted | |||||||||||||||||
| Net loss per common share — basic and diluted | $ | ( | $ | ( | $ | ( | |||||||||||
The accompanying notes are an integral part of these consolidated financial statements.
72
Axogen, Inc.
Consolidated Statements of Changes in Shareholders’ Equity
Years ended December 31, 2025, 2024 and 2023
(in thousands, except share amounts)
| Common Stock | Additional Paid-in Capital | Accumulated Deficit | Total Shareholders’ Equity | ||||||||||||||||||||||||||
| Shares | Amount | ||||||||||||||||||||||||||||
| Balance, December 31, 2022 | $ | $ | $ | ( | $ | ||||||||||||||||||||||||
| Stock-based compensation | — | — | — | ||||||||||||||||||||||||||
| Issuance of restricted and performance stock units | ( | — | |||||||||||||||||||||||||||
| Exercise of stock options and employee stock purchases under the ESPP | — | ||||||||||||||||||||||||||||
| Net loss | — | — | — | ( | ( | ||||||||||||||||||||||||
| Balance, December 31, 2023 | ( | ||||||||||||||||||||||||||||
| Stock-based compensation | — | — | — | ||||||||||||||||||||||||||
| Issuance of restricted and performance stock units | ( | — | |||||||||||||||||||||||||||
| Exercise of stock options and employee stock purchases under the ESPP | — | ||||||||||||||||||||||||||||
| Net loss | — | — | — | ( | ( | ||||||||||||||||||||||||
| Balance, December 31, 2024 | ( | ||||||||||||||||||||||||||||
| Stock-based compensation | — | — | — | ||||||||||||||||||||||||||
| Issuance of restricted and performance stock units | ( | — | |||||||||||||||||||||||||||
| Exercise of stock options and employee stock purchases under the ESPP | — | ||||||||||||||||||||||||||||
| Net loss | — | — | — | ( | ( | ||||||||||||||||||||||||
| Balance, December 31, 2025 | $ | $ | $ | ( | $ | ||||||||||||||||||||||||
The accompanying notes are an integral part of these consolidated financial statements.
73
Axogen, Inc.
Consolidated Statements of Cash Flows
Years ended December 31, 2025, 2024 and 2023
(in thousands)
| Years Ended December 31, | |||||||||||||||||
| 2025 | 2024 | 2023 | |||||||||||||||
| Cash flows from operating activities: | |||||||||||||||||
| Net loss | $ | ( | $ | ( | $ | ( | |||||||||||
| Adjustments to reconcile net loss to net cash provided by (used in) operating activities: | |||||||||||||||||
| Depreciation | |||||||||||||||||
| Amortization of right-of-use assets | |||||||||||||||||
| Amortization of intangible assets | |||||||||||||||||
| Amortization of debt discount and deferred financing fees | |||||||||||||||||
| Loss on disposal of equipment | |||||||||||||||||
| Provision for (recovery of) bad debts | ( | ||||||||||||||||
| Investment gains, net | ( | ( | ( | ||||||||||||||
| Change in fair value of debt derivative liabilities | ( | ( | |||||||||||||||
| Impairment of long-lived assets | |||||||||||||||||
| Stock-based compensation expense | |||||||||||||||||
| Change in operating assets and liabilities: | |||||||||||||||||
| Accounts receivable | ( | ( | |||||||||||||||
| Inventory | ( | ( | ( | ||||||||||||||
| Prepaid expenses and other assets | ( | ( | |||||||||||||||
| Accounts payable and accrued expenses | ( | ||||||||||||||||
| Operating lease obligations | ( | ( | ( | ||||||||||||||
| Cash paid for interest portion of financing lease obligations | ( | ( | ( | ||||||||||||||
| Other long-term liabilities | ( | ||||||||||||||||
| Net cash provided by (used in) operating activities | ( | ||||||||||||||||
| Cash flows from investing activities: | |||||||||||||||||
| Purchase of property and equipment | ( | ( | ( | ||||||||||||||
| Purchase of investments | ( | ( | ( | ||||||||||||||
| Proceeds from sale of investments | |||||||||||||||||
| Cash payments for intangible assets | ( | ( | ( | ||||||||||||||
| Net cash (used in) provided by investing activities | ( | ( | |||||||||||||||
| Cash flows from financing activities: | |||||||||||||||||
| Cash paid for debt portion of financing lease obligations | ( | ( | ( | ||||||||||||||
| Proceeds from exercise of stock options and ESPP stock purchases | |||||||||||||||||
| Net cash provided by financing activities | |||||||||||||||||
| Net increase (decrease) in cash and cash equivalents, and restricted cash | ( | ||||||||||||||||
| Cash and cash equivalents, and restricted cash, beginning of period | |||||||||||||||||
| Cash and cash equivalents, and restricted cash, end of period | $ | $ | $ | ||||||||||||||
| Supplemental disclosures of cash flow activity: | |||||||||||||||||
| Cash paid for interest, net of capitalized interest | $ | $ | $ | ||||||||||||||
| Supplemental disclosure of noncash investing and financing activities: | |||||||||||||||||
| Acquisition of property and equipment change in accounts payable and accrued expenses | $ | ( | $ | $ | |||||||||||||
| Obtaining a right-of-use asset in exchange for a lease liability | $ | $ | $ | ||||||||||||||
| Acquisition of intangible assets change in accounts payable and accrued expenses | $ | ( | $ | $ | |||||||||||||
The accompanying notes are an integral part of these consolidated financial statements.
74
Axogen, Inc.
Notes to Consolidated Financial Statements
Years ended December 31, 2025, 2024 and 2023
(in thousands, except share and per share amounts)
1.Nature of Business
Axogen, Inc. (together with its wholly-owned subsidiaries, the “Company”) was incorporated in Minnesota. The Company’s business is focused on the science, development and commercialization of the technologies used for peripheral nerve regeneration and repair. The Company’s products include AVANCE® (acellular nerve allograft-arwx), Avance® Nerve Graft, Axoguard Nerve Connector®, Axoguard Nerve Protector®, Axoguard HA+ Nerve Protector™, Axoguard Nerve Cap®, and Avive+ Soft Tissue Matrix™. The Company is headquartered in Florida. The Company has processing, warehousing, and distribution facilities in Ohio and Texas.
2. Summary of Significant Accounting Policies
Basis of Presentation
Use of Estimates
The preparation of consolidated financial statements in conformity with U.S. GAAP requires management to make estimates and assumptions that affect the reported amounts of assets and liabilities and the disclosure of contingent assets and liabilities as of the date of the financial statements and the reported amounts of revenues and expenses during the reporting period. The significant estimates affecting the amounts reported or disclosed in the consolidated financial statements include the realizable value of inventories, the valuation of stock-based compensation, the valuation of derivative instruments, and the fair value of debt instruments. Other estimates that affect the amounts reported or disclosed in the consolidated financial statements include the allowance for doubtful accounts, the useful life and recoverability of long-lived assets, incremental borrowing rates for operating leases, and accounting for income taxes, including the realizability of deferred tax assets and the related valuation allowance. The Company bases its estimates on historical and anticipated results, trends, and various other assumptions that management believes are reasonable under the circumstances, including assumptions as to future events. Actual results may differ from those estimates.
Risk and Uncertainties
The Company is dependent on its suppliers, including single source suppliers, some of which are outside of the U.S., and the inability of these suppliers to deliver necessary components of their products in a timely manner at prices, quality levels and volumes acceptable to the Company, or the Company’s inability to effectively manage these components from its suppliers, could have a material adverse effect on the Company’s business, financial condition and operating results.
Cash and Cash Equivalents
Cash and cash equivalents consist of short-term, highly liquid investments with original maturities of three months or less from the date of acquisition. Certain of the Company’s cash and cash equivalents balances exceed Federal Deposit Insurance Corporation (“FDIC”) insured limits or are invested in money market accounts with investment banks that are not FDIC-insured. The Company places its cash and cash equivalents in what they believe to be credit-worthy financial institutions. As of December 31, 2025, $35,048 of the cash and cash equivalents balance was in excess of FDIC limits or not FDIC-insured.
Restricted Cash
75
Axogen, Inc.
Notes to Consolidated Financial Statements - Continued
Years ended December 31, 2025, 2024 and 2023
(in thousands, except share and per share amounts)
The following table provides a reconciliation of cash and cash equivalents, and restricted cash reported on the Consolidated Balance Sheets that sum to the total of the same reported on the Consolidated Statements of Cash Flows as of the periods presented:
| (in thousands) | December 31, 2025 | December 31, 2024 | |||||||||
| Cash and cash equivalents | $ | $ | |||||||||
| Restricted cash | |||||||||||
| Total cash and cash equivalents, and restricted cash shown on the Consolidated Statements of Cash Flows | $ | $ | |||||||||
Investments
Investments, consisting of U.S. Treasuries, are classified as available-for-sale and have maturities less than one year as of December 31, 2025. Investments are carried at fair value based upon quoted market prices. The Company elected the fair value option (“FVO”) for all of its available-for-sale investments. The FVO election results in all changes in unrealized gains and losses being included in Investment income on the Consolidated Statements of Operations.
Accounts Receivable and Allowance for Doubtful Accounts
Accounts receivable are recorded at invoiced amounts and do not bear interest. The Company grants credit to customers in the normal course of business, but generally does not require collateral or other security to support its receivables.
Concentration Risk
Credit Risk
Financial instruments, which potentially subject the Company to concentrations of credit risk, consist principally of cash and cash equivalents, which are held at major financial institutions, and trade receivables.
None of the Company’s customers accounted for 10% or more of the consolidated revenues or accounts receivable during the years ended December 31, 2025, 2024 and 2023.
Inventory
Inventory, consisting of materials, direct labor, and manufacturing overhead, are stated at the lower of cost or net realizable value. At each balance sheet date, the Company evaluates inventory for excess quantities, obsolescence or shelf life.
The Company monitors the shelf life of its products and historical expiration and spoilage trends, and reserves for inventory based on the estimated amount of inventory that will not be distributed before expiration or spoilage. To estimate the amount of inventory that will expire or spoil prior to being distributed, the Company reviews inventory quantities on hand, historical and projected distribution levels, historical expiration trends, and historical spoilage trends. The Company’s calculation of the amount of inventory that will expire prior to distribution has three components: (i) a spoilage-based component that compares historical spoilage rates to inventory quantities on hand, (ii) a demand or consumption-based component that compares projected distribution to inventory quantities on hand; and (iii) an expiring inventory component that assesses the risk related to inventory that is near expiration. The Company’s model assumes that inventory will be distributed on a first-in-first-out basis. Due to the nature of the inventory (surgical implants with expiration dates) and the fact that a
76
Axogen, Inc.
Notes to Consolidated Financial Statements - Continued
Years ended December 31, 2025, 2024 and 2023
(in thousands, except share and per share amounts)
significant portion of the Company’s inventory is at medical facility consignment locations, estimating the amount of spoilage, the amount of inventory that will expire, and the amount of inventory that should be reserved for involves significant judgments and estimates.
Property and Equipment, Net
Property and equipment, net are stated at historical cost less accumulated depreciation and amortization. Additions and improvements that extend the lives of the assets are capitalized, while expenditures for repairs and maintenance are expensed as incurred. Leasehold improvements are amortized on a straight-line basis over the shorter of the asset’s estimated useful life or the remaining lease term. Depreciation is calculated on a straight-line basis over the estimated useful lives of the assets ranging from to 39 years.
Gains or losses on the disposition of property and equipment are recorded in the period incurred and recorded in General and administrative costs and expenses on the Consolidated Statements of Operations.
Capitalized Interest
Intangible Assets, Net
Intangible assets are recorded at cost and include patents and patent application costs and trademarks. Intangible assets with finite lives are amortized on a straight-line basis over their estimated useful lives and reported net of accumulated amortization. Amortization expense is recorded in General and administrative costs and expenses on the Consolidated Statements of Operations. The useful lives of intangible assets are as follows:
•Patents: up to 20 years
•Trademarks: indefinite lived
Impairment of Long-Lived Assets
The Company analyzes long-lived assets (asset groups), including property and equipment and definite-lived intangible assets, for impairment whenever events or changes in circumstances indicate that the carrying amounts may not be recoverable. An impairment is recognized when the estimated undiscounted cash flows generated by those assets is less than the carrying amounts of such assets. If it is determined that a long-lived asset (asset groups) is not recoverable, an impairment loss would be calculated based on the excess of the carrying value of the long-lived asset (asset groups) over the fair value of the long-lived asset (asset groups). During the year ended December 31, 2025, the Company had long-lived asset impairment of $64 . There were no
Global Nerve Foundation
Periodically, the Company may make contributions to the Global Nerve Foundation (“GNF”), a related party, due to certain executives of the Company being members of GNF’s board of directors. The GNF was incorporated in 2021 exclusively for charitable, educational, and scientific purposes and qualifies under Internal Revenue Code 501(c)(3) as an exempt private foundation. Under its charter, the GNF engages in activities that focus on improving the awareness and care of patients with
77
Axogen, Inc.
Notes to Consolidated Financial Statements - Continued
Years ended December 31, 2025, 2024 and 2023
(in thousands, except share and per share amounts)
peripheral nerve injuries through grants, contributions and other appropriate means. The GNF is a separate legal entity and is not a subsidiary of the Company; therefore, its results are not included in the accompanying consolidated financial statements.
On July 20, 2024, the Company entered into a Qualified Founding Partner Agreement (the “Partner Agreement”) which grants certain benefits to the Company related to the GNF’s programming and marketing. The Partner Agreement terminates on December 31, 2031. The Company may terminate the Partner Agreement early by giving notice of termination between January 2 and January 31 in any calendar year; however, if the Company terminates the Partner Agreement early, it will be required to make the next two payments per the payment schedule in a total amount not to exceed $100 . Per the terms of the Partner Agreement, the Company contributed $175 and $100 to the GNF during the years ended December 31, 2025 and 2024, respectively, and will contribute $125 in 2026. These contributions were recorded in Sales and marketing costs and expenses on the Consolidated Statements of Operations.
Fair Value
The Company uses fair value measurements to record fair value adjustments to certain assets and liabilities and to determine fair value disclosures. Cash equivalents, investments, and derivative instruments are recorded at fair value on a recurring basis. Fair value is defined as the price that would be received upon the sale of an asset or paid to transfer a liability in an orderly transaction between market participants on the measurement date. Valuation techniques used to measure fair value maximize the use of observable inputs and minimize the use of unobservable inputs. The fair value hierarchy defines a three-level valuation hierarchy for classification and disclosure of fair value measurements as follows:
Level 1 – Quoted prices in active markets for identical assets or liabilities.
Level 2 – Inputs other than Level 1 that are observable, either directly or indirectly, such as quoted prices for similar assets or liabilities; quoted prices in markets that are not active; or other inputs that are observable or can be corroborated by observable market data for substantially the full term of the assets or liabilities.
Level 3 – Unobservable inputs that are supported by little or no market activity and that are significant to the fair value of the assets or liabilities.
Derivative Instruments
The Company reviews its debt instruments in determining whether there are embedded derivative instruments, which are required to be bifurcated and accounted for separately as a derivative financial instrument. Embedded derivatives that are not clearly and closely related to the debt host are bifurcated and recognized at fair value on the Consolidated Balance Sheets with changes in fair value recognized as either a gain or loss on the Consolidated Statement of Operations for each reporting period. The fair value of embedded derivatives is measured based on equity markets and interest rates, as well as an estimate of the Company’s nonperformance risk adjustment. This estimate includes an option adjusted spread and an estimate of the Company’s risk-free rate.
Leases
The Company determines if a contract contains a lease at the inception date and determines the lease classification, recognition, and measurement at commencement date. All operating lease commitments with a lease term greater than 12 months are recognized as right-of-use assets and obligations on a discounted basis on the Consolidated Balance Sheets. Leases with an initial term of 12 months or less are not recorded on the Consolidated Balance Sheets.
The Company classifies a lease based on whether the arrangement is effectively a purchase of the underlying asset. Leases that transfer the control of the underlying asset are classified as finance leases and all others are classified as operating leases. Interest and amortization expense are recognized for operating leases on a straight-line basis. If a change to the lease term leads to a reassessment of the lease classification and remeasurement, assumptions such as the discount rate and variable rents based on a rate or index will be updated as of the remeasurement date. If an arrangement is modified, the Company will reassess whether the arrangement contains a lease. Any subsequent changes in lease payments are recognized when incurred, unless the change requires a remeasurement of the lease liability.
Certain of the Company’s leases include options for the Company to extend the lease term. The exercise of a lease renewal option is generally at the Company’s sole discretion. Certain of the Company’s lease agreements include provisions for the
78
Axogen, Inc.
Notes to Consolidated Financial Statements - Continued
Years ended December 31, 2025, 2024 and 2023
(in thousands, except share and per share amounts)
Company to reimburse the lessor for common area maintenance, real estate taxes, and insurance, which the Company accounts for as variable lease costs. The Company’s lease agreements do not contain any material residual value guarantees or material restrictive covenants.
The Company accounts for its sublease income on a net basis, recording the sublease income with the operating lease expense on its Consolidated Statements of Operations.
Revenue Recognition
The Company enters into contracts to sell and distribute products and services to hospitals and surgical facilities for use in caring for patients with peripheral nerve damage or transection. Revenue is recognized when the Company transfers control of the products and services to the Company’s customers when the product is shipped or when it is delivered to the customer depending on the agreement. Products are primarily transferred to customer at a point in time.
A portion of the Company’s product revenue is generated from consigned inventory maintained at hospitals and independent sales agencies, and also from inventory physically held by field sales representatives. For these types of product sales, the Company retains control until the product has been used or implanted, at which time revenue is recognized.
In the case of products or services sold to a customer under a distribution or purchase agreement, the customers are granted exclusive distribution rights to sell the implants internationally in a territory defined by the contract. These international distributor agreements contain provisions that allow the Company to terminate the distribution agreement with the distributor, and upon termination, the right to repurchase inventory from the distributor at the distributor’s cost. The Company has determined that its contractual rights to repurchase distributor inventory upon termination of the distributor agreement are not substantive and do not impact the timing of when control transfers; and therefore, the Company has determined it is appropriate to recognize revenue when: (i) the product is shipped via common carrier; or (ii) the product is delivered to the customer or distributor, depending on the terms of the agreement. Determining the timing of revenue recognition for such contracts is subject to judgment because an evaluation must be made regarding the distributor’s ability to direct the use of, and obtain substantially all of the remaining benefits from, the implants received from the Company. Changes in these assessments could have an impact on the timing of revenue recognition from sales to distributors. The Company accounts for shipping and handling activities as a fulfillment cost rather than a separate performance obligation. Amounts billed to customers for shipping and handling are included as part of the transaction price and recognized as revenue when control of the underlying products is transferred to the customer.
The Company operates in a single reportable segment of peripheral nerve repair, offers similar products to its customers, and enters into consistently structured arrangements with similar types of customers. As such, the Company does not disaggregate revenue from contracts with customers as the nature, amount, timing, and uncertainty of revenue and cash flows does not materially differ within and among the contracts with customers.
Government Assistance
Government assistance and grants are recognized when there is reasonable assurance that the Company has met the requirements of the assistance and there is reasonable assurance that the grant will be received. The Company received government grants of $717 and $393 during the years ended December 31, 2024 and 2023, respectively. The Company did not receive any government grants during the year ended December 31, 2025.
79
Axogen, Inc.
Notes to Consolidated Financial Statements - Continued
Years ended December 31, 2025, 2024 and 2023
(in thousands, except share and per share amounts)
the Consolidated Statement of Operations during the year ended December 31, 2024. Deferred grants totaling $131 and $330 were recorded in Accounts payable and accrued expenses as of December 31, 2025 and 2024, respectively, on the Consolidated Balance Sheets.
Cost of Goods Sold
Cost of goods sold includes materials, direct labor, and manufacturing overhead costs related to each product sold or produced, including processing, quality assurance labor, scrap, and inbound freight costs, as well as facility, warehousing and overhead supporting the Company’s manufacturing operations. All of the Company’s manufacturing costs are included in Cost of goods sold on the Consolidated Statements of Operations.
Research and Development Costs and Expenses
Shipping and Handling
Income Taxes
The Company uses the asset and liability method to account for income taxes in accordance with the authoritative guidance for income taxes. Under this method, deferred tax assets and liabilities are determined based on future tax consequences attributable to differences between the financial statement carrying amounts of existing assets and liabilities and their respective tax bases, and tax loss and credit carryforwards. Deferred tax assets and liabilities are measured using enacted tax rates applied to taxable income in the years in which those temporary differences are expected to be recovered or settled. The effect on deferred tax assets and liabilities of a change in tax rates is recognized in income in the period that includes the enactment date. A valuation allowance is established when necessary to reduce deferred tax assets to the amount expected to be realized.
The Company recognizes the effect of income tax positions only if those positions are more likely than not of being sustained. Recognized income tax positions are measured at the largest amount that has a greater than 50% likelihood of being realized. Changes in recognition or measurement are reflected in the period in which the change in judgment occurs.
Stock-Based Compensation
Stock-based compensation is in the form of stock options, restricted stock units (“RSU”), and performance stock units (“PSU”) granted to employees and directors. Stock-based compensation expense is based on the fair value of the stock options, RSUs and PSUs.
The Company estimates the fair value of each stock option award on the date of grant using a multiple-point Black-Scholes option-pricing model. In addition, the Company estimates the grant date fair value of stock options granted to employees at a premium price based on market conditions, such as the trading price of the Company’s common stock, using a Monte Carlo simulation option-pricing model.
The Company estimates the fair value of RSU grants based upon the grant date closing market price of the Company’s common stock.
80
Axogen, Inc.
Notes to Consolidated Financial Statements - Continued
Years ended December 31, 2025, 2024 and 2023
(in thousands, except share and per share amounts)
The fair value of the PSU grants is based on the Company’s closing stock price on the grant date and its estimate of achieving the applicable performance target goals. The number of PSUs that will ultimately be earned is based upon the Company’s performance as measured against specified targets over the measurement period. Expectations related to the achievement of performance goals associated with PSU grants is assessed as of each reporting period and is used to determine whether PSU grants are expected to vest. If performance-based milestones related to PSU grants are not met or not expected to be met, any compensation expense recognized associated with such grants is reversed.
The grant date fair value of the Total Shareholder Return (“TSR”) PSU grants (“TSR PSUs”) is calculated using a Monte Carlo simulation. The number of TSR PSUs that will be earned is based upon the achievement of stock price hurdles (“Price Targets”) over the measurement period. Compensation expense is recognized regardless if the Price Targets are satisfied. Compensation expense is reversed if an employee’s employment is terminated prior to satisfying the requisite service period.
The grant date fair value of the PSU grants tied to revenue compounded annual growth rate (“CAGR”) goals, subject to modification based on the Company’s TSR relative to the TSR of companies within its industry (“Relative TSR”) over the performance period (the “CAGR TSR PSU”) is determined based on the fair value of our common stock on the date of grant and our estimate of achieving the applicable revenue CAGR goals. The estimated grant date fair value of the CAGR TSR PSUs is calculated using a Monte Carlo simulation. Compensation expense for the CAGR TSR PSUs is recorded ratably over the performance period. The number of shares delivered to recipients and the related compensation cost recognized as an expense will be based on the actual performance metrics as set forth in the applicable CAGR TSR PSU award agreements. If the performance-based milestones related to the CAGR TSR PSU grants are not met or not expected to be met, any compensation expense recognized associated with such grants is reversed.
The Company recognizes expense for all stock-based compensation awards, including stock options, RSUs, and PSUs granted to employees eligible for retirement, as defined within the award notice and allowing for continued vesting post-retirement, over the retirement notice period and continuously updates its estimate of expense over the notice period each reporting period if a retirement notice has not been provided.
The Company recognizes compensation expense related to the Employee Stock Purchase Plan (“ESPP”) based on the estimated fair value of the options on the date of grant. The Company estimates the grant date fair value, and the resulting stock-based compensation expense, using a Black-Scholes option pricing model for each purchase period. The grant date fair value is expensed on a straight-line basis over the offering period.
The determination of fair value using option-pricing models, as indicated above, is affected by the Company’s stock price, as well as assumptions regarding several subjective variables. These variables include, but are not limited to, the Company’s expected stock price volatility over the expected term of the awards. The Company determines the expected term of each award giving consideration to the contractual terms, vesting schedules, and post-vesting forfeitures. The Company uses the risk-free interest rate on the implied yield available on U.S. Treasury issues with an equivalent remaining term approximately equal to the expected term of the award. The value of the portion of the award that is ultimately expected to vest is recognized as expense over the requisite service periods on the Consolidated Statements of Operations. The expense is reduced for forfeitures as they occur.
Net Loss Per Share
Basic net loss per common share is computed by dividing reported net loss by the weighted average number of common shares outstanding during the period without consideration of potentially dilutive securities. Diluted net loss per share reflects the potential dilution that could occur if contracts to issue common stock were exercised or converted into common stock of the Company. Diluted net loss per share is the same as basic net loss per common share for all periods presented, since the effect of the potentially dilutive securities are anti-dilutive. Potential dilutive common share equivalents consist of the incremental common shares issuable upon exercise of vested stock options, RSUs, and PSUs.
Recently Issued Accounting Pronouncements
In November 2024, the Financial Accounting Standards Board (“FASB”) issued Accounting Standards (“ASU”) Update 2024-03 — Income Statement - Reporting Comprehensive Income - Expense Disaggregation Disclosures (Subtopic 220-40) — Disaggregation of Income Statement Expenses (“ASU 2024-03”), and in January 2025, the FASB issued ASU No. 2025-01, Income Statement - Reporting Comprehensive Income - Expense Disaggregation Disclosures (Subtopic 220-40): Clarifying the Effective Date (“ASU 2025-01”). ASU 2024-03 requires additional disclosure of the nature of expenses included in the income
81
Axogen, Inc.
Notes to Consolidated Financial Statements - Continued
Years ended December 31, 2025, 2024 and 2023
(in thousands, except share and per share amounts)
statement as well as disclosures about specific types of expenses included in the expense captions presented in the income statement. ASU 2024-03, as clarified by ASU 2025-01, is effective for annual periods beginning after December 15, 2026 and interim reporting periods beginning after December 15, 2027. Both early adoption and retrospective application are permitted. The Company expects to enhance annual expense disclosures based on the new requirements.
In September 2025, the FASB issued ASU 2025-06 — Intangibles - Goodwill and Other - Internal-Use Software (Subtopic 350-40): Targeted Improvements to the Accounting for Internal-Use Software (“ASU 2025-06”). ASU 2025-06 was issued to modernize the accounting for software costs that are accounted for under Subtopic 350-40, including removing reference to “project stages” and adding the “probable-to-complete recognition threshold.” ASU 2025-06 is effective for annual periods beginning after December 15, 2027, and interim reporting periods within those annual reporting periods, with early adoption permitted. The Company is currently evaluating these new requirements.
In December 2025, the FASB issued ASU 2025-11 — Interim Reporting (Topic 270): Narrow-Scope Improvements (“ASU 2025-11”). ASU 2025-11 clarifies the guidance in Accounting Standards Codification (“ASC 270”), adding a comprehensive list of required interim disclosures and a principle that requires entities to disclose events since the end of the last annual reporting period that have a material impact on the entity. ASU 2025-11 is effective for interim periods within annual periods beginning after December 15, 2027, with early adoption permitted. The Company is currently evaluating these new disclosure requirements.
Recently Adopted Accounting Pronouncements
In December 2023, the FASB issued ASU 2023-09 — Income Taxes (Topic 740) - Improvements to Income Tax Disclosures (“ASU 2023-09”). The new guidance provides for disclosure on an annual basis of the following: (i) specific categories in the rate reconciliation and (ii) additional information for reconciling items that meet a quantitative threshold of greater than 5% of the amount computed by multiplying pretax income (or loss) by the applicable statutory income tax rate. The Company adopted ASU 2023-09 prospectively effective December 31, 2025. See Note 12 - Income Taxes for income tax reporting disclosures based on the new ASU 2023-09 requirements.
All other ASUs issued and not yet effective as of December 31, 2025, and through the date of this report, were assessed and determined to be either not applicable or are expected to have minimal impact on the Company’s current or future financial position or results of operations.
3. Inventory
Inventory consists of the following as of the periods presented:
| (in thousands) | December 31, 2025 | December 31, 2024 | |||||||||
| Finished goods | $ | $ | |||||||||
| Work in process | |||||||||||
| Raw materials | |||||||||||
| Inventory | $ | $ | |||||||||
The provision for inventory write-down is as follows for the periods presented:
| Years Ended December 31, | |||||||||||||||||
| (in thousands) | 2025 | 2024 | 2023 | ||||||||||||||
| Provision for inventory write-down | $ | $ | $ | ||||||||||||||
As of December 31, 2025 and 2024, the Company reserved $2,202 and $1,630 , respectively, for potential losses relating to inventory.
82
Axogen, Inc.
Notes to Consolidated Financial Statements - Continued
Years ended December 31, 2025, 2024 and 2023
(in thousands, except share and per share amounts)
4.Property and Equipment, Net
Property and equipment, net consist of the following as of the periods presented:
| (in thousands) | December 31, 2025 | December 31, 2024 | |||||||||
| Land | $ | $ | |||||||||
| Building | |||||||||||
| Leasehold improvements | |||||||||||
| Processing equipment | |||||||||||
| Furniture and equipment | |||||||||||
| Finance lease right-of-use assets | |||||||||||
| Projects in process | |||||||||||
| Property and equipment, at cost | |||||||||||
| Less: accumulated depreciation and amortization | ( | ( | |||||||||
| Property and equipment, net | $ | $ | |||||||||
Depreciation expense is as follows for the periods presented:
| Years Ended December 31, | |||||||||||||||||
| (in thousands) | 2025 | 2024 | 2023 | ||||||||||||||
| Depreciation expense | $ | $ | $ | ||||||||||||||
5. Intangible Assets, Net
Intangible assets consist of the following as of the periods presented:
| December 31, 2025 | December 31, 2024 | ||||||||||||||||||||||||||||||||||
| (in thousands) | Gross Carrying Amount | Accumulated Amortization | Net Carrying Amount | Gross Carrying Amount | Accumulated Amortization | Net Carrying Amount | |||||||||||||||||||||||||||||
| Amortizable intangible assets: | |||||||||||||||||||||||||||||||||||
| Patents | $ | $ | ( | $ | $ | $ | ( | $ | |||||||||||||||||||||||||||
| Unamortized intangible assets: | |||||||||||||||||||||||||||||||||||
| Trademarks | — | — | |||||||||||||||||||||||||||||||||
| Total intangible assets | $ | $ | ( | $ | $ | $ | ( | $ | |||||||||||||||||||||||||||
The amortization expense is as follows for the periods presented:
Years Ended December 31, | |||||||||||||||||
| (in thousands) | 2025 | 2024 | 2023 | ||||||||||||||
| Amortization expense | $ | $ | $ | ||||||||||||||
83
Axogen, Inc.
Notes to Consolidated Financial Statements - Continued
Years ended December 31, 2025, 2024 and 2023
(in thousands, except share and per share amounts)
As of December 31, 2025, future amortization of patents is as follows:
| Years Ending December 31, | (in thousands) | ||||
| 2026 | $ | ||||
| 2027 | |||||
| 2028 | |||||
| 2029 | |||||
| 2030 | |||||
| Thereafter | |||||
| Total | $ | ||||
License Agreements
The Company had multiple license agreements with the University of Florida Research Foundation and the University of Texas at Austin (the “License Agreements”) in which the Company acquired exclusive worldwide licenses for underlying technology used in repairing and regenerating nerves. The licensed technologies included the rights to issue patents and patents pending in the U.S. and international markets. The License Agreement with the University of Texas expired in September 2023 and the royalty obligations associated with the License Agreement with the University of Florida Research Foundation expired in December 2023.
The Company paid royalty fees ranging from 1 % to 3 % under the License Agreements based on net sales of licensed products. Also, when the Company paid royalties to more than one licensor for sales of the same product, a royalty stack cap applied, capping total royalties at 3.75 %.
6. Fair Value Measurement
The following tables represent the Company’s fair value hierarchy for its financial assets and liabilities measured at fair value on a recurring basis as of the periods presented:
| December 31, 2025 | |||||||||||||||||||||||
| (in thousands) | Level 1 | Level 2 | Level 3 | Total | |||||||||||||||||||
| Assets: | |||||||||||||||||||||||
| Money market funds | $ | $ | $ | $ | |||||||||||||||||||
| U.S. Treasuries | |||||||||||||||||||||||
| Total assets | $ | $ | $ | $ | |||||||||||||||||||
| Liabilities: | |||||||||||||||||||||||
| Debt derivative liabilities | $ | $ | $ | $ | |||||||||||||||||||
84
Axogen, Inc.
Notes to Consolidated Financial Statements - Continued
Years ended December 31, 2025, 2024 and 2023
(in thousands, except share and per share amounts)
| December 31, 2024 | |||||||||||||||||||||||
| (in thousands) | Level 1 | Level 2 | Level 3 | Total | |||||||||||||||||||
| Assets: | |||||||||||||||||||||||
| Money market funds | $ | $ | $ | $ | |||||||||||||||||||
| U.S. Treasuries | |||||||||||||||||||||||
| Total assets | $ | $ | $ | $ | |||||||||||||||||||
| Liabilities: | |||||||||||||||||||||||
| Debt derivative liability | $ | $ | $ | $ | |||||||||||||||||||
The changes in Level 3 liabilities measured at fair value on a recurring basis are as follows for the periods presented:
| (in thousands) | Debt Derivative Liabilities | ||||
| Balance, December 31, 2023 | $ | ||||
| Change in fair value included in net loss | ( | ||||
| Balance, December 31, 2024 | |||||
| Change in fair value included in net loss | |||||
| Balance, December 31, 2025 | $ | ||||
There were no changes in the levels or methodology of the measurement of financial assets or liabilities during the years ended December 31, 2025 and 2024.
The debt derivative liabilities are measured using a ‘with and without’ valuation model to compare the fair value of each tranche of the credit facility the Company has with Oberland Capital and its affiliates, TPC Investments II LP and Argo LLC (“Credit Facility”) including the identified embedded derivative feature and the fair value of a plain vanilla note with the same terms. The fair value of the Credit Facility including the embedded derivative features was determined using a probability-weighted expected return model based on three potential settlement scenarios for the Credit Facility included in the tables below. The estimated settlement value of each scenario, which would include any required make-whole payment (see Note 9 - Long-Term Debt, Net of Debt Discount and Financing Fees), is then discounted to present value using a discount rate that is derived based on the initial terms of the Credit Facility at issuance and corroborated utilizing a synthetic credit rating analysis.
The significant inputs, as of the periods presented, that are included in the valuation of the debt derivative liability - first tranche include:
| Input | December 31, 2025 | December 31, 2024 | |||||||||||||||
| Remaining term (years) | |||||||||||||||||
| Maturity date | June 30, 2027 | June 30, 2027 | |||||||||||||||
| Coupon rate | |||||||||||||||||
| Revenue participation payments | Maximum each year | Maximum each year | |||||||||||||||
| Discount rate | (1) | % | (1) | ||||||||||||||
| Probability of mandatory prepayment after 2025 | (1) | (1) | |||||||||||||||
| Estimated timing of mandatory prepayment event after 2025 | March 31, 2026 | (1) | March 31, 2026 | (1) | |||||||||||||
| Probability of optional prepayment event | (1) | (1) | |||||||||||||||
| Estimated timing of optional prepayment event | January 31, 2026 | (1) | December 31, 2025 | (1) | |||||||||||||
Probability of note held-to-maturity (2) | (1) | (1) | |||||||||||||||
__________
(1)Represents a significant unobservable input.
(2)See Maturity date in table.
85
Axogen, Inc.
Notes to Consolidated Financial Statements - Continued
Years ended December 31, 2025, 2024 and 2023
(in thousands, except share and per share amounts)
The significant inputs, as of the periods presented, that are included in the valuation of the debt derivative liability - second tranche include:
| Input | December 31, 2025 | December 31, 2024 | |||||||||||||||
| Remaining term (years) | |||||||||||||||||
| Maturity date | June 30, 2028 | June 30, 2028 | |||||||||||||||
| Coupon rate | |||||||||||||||||
| Revenue participation payments | Maximum each year | Maximum each year | |||||||||||||||
| Discount rate | % | (1) | % | (1) | |||||||||||||
| Probability of mandatory prepayment after 2025 | (1) | (1) | |||||||||||||||
| Estimated timing of mandatory prepayment event after 2025 | March 31, 2026 | (1) | March 31, 2026 | (1) | |||||||||||||
| Probability of optional prepayment event | (1) | (1) | |||||||||||||||
| Estimated timing of optional prepayment event | January 31, 2026 | (1) | December 31, 2025 | (1) | |||||||||||||
Probability of note held-to-maturity (2) | % | (1) | % | (1) | |||||||||||||
__________
(1)Represents a significant unobservable input.
(2)See Maturity date in table.
The fair value of cash, restricted cash, accounts receivable, and accounts payable and accrued expenses approximates the carrying values because of the short-term nature of these instruments. The carrying value and fair value of the Credit Facility were $50,000 and $53,135 at December 31, 2025, respectively, and $47,496 and $51,307 at December 31, 2024, respectively. See Note 9 - Long-Term Debt, Net of Debt Discount and Financing Fees.
7.Accounts Payable and Accrued Expenses
Accounts payable and accrued expenses consist of the following as of the periods presented:
| (in thousands) | December 31, 2025 | December 31, 2024 | |||||||||
| Accounts payable | $ | $ | |||||||||
| Accrued expenses | |||||||||||
| Accrued compensation | |||||||||||
| Accounts payable and accrued expenses | $ | $ | |||||||||
8. Leases
The Company leases administrative, manufacturing, research, and distribution facilities through operating leases. Several leases include fixed payments including rent and non-lease components such as common-area or other maintenance costs.
The components of total operating lease expense and sublease income are as follows for the periods presented:
| Years Ended December 31, | |||||||||||||||||
| (in thousands) | 2025 | 2024 | 2023 | ||||||||||||||
| Operating lease costs | $ | $ | $ | ||||||||||||||
| Short term lease costs | |||||||||||||||||
| Variable lease costs | |||||||||||||||||
| Total operating lease expense | $ | $ | $ | ||||||||||||||
| Sublease income | $ | $ | $ | ||||||||||||||
86
Axogen, Inc.
Notes to Consolidated Financial Statements - Continued
Years ended December 31, 2025, 2024 and 2023
(in thousands, except share and per share amounts)
Supplemental balance sheet information related to the operating and financing leases is as follows as of the periods presented:
| (dollars in thousands) | December 31, 2025 | December 31, 2024 | |||||||||
| Operating Leases | |||||||||||
| Right-of-use operating lease assets | $ | $ | |||||||||
| $ | $ | ||||||||||
| $ | $ | ||||||||||
| Financing Leases | |||||||||||
Right-of-use finance lease assets, net of accumulated amortization (1) | $ | $ | |||||||||
| $ | $ | ||||||||||
| $ | $ | ||||||||||
| Weighted average remaining lease term - operating leases | |||||||||||
| Weighted average remaining lease term - finance leases | |||||||||||
| Weighted average discount rate - operating leases | % | % | |||||||||
| Weighted average discount rate - finance leases | % | % | |||||||||
__________
(1)Financing leases are included in Property and equipment, net on the Consolidated Balance Sheets.
Future minimum lease payments under operating and financing leases as of December 31, 2025 are as follows:
| Years ending December 31, | (in thousands) | ||||
| 2026 | $ | ||||
| 2027 | |||||
| 2028 | |||||
| 2029 | |||||
| 2030 | |||||
| Thereafter | |||||
| Total | |||||
| Less: Imputed interest | ( | ||||
| Total lease obligations | |||||
| Less: Current maturities of long-term obligations | ( | ||||
| Long-term lease obligations | $ | ||||
Lease modifications
The Company accounts for lease revisions as a lease modification when the modification effectively terminates the existing lease and creates a new lease. No lease modifications were recorded during the years ended December 31, 2025 and 2024.
Sublease Agreements
87
Axogen, Inc.
Notes to Consolidated Financial Statements - Continued
Years ended December 31, 2025, 2024 and 2023
(in thousands, except share and per share amounts)
9. Long-Term Debt, Net of Debt Discount and Financing Fees
Long-term debt, net of debt discount and financing fees consists of the following:
| (in thousands) | December 31, 2025 | December 31, 2024 | |||||||||
| Credit Facility - first tranche | $ | $ | |||||||||
| Credit Facility - second tranche | |||||||||||
| Less: Unamortized debt discount and deferred financing fees | ( | ( | |||||||||
| Long-term debt, net of debt discount and financing fees | $ | $ | |||||||||
Credit Facility
On June 29, 2023, the Company amended its Credit Facility with Oberland Capital and its affiliates, TPC Investments II LP and Argo LLC (collectively, the “Lender”), to transition the base interest rate from three-month LIBOR to the forward looking term rate based on the secured overnight financing rate as set by the Federal Reserve Bank of New York plus 0.10 % (“Adjusted SOFR”). The Company obtained the first tranche of $35,000 at closing on June 30, 2020. On June 30, 2021, the second tranche of $15,000 was drawn down by the Company.
Each tranche under the Credit Facility requires quarterly interest payments for seven years . Interest is calculated as 7.5 % plus the greater of Adjusted SOFR or 2.0 % (11.59 % as of December 31, 2025); provided that the interest rate shall never be less than 9.5 %. Each tranche of the Credit Facility has a term of seven years from the date of issuance (with the first tranche issued on June 30, 2020, maturing on June 30, 2027, and the second tranche issued on June 30, 2021, maturing on June 30, 2028). In connection with the Credit Facility, the Company entered into a revenue participation agreement (the “Revenue Participation Agreement”) with the Lender, which provided that, among other things, a quarterly royalty payment as a percentage of the Company’s net revenues up to $70,000 in any given year, after April 1, 2021, ending on the date upon which all amounts owed under the Credit Facility have been paid in full. This structure results in approximately 1.5 % per year of additional interest payments on the outstanding loan amount. The Company recorded interest expense for this Revenue Participation Agreement of $756 5,992 , $6,475 and $6,436 , respectively, to the Lender. The Company capitalized interest of $5,285 for the year ended December 31, 2023 towards the costs to construct and retrofit its Axogen Processing Center facility (the “APC Facility”) in Vandalia, Ohio, which was completed during 2023.
The amounts outstanding under the Credit Facility may be accelerated upon certain events, including: (a) required mandatory prepayments upon an asset sale; (b) in the event the Company is subject to (i) any litigation brought by a Governmental Authority (as defined in the Credit Facility) including intervention after litigation is commenced by a Person (as defined in the Credit Facility), or (ii) any final administrative action by a Governmental Authority, in each case arising out of or in connection with any of the Company’s registry studies, payments made to doctors or training activities with respect to healthcare professionals (excluding certain final administrative actions that have been fully and finally resolved by the parties pursuant to a settlement agreement) or (c) upon the occurrence of an event of default (either automatically or at the option of the Lender depending on the nature of the event). In addition, the Company has the right to prepay any amounts outstanding under the Credit Facility. Upon maturity or upon such earlier repayment of the Credit Facility, the Company will repay the principal balance and provide a make-whole payment calculated to generate an internal rate of return to the Lender equal to 11.5 %, less the total of all quarterly interest and royalty payments previously paid to the Lender (the “Make-Whole Payment”). See Note 15 - Commitments and Contingencies for further information related to the Make-Whole Payment calculation.
Upon the occurrence of an event of default, the interest rate incurred on amounts outstanding under the Credit Facility will be increased by 4 %. The Credit Facility includes a financial covenant requiring the Company to achieve certain revenue targets each quarter. As of December 31, 2025, the Company was in compliance with all the covenants. In the event of a failure to meet such covenants, the Company may avoid a default by electing to be subject to a liquidity covenant and meeting all of the obligations required by such covenant. The borrowings under the Credit Facility are secured by substantially all of the assets of the Company.
88
Axogen, Inc.
Notes to Consolidated Financial Statements - Continued
Years ended December 31, 2025, 2024 and 2023
(in thousands, except share and per share amounts)
Unamortized Debt Discount and Financing Fees
The unamortized debt discount consists of the remaining initial fair values of the embedded derivatives related to the Credit Facility. The unamortized debt discount and deferred financing fees for the Credit Facility were recorded as a contra-liability to Long-term debt on the Consolidated Balance Sheets.
Amortization of debt discount and deferred financing fees for the years ended December 31, 2025, 2024 and 2023 was $891 , $893 and $891 , respectively, and recorded in Interest expense on the Consolidated Statements of Operations, using the effective interest rate method.
Termination of the Credit Facility
On January 20, 2026, the Company entered into a payoff letter with the Lender (the “Payoff Letter”). Pursuant to the Payoff Letter, the final payoff amount was $69,707 (the “Payoff Amount”), so long as the payoff date was on or before February 15, 2026. Upon payment of the Payoff Amount on January 28, 2026 and satisfaction of the other conditions specified in the Payoff Letter, all obligations under the Credit Facility, including the Make-Whole Payment, were paid in full, all liens and security interests securing such obligations were released, and the Credit Facility and related loan documents were terminated, subject to certain customary surviving provisions. Payment of the Payoff Amount was funded by proceeds from an underwritten public offering of an aggregate 4,600,000 shares of the Company’s common stock, $0.01 par value per share, at a public offering price of $31.00 per share (the “Offering”) completed by the Company on January 23, 2026. See Note 16 - Subsequent Events for additional details regarding the Offering.
Other Credit Facilities
10.Basic and Diluted Loss Per Common Share
The following reflects the net loss attributable to common shareholders and share data used in the basic and diluted earnings per share computations using the two-class method for the periods presented:
| Years Ended December 31, | |||||||||||||||||
| (in thousands, except share and per share amounts) | 2025 | 2024 | 2023 | ||||||||||||||
| Numerator: | |||||||||||||||||
| Net loss | $ | ( | $ | ( | $ | ( | |||||||||||
| Denominator: | |||||||||||||||||
| Weighted average common shares outstanding — basic and diluted | |||||||||||||||||
| Net loss per common share — basic and diluted | $ | ( | $ | ( | $ | ( | |||||||||||
Anti-dilutive shares excluded from the calculation of diluted earnings per share (1) | |||||||||||||||||
| Stock options | |||||||||||||||||
| Restricted and performance stock units | |||||||||||||||||
__________
(1)These common equivalent shares are not included in the diluted per share calculations as they would be dilutive if the Company was in a net income position.
89
Axogen, Inc.
Notes to Consolidated Financial Statements - Continued
Years ended December 31, 2025, 2024 and 2023
(in thousands, except share and per share amounts)
11. Stock-Based Compensation
The Company maintains two stock-based incentive plans: (i) The Axogen, Inc. Fourth Amended and Restated 2019 Long-Term Incentive Plan (“2019 Plan”) which provides incentives through the grants of stock options, non-qualified stock options, PSUs and RSUs to employees, directors and consultants which replaced the Company’s 2010 Stock Incentive Plan and (ii) The Axogen 2017 Employee Stock Purchase Plan (“2017 ESPP”).
At the June 19, 2025 Annual Shareholder Meeting, approval was received to increase the number of shares available under the 2019 Plan from 10,500,000 to 13,400,000 .
During the year ended December 31, 2025, the Company issued 148,000 RSUs and 58,000 PSUs as inducement grants to certain employees in accordance with Nasdaq Listing Rule 5635(c)(4). As of December 31, 2025, there were 4,833,771 shares of common stock available for future grant under the 2019 Plan.
Stock-based compensation expense is included in the following line items on the accompanying Consolidated Statements of Operations for the periods presented:
| Years Ended December 31, | |||||||||||||||||
| (in thousands) | 2025 | 2024 | 2023 | ||||||||||||||
| Costs of goods sold | $ | $ | $ | ||||||||||||||
| Sales and marketing | |||||||||||||||||
| Research and development | |||||||||||||||||
| General and administrative | |||||||||||||||||
| Total non-cash stock-based compensation | $ | $ | $ | ||||||||||||||
Stock Options
Stock options granted to employees typically vest 50 % two years after the grant date and 12.5 % every six months thereafter for the remaining two-year period until fully vested after four years . Stock options granted to directors and certain options granted from time to time to certain executive officers vest ratably over three years . Options typically have terms of ten years . The Company estimates the fair value of each option award on the date of grant using a multiple-point Black-Scholes option-pricing model. In addition, the Company estimates the grant date fair value of stock options granted to employees at a premium price based on market conditions, such as the trading price of the Company’s common stock, using a Monte Carlo simulation option-pricing model. The value of the portion of the award that is ultimately expected to vest is recognized as expense over the requisite service periods in the Company’s Consolidated Statements of Operations. The expense is reduced for forfeitures as they occur.
The following weighted-average assumptions were used in the calculation of fair value for stock options granted for the periods presented:
| Years Ended December 31, | |||||||||||
| 2024 | 2023 | ||||||||||
| Expected term (in years) | |||||||||||
| Expected volatility | % | % | |||||||||
| Risk free interest rate | % | % | |||||||||
| Expected dividends | % | % | |||||||||
90
Axogen, Inc.
Notes to Consolidated Financial Statements - Continued
Years ended December 31, 2025, 2024 and 2023
(in thousands, except share and per share amounts)
The following table summarizes the Company’s stock option activity for the year ended December 31, 2025:
| Options | Weighted Average Exercise Price | Weighted Average Remaining Contractual Life (Years) | Aggregate Intrinsic Value (in thousands) | ||||||||||||||||||||
| Outstanding at December 31, 2024 | $ | $ | |||||||||||||||||||||
| Forfeited | ( | $ | |||||||||||||||||||||
| Exercised | ( | $ | |||||||||||||||||||||
Outstanding at December 31, 2025 (1) | $ | $ | |||||||||||||||||||||
| Exercisable at December 31, 2025 | $ | $ | |||||||||||||||||||||
__________
The weighted-average grant-date fair value of stock options granted during the years ended December 31, 2024 and 2023 was $4.33 and $4.72 , respectively.
The total intrinsic value of options exercised for the years ended December 31, 2025, 2024 and 2023 was $21,204 , $2,031 and $1,710 , respectively.
As of December 31, 2025, there was approximately $697 of total unrecognized compensation costs related to unvested stock options. These costs are expected to be recognized over a weighted-average period of 1.2 years.
Restricted Stock Units
RSUs granted to employees have a requisite service period of four years . The RSUs granted to directors and certain RSUs granted from time to time to certain executive officers and vice presidents have a requisite service period of three years , while certain of these RSUs have a requisite service period of one year . The Company estimates the fair value of RSU grants based upon the grant date closing market price of the Company’s common stock. The Company expenses the fair value of RSUs on a straight-line basis over the requisite service period.
The following table summarizes the activity for RSUs for the year ended December 31, 2025:
| Outstanding Restricted Stock Units | |||||||||||||||||||||||
Restricted Stock Units | Weighted Average Fair Value at Date of Grant per Share | Weighted Average Remaining Vesting Life (Years) | Aggregate Intrinsic Value (in thousands) | ||||||||||||||||||||
| Unvested at December 31, 2024 | $ | $ | |||||||||||||||||||||
Granted (1) | $ | ||||||||||||||||||||||
| Released | ( | $ | |||||||||||||||||||||
Forfeited (2) | ( | $ | |||||||||||||||||||||
| Unvested at December 31, 2025 | $ | $ | |||||||||||||||||||||
__________
(1)RSUs granted include 148,000 inducement shares in accordance with Nasdaq Listing Rule 5635(c)(4).
(2)RSUs forfeited include 150,000 inducement shares in accordance with Nasdaq Listing Rule 5635(c)(4).
The weighted-average grant-date fair value of RSUs granted during the years ended December 31, 2025, 2024 and 2023 was $16.58 , $8.79 and $8.10 , respectively.
As of December 31, 2025, there was approximately $23,032 of total unrecognized compensation costs related to unvested restricted stock. These costs are expected to be recognized over a weighted-average period of 2.5 years.
91
Axogen, Inc.
Notes to Consolidated Financial Statements - Continued
Years ended December 31, 2025, 2024 and 2023
(in thousands, except share and per share amounts)
Performance Stock Units
The Company estimates the fair value of PSUs based on its closing stock price at the time of grant and its estimate of achieving the applicable performance target goals and records compensation expense as the milestones are achieved. The number of shares delivered to recipients and the related compensation cost recognized as an expense will be based on the actual performance metrics as set forth in the applicable PSU award agreement. The amount actually awarded will be based upon achievement of the performance measures.
The Company’s CAGR TSR PSUs generally have a requisite service period of three years and are subject to vesting conditions based on goals defined within the award. The grant date fair value of the CAGR TSR PSUs is calculated using a Monte Carlo simulation. The Company expenses their fair value over the requisite service period. Over the performance period, the number of shares of common stock that will ultimately vest and be issued and the related compensation expense will be adjusted based upon the Company’s estimate of achieving such performance target.
The Company’s TSR PSUs generally have a requisite service period of three years and are subject to vesting conditions based on goals defined within the award. The grant date fair value of the TSR PSUs is calculated using a Monte Carlo simulation. The Company expenses their fair value over the requisite service period. Over the performance period, the number of shares of common stock that will ultimately vest and be issued will be adjusted based upon the Company’s estimate of achieving such performance target.
PSUs issued in 2017 and 2019 tied to the achievement of certain milestones have performance periods through December 31, 2025 and a requisite service period of one year after the milestone achievement date but not sooner than one year after the grant date. PSUs issued in 2018 tied to the achievement of certain milestones have performance periods through January 1, 2025 and a requisite service period of one year after the milestone achievement date but not sooner than one year after the grant date.
PSUs issued in 2024 tied to the achievement of certain milestones have performance periods through December 31, 2025 and requisite service periods through the date of the milestone achievement but not sooner than one year after the grant date. The Company expenses the fair value upon the achievement of such milestone and subsequent requisite period.
The following table summarizes the activity for PSUs for the year ended December 31, 2025:
| Outstanding Performance Stock Units | |||||||||||||||||||||||
Performance Stock Units | Weighted Average Fair Value at Date of Grant per Share | Weighted Average Remaining Vesting Life (Years) | Aggregate Intrinsic Value (in thousands) | ||||||||||||||||||||
| Unvested at December 31, 2024 | $ | $ | |||||||||||||||||||||
Granted (1) | $ | ||||||||||||||||||||||
| Released | ( | $ | |||||||||||||||||||||
| Forfeited | ( | $ | |||||||||||||||||||||
| Unvested at December 31, 2025 | $ | $ | |||||||||||||||||||||
Vested and deferred (2) | $ | $ | |||||||||||||||||||||
__________
(1)PSUs granted include 58,000 inducement shares in accordance with Nasdaq Listing Rule 5635(c)(4).
(2)Represents PSUs tranches that vested based on achievement of performance metrics for the 2025, 2024 and 2023 performance periods. The related shares were released but deferred in accordance with the terms of the award agreements, with the release of shares occurring upon the earlier of termination of employment or the final vesting date of the full PSU award in February 2026.
The weighted-average grant-date fair value of PSUs granted during the years ended December 31, 2025, 2024 and 2023 was $21.82 , $9.11 and $8.29 , respectively.
As of December 31, 2025, there was approximately $22,595 of total unrecognized compensation costs related to unvested PSU awards. These costs are expected to be recognized over a weighted-average period of 1.5 years.
92
Axogen, Inc.
Notes to Consolidated Financial Statements - Continued
Years ended December 31, 2025, 2024 and 2023
(in thousands, except share and per share amounts)
During 2017, 2018 and 2019, the Company issued PSU awards to certain employees related to their work on the Company’s Biologics License Application (“BLA”). The number of shares was allocated to certain milestones related to the BLA submission to and approval by the FDA. The performance measure is based upon achieving each of the specific milestones and will vest 50 50 one year one year after the grant date. As of December 31, 2025, substantially all of the awards issued related to achieving BLA milestones had vested. The Company generally recognizes expense on these milestones upon the achievement. During the three months ended December 31, 2025, the Company recognized $8,485 of PSU expense related to the approval of the BLA.
During 2023, the Company issued PSU awards to certain officers and employees tied to revenue from 2023 to 2025 with a pay-out range from 0 % to 150 % upon achievement of specific revenue targets. These awards were achieved at 112.6 %, vested in the fourth quarter of 2025 and will be released in the first quarter of 2026.
During 2024, the Company issued TSR PSU awards to certain officers and employees tied to the Company’s share price targets with a pay-out range from 0 % to 200 % upon achievement of specific average share prices over a 30 day trading period immediately preceding the end of a performance period of February 2, 2024 through February 22, 2027.
During 2025, the Company issued CAGR TSR PSU awards to certain officers and employees tied to CAGR of revenue from 2025 to 2027, subject to modification based on the Company’s TSR relative to the TSR of certain peer companies.
The following weighted-average assumptions were used in calculation of fair value for PSUs granted using the Monte Carlo simulation pricing model for the periods presented:
| Years Ended December 31, | |||||||||||
| 2025 | 2024 | ||||||||||
| Expected term (in years) | |||||||||||
| Expected volatility | % | % | |||||||||
| Risk free interest rate | % | % | |||||||||
| Expected dividends | % | % | |||||||||
Employee Stock Purchase Plan
The 2017 ESPP allows eligible employees to acquire shares of the Company’s common stock through payroll deductions at a discount to market price (currently 15.0 %) of the lesser of the closing price of the Company’s common stock on the first day or last day of the offering period. The offering period is currently six months . Participants may not purchase more than $25 or 3,000 shares of the Company’s common stock in a calendar year through the ESPP. The Company estimates the grant date fair value, and the resulting stock-based compensation expense, using a Black-Scholes option pricing model for each purchase period. Stock-based compensation expense related to the 2017 ESPP, included in total stock-based compensation expense, was $442 , $371 and $333 for the years ended December 31, 2025, 2024 and 2023, respectively.
The following are the weighted average assumptions used in the valuation of ESPP options for the periods presented:
| Years Ended December 31, | |||||||||||||||||
| 2025 | 2024 | 2023 | |||||||||||||||
| Expected term | |||||||||||||||||
| Expected volatility | |||||||||||||||||
| Risk-free interest rate | |||||||||||||||||
| Expected dividends | |||||||||||||||||
93
Axogen, Inc.
Notes to Consolidated Financial Statements - Continued
Years ended December 31, 2025, 2024 and 2023
(in thousands, except share and per share amounts)
12. Income Taxes
Deferred income taxes are accounted for using the balance sheet approach, which requires recognition of deferred tax assets and liabilities for the expected future consequences of temporary differences between the financial reporting basis and the tax basis of assets and liabilities, as measured by enacted state and federal tax rates. Deferred tax assets and deferred tax liabilities are as follows:
| (in thousands) | December 31, 2025 | December 31, 2024 | |||||||||
| Deferred tax assets: | |||||||||||
| Net operating loss carryforwards | $ | $ | |||||||||
| Inventory write down | |||||||||||
| Interest limitation | |||||||||||
| Allowance for doubtful accounts | |||||||||||
| Lease obligations | |||||||||||
| Stock-based compensation | |||||||||||
| Capitalized research and development costs | |||||||||||
| Debt derivative liabilities | |||||||||||
| Charitable contributions | |||||||||||
| Accrued compensation | |||||||||||
| Total deferred tax assets | |||||||||||
| Deferred tax liabilities: | |||||||||||
| Depreciation | ( | ( | |||||||||
| Amortization | ( | ( | |||||||||
| Right-of-use asset | ( | ( | |||||||||
| Contract liabilities | ( | ||||||||||
| Total deferred tax liabilities | ( | ( | |||||||||
| Net deferred tax assets | |||||||||||
| Valuation allowance | $ | ( | $ | ( | |||||||
A valuation allowance is provided to reduce the deferred tax assets reported if, based on the weight of the evidence, it is more-likely-than-not that a portion or none of the deferred tax assets will be realized. As of December 31, 2025 and 2024, management assessed the realizability of deferred tax assets. After consideration of all the evidence, including reversal of deferred tax liabilities, future taxable income and other factors, management determined that a full valuation allowance was necessary as of December 31, 2025 and 2024. The valuation allowance decreased by $557 and increased by $315 during 2025 and 2024, respectively. During 2025, the decrease was primarily driven by changes in depreciation expense and research and development costs resulting from the introduction of the One Big Beautiful Bill Act. During 2024, the increase was primarily due to higher capitalized research and development costs for tax purposes.
94
Axogen, Inc.
Notes to Consolidated Financial Statements - Continued
Years ended December 31, 2025, 2024 and 2023
(in thousands, except share and per share amounts)
The difference between the financial statement income tax benefit and the income tax benefit using statutory rates is primarily due to the valuation allowance and non-deductible permanent items such as meals, entertainment and equity compensation. The Company’s effective income tax rate differs from the statutory federal income tax rate as follows:
Year Ended December 31, 2025 | |||||||||||
(dollars in thousands) | % Impact | $ Impact | |||||||||
| % | $ | ( | |||||||||
| State taxes, net of federal benefit | ( | ||||||||||
| Nontaxable and nondeductible items: | |||||||||||
| Nondeductible executive compensation under IRC 162(m) | ( | ||||||||||
| Nondeductible equity-based compensation permanent items | ( | ||||||||||
| Other | ( | ||||||||||
| Other | ( | ||||||||||
| Change in valuation allowance | ( | ||||||||||
| Effective income tax rate | % | $ | |||||||||
| Years Ended December 31, | |||||||||||
| 2024 | 2023 | ||||||||||
| % | % | ||||||||||
| State taxes, net of federal benefit | |||||||||||
Nontaxable and nondeductible items | ( | ( | |||||||||
| Other | ( | ( | |||||||||
| Change in valuation allowance | ( | ( | |||||||||
| Effective income tax rate | % | % | |||||||||
As of December 31, 2025, the Company had tax-effected net operating loss (“NOL”) carryforwards of $46,519 to offset future taxable income. The TCJA enacted significant changes to NOL utilization. NOLs generated after January 1, 2018 limit the NOL utilization to 80% of taxable income. The remaining 20% is carried forward to subsequent years. NOLs incurred in tax years beginning on or after January 1, 2018 are carried forward indefinitely. NOLs incurred in tax years prior to January 1, 2018 are subject to a twenty-year carryforward before expiring. A portion of the NOL carryforwards may expire due to limitations imposed by Section 382 of the Internal Revenue Code. Future utilization of the available NOL carryforwards may be limited under Internal Revenue Code Section 382 as a result of changes in ownership.
The Company files U.S. federal and state income tax returns in jurisdictions with varying statutes of limitations. In the normal course of business, the Company is subject to examination by taxing authorities throughout the U.S. These examinations could include examining the timing and amount of deductions, the allocation of income among various tax jurisdictions and compliance with federal, state, and local laws. The Company’s remaining open tax years subject to examination by federal tax authorities include the years ended December 31, 2021 through 2024. The Internal Revenue Service is currently examining the Company’s 2021 federal income tax return. The Company’s remaining open tax years subject to examination by state and foreign tax authorities include the years ended December 31, 2020 through 2024. However, for tax years 2004 through 2017, federal and state taxing authorities may examine and adjust loss carryforwards in the years in which those loss carryforwards are ultimately utilized.
The TCJA, enacted in 2018, subjects a U.S. shareholder to tax on global intangible low-taxed income (“GILTI”) earned by certain foreign subsidiaries. The Company has elected to account for GILTI in the year the tax is incurred.
The Company has no
95
Axogen, Inc.
Notes to Consolidated Financial Statements - Continued
Years ended December 31, 2025, 2024 and 2023
(in thousands, except share and per share amounts)
13. Segments
The Company determines its operating segments in accordance with FASB ASC 280, Segment Reporting (“ASC 280”). ASC 280 defines operating segments as components where discrete financial information is regularly reviewed by the chief operating decision maker (“CODM”), which for the Company is the Chief Executive Officer (“CEO”), to determine resource allocation and assess performance. As such, based on the way the CODM monitors and makes decisions affecting operations, the Company has concluded that it has one one operating segment and is managed on a consolidated basis, the measure of profit or loss is consolidated net income or loss. The metrics are used to review operating trends, to perform analytical comparisons between periods and to monitor budget to actual variances. See the Consolidated Statements of Operations.
Geographic Areas
International revenues are defined as revenues generated from sales to customers outside of the U.S. The following table details total revenues by major geographic area for the periods indicated:
| Years Ended December 31, | |||||||||||||||||
| (in thousands) | 2025 | 2024 | 2023 | ||||||||||||||
| U.S. | $ | $ | $ | ||||||||||||||
| International | |||||||||||||||||
| Total revenues | $ | $ | $ | ||||||||||||||
As of December 31, 2025 and 2024, all of the Company’s long-lived assets were held within the U.S.
14. Retirement Plan
The Company sponsors the Axogen 401(k) plan (the “401(k) Plan”), a defined contribution plan covering substantially all employees of the Company. All full-time employees who have attained the age of 18 are eligible to participate in the 401(k) Plan. Eligibility is immediate upon employment and enrollment is available any time during employment. Participating employees may make annual pretax contributions to their accounts up to a maximum amount as limited by law. The 401(k) Plan requires the Company to make 100 % matching contributions on up to 3 % of the employee’s annual salary and 50 % matching contributions on up to the next 2 % of the employee’s annual salary as long as the employee participates in the 401(k) Plan. Employee contributions and Company contributions made prior to January 1, 2025 vest immediately. Company contributions made subsequent to January 1, 2025 vest after two years of service, as defined by the 401(k) Plan. Employer contributions to the 401(k) Plan were $1,967 , $1,669 and $1,612 for the years ended December 31, 2025, 2024 and 2023, respectively.
15. Commitments and Contingencies
Service Agreements
The Company pays a third party a facility fee for the use of clean-rooms, manufacturing, storage, and office space and for services in support of its tissue processing including for routine sterilization of daily supplies, providing disposable supplies and microbial services, and office support pursuant to a License and Services Agreement, as amended (the “License and Services Agreement”). Pursuant to the License and Services Agreement, the Company recorded expenses of $915 , $910 and $2,327 for the years ended December 31, 2025, 2024 and 2023, respectively, in Cost of goods sold. The License and Services Agreement was amended on December 21, 2023, extending the term through December 31, 2026. The License and Services Agreement may be terminated by either party by providing an 18 month written notice. While the Company ended its utilization of the same third party for Avance Nerve Graft in the fourth quarter of 2023, the Company continues to utilize the same third party for processing and packaging of Avive+ Soft Tissue Matrix.
In December 2011, the Company entered into a Master Services Agreement for Clinical Research and Related Services. The Company was required to pay $151 upon execution of this agreement and the remainder monthly based on activities associated with the execution of the Company’s Phase III pivotal clinical trial to support the BLA for Avance. The Master Services Agreement for Clinical Research and Related Services was completed in 2023. Payments made under this agreement were $191 for the year ended December 31, 2023.
96
Axogen, Inc.
Notes to Consolidated Financial Statements - Continued
Years ended December 31, 2025, 2024 and 2023
(in thousands, except share and per share amounts)
Distribution and Supply Agreements
On August 27, 2008, the Company entered into an exclusive distribution agreement with Cook Biotech Incorporated (acquired on January 31, 2024 by RTI Surgical, Inc. and rebranded on December 17, 2024 as Evergen) (“Evergen”), to distribute the Axoguard Nerve Connector and Axoguard Nerve Protector products worldwide and the parties subsequently amended the agreement on August 4, 2023 (the “Distribution Agreement”). The Distribution Agreement expires on December 31, 2030. The Distribution Agreement establishes a formula for the transfer cost of the Axoguard Nerve Connector and Axoguard Nerve Protector products and requires certain minimum purchases by the Company, although, through mutual agreement, the parties have not established such minimums; and, to date, have not enforced such provision. Under the Distribution Agreement, the Company provides purchase orders to Evergen, and Evergen fulfills the purchase orders. The Distribution Agreement allows for termination provisions for both parties. The loss of the ability to sell the Axoguard Nerve Connector and Axoguard Nerve Protector products could have a material adverse effect on the Company’s business until other replacement products would be available.
The Company entered into the Nerve End Cap Supply Agreement dated June 27, 2017, as amended on April 6, 2020 and August 4, 2023 (the “Amended Supply Agreement”) with Evergen whereby Evergen is the exclusive contract manufacturer of the Axoguard Nerve Cap. The Amended Supply Agreement expires on December 31, 2030. The Amended Supply Agreement establishes the terms and conditions in which Evergen will manufacture the product for the Company. Under the Amended Supply Agreement, the Company provides purchase orders to Evergen and Evergen fulfills the purchase orders. The Amended Supply Agreement allows for termination provisions for both parties. The loss of the ability to sell the Axoguard Nerve Cap product could have a material adverse effect on the Company’s business until other replacement products would be available.
In May 2023, the Company entered into a Supply and Manufacturing Agreement (“Axoguard HA+ Nerve Protector Supply Agreement”) with Evergen whereby Evergen is the exclusive contract manufacturer of the Axoguard HA + Nerve Protector. The Axoguard HA+ Nerve Protector Supply Agreement expires on July 1, 2030. The Axoguard HA+ Nerve Protector Supply Agreement establishes the terms and condition in which Evergen will manufacture, package, label and deliver the product to the Company. Under the Axoguard HA+ Nerve Protector Supply Agreement, the Company provides purchase orders to Evergen, and Evergen fulfills the purchase orders. The Axoguard HA+ Nerve Protector Supply Agreement allows for termination provisions for both parties. The loss of the ability to sell the Axoguard HA + Nerve Protector product could have a material adverse effect on the Company’s business until other replacement products would be available.
Insurance Financing Agreement
The Company entered into an Insurance Financing Agreement on December 31, 2024. Outstanding payments owed of $1,255 under the Insurance Financing Agreement were included in Prepaid expenses and other on the Consolidated Balance Sheet as of December 31, 2024.
Processing Facilities
The Company is highly dependent on the continued availability of its processing facilities at its APC Facility in Vandalia, Ohio and the facility it leases Dayton, Ohio and could be harmed if the physical infrastructure of these facilities is unavailable for any prolonged period of time.
Certain Economic Development Grants
The Company obtained certain economic development grants from state and local authorities totaling up to $2,685 including $1,250 of cash grants to offset costs to acquire and develop the APC Facility. Certain of these economic development grants were subject to fixed asset investments and job creation milestones by December 31, 2024, and have clawback clauses if the Company does not meet the job creation milestones. The Company has not met certain job creation milestones and has requested a reduction or waiver of clawbacks or extensions from the grant authorities to extend the job creation milestones. In October 2025, the Company received notification from certain grant authorities that the Company is expected to satisfy our job creation milestone, assuming we continue to maintain certain headcount and payroll amount thresholds through December 31, 2026. If the Company is unable to maintain minimum requirements under our grant agreements the Company could be obligated to pay back up to approximately $950 as of December 31, 2025 related to these grants. As of December 31, 2025, the Company had received $1,250 in cash grants related to these economic development grants during the years ended 2021 and 2020.
97
Axogen, Inc.
Notes to Consolidated Financial Statements - Continued
Years ended December 31, 2025, 2024 and 2023
(in thousands, except share and per share amounts)
Fair Value of the Debt Derivative Liabilities
The fair value of the debt derivative liabilities is $3,886 as of December 31, 2025. The fair value of the debt derivative liabilities was determined using a probability-weighted expected return model based upon three potential settlement scenarios for the Credit Facility which are described in Note 2 - Summary of Significant Accounting Policies – Derivative Instruments. The estimated settlement value of each scenario includes any required Make-Whole Payment, see Note 9 - Long-Term Debt, Net of Debt Discount and Financing Fees, and then discounted to present value using a discount rate that is derived based upon the initial terms of the Credit Facility at issuance and corroborated utilizing a synthetic rating analysis. The calculated fair values under the three scenarios are then compared to the fair value of a plain vanilla note, with the difference reflecting the fair value of the debt derivative liabilities. The Company estimated the Make-Whole Payment required under each scenario according to the terms of the Credit Facility to generate an internal rate of return equal to 11.5 % through the scheduled maturity dates, less the total of all quarterly interest and royalty payments previously paid to the Lender. The calculation utilized the XIRR function in Microsoft Excel as required by the Credit Facility.
The Company is aware that the Lender has an alternative interpretation of the calculation of the Make-Whole Payment. The Company estimates the top end of the range of the Make-Whole Payment under an alternative interpretation to be approximately $15,500 for the first tranche of the Credit Facility due on June 30, 2027, and approximately $8,200 for the second tranche of the Credit Facility due on June 30, 2028. Under the alternative interpretation of the calculation, if the Credit Facility was to be prepaid in whole as of December 31, 2025, the Make-Whole Payment, in excess of the outstanding principal of the Credit Facility, is estimated to be approximately $23,700 .
In January 2026, the Company entered into a Payoff Letter with the Lender pursuant to which the final Payoff Amount for the Credit Facility, including the Make-Whole Payment, was $69,707 . On January 28, 2026, the Company remitted the Payoff Amount to the Lender and after satisfaction of all other conditions specified in the Payoff Letter, all obligations under the Credit Facility were paid in full. See Note 9 - Long-Term Debt, Net of Debt Discount and Financing Fees for additional details regarding the Payoff Letter and termination of the Credit Facility.
Other Commitments
Certain executive officers of the Company are parties to employment contracts. Such contracts have severance payments for certain conditions including change of control.
Legal Proceedings
The Company is and may be subject to various claims, lawsuits, and proceedings in the ordinary course of the Company’s business. Such matters are subject to many uncertainties and outcomes are not predictable with assurance. While there can be no assurances as to the ultimate outcome of any legal proceeding or other loss contingency involving the Company. In the opinion of management, such claims are either adequately covered by insurance or otherwise indemnified, or are not expected, individually or in the aggregate, to result in a material, adverse effect on the Company's financial condition, results of operations or cash flows. However, it is possible that the Company’s results of operations, financial position and cash flows in a particular period could be materially affected by these contingencies.
16. Subsequent Events
On January 21, 2026, the Company entered into an underwriting agreement (the “Underwriting Agreement”) with Wells Fargo Securities, LLC and Mizuho Securities USA LLC, as representatives of the underwriters (the “Underwriters”). Pursuant to the terms and conditions of the Underwriting Agreement, the Company agreed to sell 4,000,000 shares of its common stock, $0.01 31.00 per share, plus an additional 600,000 shares sold pursuant to the Underwriters’ option to purchase additional shares. The Offering closed on January 23, 2026 with a sale of 4,600,000 shares.
The Company received net proceeds from the Offering of $133,338 , after deducting the underwriting discounts and commissions, and expenses in connection with the Offering. Net proceeds from the Offering were used for the early payoff and termination of the Credit Facility for a final repayment amount of $69,707 . The remaining net proceeds will be used for working capital, capital expenditures and other general corporate purposes. See Note 9 - Long-Term Debt, Net of Debt Discount and Financing Fees for details regarding the termination of the Credit Facility.
98
ITEM 9. CHANGES IN AND DISAGREEMENTS WITH ACCOUNTANTS ON ACCOUNTING AND FINANCIAL DISCLOSURE
None.
ITEM 9A. CONTROLS AND PROCEDURES
Evaluation of Disclosure Controls and Procedures
We maintain “disclosure controls and procedures” as defined in Rules 13a-15(e) and 15d-15(e) under the Securities Exchange Act of 1934, as amended (the “Exchange Act”), that are designed to ensure that information required to be disclosed by us in reports we file or submit under the Exchange Act is recorded, processed, summarized and reported within the time periods specified in the SEC’s rules and forms, and that such information is accumulated and communicated to our management, including our principal executive officer and principal financial officer, and Board of Directors, as appropriate, to allow timely decisions regarding required disclosure. In designing and evaluating our disclosure controls and procedures, management recognizes that disclosure controls and procedures, no matter how well conceived and operated, can provide only reasonable assurance of achieving the desired objectives, and we necessarily are required to apply our judgment in evaluating the cost-benefit relationship of possible disclosure controls and procedures.
Our management, including our principal executive officer and principal financial officer, evaluated the effectiveness of our disclosure controls and procedures as of December 31, 2025, and concluded that our disclosure controls and procedures were effective.
Changes in Internal Control Over Financial Reporting
There were no changes in our internal control over financial reporting during the three months ended December 31, 2025, that materially affected, or are reasonably likely to materially affect, our internal control over financial reporting (as defined in Rules 13a-15(d) or 15d-15(f) of the Exchange Act).
Management’s Annual Report on Internal Control Over Financial Reporting
Our management is responsible for establishing and maintaining internal control over financial reporting, as such term is defined in Rules 13a-15(f) and 15d-15(f) of the Exchange Act. Our internal control system is designed to provide reasonable assurance regarding the reliability of financial reporting and the preparation of financial statements for external purposes in accordance with U.S. GAAP and includes those policies and procedures that:
•Pertain to the maintenance of records that in reasonable detail accurately and fairly reflect the transactions and dispositions of our assets;
•Provide reasonable assurance that transactions are recorded as necessary to permit preparation of financial statements in accordance with generally accepted accounting principles, and that our receipts and expenditures are being made only in accordance with authorizations of our management and directors; and
•Provide reasonable assurance regarding prevention or timely detection of unauthorized acquisition, use or disposition of our assets that could have a material effect on the consolidated financial statements.
Because of inherent limitations, a system of internal control over financial reporting may not prevent or detect misstatements. Also, projections of any evaluation of effectiveness to future periods are subject to the risk that controls may become inadequate due to a change in conditions, or that the degree of compliance with the policies or procedures may deteriorate.
Our management, including our principal executive officer and principal financial officer, evaluated the effectiveness of the design and operation of our internal control over financial reporting as of December 31, 2025. In making this assessment, our management used the criteria set forth by the Committee of Sponsoring Organizations of the Treadway Commission in Internal Control-Integrated Framework (2013). Based on their evaluation, the principal executive officer and principal financial officer concluded that our internal controls over financial reporting were effective.
Our independent registered public accounting firm, Deloitte & Touche LLP, who audited the consolidated financial statements included in this Annual Report on Form 10-K, has issued an attestation report on the effectiveness of management’s internal control over financial reporting as of December 31, 2025.
99
ITEM 9B. OTHER INFORMATION
During the three months ended December 31, 2025, none of our directors or officers adopted terminated
During the three months ended December 31, 2025, the Company did not adopt, modify or terminate a “Rule 10b5-1 trading arrangement” or a “non-Rule 10b5-1 trading arrangement” as such terms are defined under Item 408 of Regulation S-K.
ITEM 9C. DISCLOSURE REGARDING FOREIGN JURISDICTIONS THAT PREVENT INSPECTIONS
Not applicable.
100
PART III
ITEM 10. DIRECTORS, EXECUTIVE OFFICERS AND CORPORATE GOVERNANCE.
Information required by this item concerning our directors will be set forth under the caption “Election of Directors” in our definitive proxy statement for our 2026 annual meeting, which will be filed no later than 120 days after December 31, 2025, and is incorporated herein by reference.
If applicable, information required by this item concerning compliance with Section 16(a) of the Exchange Act, as amended, will be set forth under the caption “Security Ownership of Certain Beneficial Owners and Management — Delinquent Section 16(a) Reports” in our definitive proxy statement for our 2026 annual meeting, and is incorporated herein by reference.
Information required by this item concerning our audit committee, our audit committee financial expert and any material changes to the way in which security holders may recommend nominees to our Board of Directors will be set forth under the caption “Corporate Governance” in our definitive proxy statement for our 2026 annual meeting and is incorporated herein by reference.
The Board of Directors adopted a Code of Business Conduct and Ethics, which is posted on our website https://ir.axogeninc.com/governance-docs, that is applicable to all employees and directors. We will provide copies of our Code of Business Conduct and Ethics without charge upon request. To obtain a copy, please visit our website or send your written request to Investors Relations, 13631 Progress Blvd., Suite 400, Alachua, FL 32615. With respect to any amendments or waivers of this Code of Business Conduct and Ethics (to the extent applicable to our chief executive officer, principal accounting officer or controller, or persons performing similar functions) we intend to either post such amendments or waivers on our website or disclose such amendments or waivers pursuant to a Current Report on Form 8-K.
ITEM 11. EXECUTIVE COMPENSATION.
Information required by this item will be set forth under the caption “Executive Compensation” in our definitive proxy statement for our 2026 annual meeting, which will be filed no later than 120 days after December 31, 2025, and is incorporated herein by reference.
ITEM 12. SECURITY OWNERSHIP OF CERTAIN BENEFICIAL OWNERS AND MANAGEMENT AND RELATED STOCKHOLDER MATTERS.
Information required by this item concerning ownership will be set forth under the caption “Security Ownership of Certain Beneficial Owners and Management” and “Equity Compensation Plan Information” in our definitive proxy statement for our 2026 annual meeting, which will be filed no later than 120 days after December 31, 2025, and is incorporated herein by reference.
ITEM 13. CERTAIN RELATIONSHIPS AND RELATED TRANSACTIONS, AND DIRECTOR INDEPENDENCE.
Information required by this item concerning ownership will be set forth under the caption “Corporate Governance — Director Independence” and “Certain Relationships and Related Transactions” in our definitive proxy statement for our 2026 annual meeting, which will be filed no later than 120 days after December 31, 2025, and is incorporated herein by reference.
ITEM 14. PRINCIPAL ACCOUNTANT FEES AND SERVICES.
Information required by this item concerning ownership will be set forth under the caption “Ratification of Appointment of Independent Registered Public Accounting Firm” in our definitive proxy statement for our 2026 annual meeting, which will be filed no later than 120 days after December 31, 2025, and is incorporated herein by reference.
101
PART IV
ITEM 15. EXHIBITS AND FINANCIAL STATEMENT SCHEDULES
(a)Financial Statements and Financial Statement Schedules
The financial statements required by Item 15(a) are filed in Part II Item 8 of this Annual Report on Form 10-K. Schedules not included have been omitted because they are not applicable or because the required information is included in the Consolidated Financial Statements and notes thereto.
Schedule II – Valuation and Qualifying Accounts
AXOGEN, INC.
SCHEDULE II – VALUATION AND QUALIFYING ACCOUNTS
THREE YEARS ENDED DECEMBER 31, 2025, 2024 AND 2023
| (in thousands) | Balance at Beginning of Year | Additions | Deductions (Charge-offs) | Balance at End of Year | |||||||||||||||||||
| Allowance for doubtful accounts | |||||||||||||||||||||||
| 2023 | $ | $ | $ | ( | $ | ||||||||||||||||||
| 2024 | $ | $ | $ | ( | $ | ||||||||||||||||||
| 2025 | $ | $ | $ | ( | $ | ||||||||||||||||||
| Valuation allowance for deferred tax assets | |||||||||||||||||||||||
| 2023 | $ | $ | $ | $ | |||||||||||||||||||
| 2024 | $ | $ | $ | $ | |||||||||||||||||||
| 2025 | $ | $ | $ | ( | $ | ||||||||||||||||||
| Reserve for excess and obsolete inventory | |||||||||||||||||||||||
| 2023 | $ | $ | $ | ( | $ | ||||||||||||||||||
| 2024 | $ | $ | $ | ( | $ | ||||||||||||||||||
| 2025 | $ | $ | $ | ( | $ | ||||||||||||||||||
(b)Exhibits
The following exhibits are included in this Annual Report on Form 10-K or incorporated by reference in the Form 10-K.
| Exhibit Number | Description | Filings Referenced for Incorporation by Reference | ||||||||||||
| 3.1 | November 6, 2019, Form 10-Q, Exhibit 3.1 | |||||||||||||
| 3.2 | March 5, 2024, Form 10-K, Exhibit 3.2 | |||||||||||||
| 4.1 | February 24, 2020, Form 10-K, Exhibit 4.1 | |||||||||||||
| †10.2.1 | October 6, 2011, Form 8-K, Exhibit 10.2 | |||||||||||||
| 10.2.2 | July 11, 2016, Form 8-K, Exhibit 10.2.1 | |||||||||||||
| 10.2.3 | March 14, 2023, Form 10-K Exhibit 10.2.3 | |||||||||||||
102
| Exhibit Number | Description | Filings Referenced for Incorporation by Reference | ||||||||||||
| *10.3 | October 6, 2011, Form 8-K, Exhibit 10.3 | |||||||||||||
| †10.4.1 | October 6, 2011, Form 8-K, Exhibit 10.4.1 | |||||||||||||
| †10.4.2 | October 6, 2011, Form 8-K , Exhibit 10.4.2 | |||||||||||||
| †10.5.1 | October 6, 2011, Form 8-K , Exhibit 10.5 | |||||||||||||
| 10.5.2 | March 15, 2012, Form 10-K, Exhibit 10.5.2 | |||||||||||||
| 10.5.3 | March 1, 2017, Form 10-K, Exhibit 10.5.3 | |||||||||||||
| 10.5.4 | August 8, 2023, Form 10-Q, Exhibit 10.3.3 | |||||||||||||
| 10.6.1 | November 14, 2011, Form 10-Q, Exhibit 10.10 | |||||||||||||
| 10.6.2 | March 15, 2012, Form 10-K, Exhibit 10.23 | |||||||||||||
| 10.6.3 | March 12, 2013, Form 10-K, Exhibit 10.23 | |||||||||||||
| 10.6.4 | March 6, 2014, Form 10-K, Exhibit 10.10.3 | |||||||||||||
| 10.6.5 | March 18, 2016, Form 8-K, Exhibit 10.10.4 | |||||||||||||
| 10.6.6 | December 4, 2020, Form 8-K, Exhibit 10.9.5 | |||||||||||||
| 10.6.7 | July 16, 2021, Form 8-K, Exhibit 10.2 | |||||||||||||
| 10.6.8 | April 13, 2018, Form 8-K, Exhibit 10.1 | |||||||||||||
| 10.6.9 | September 21, 2018, Form 8-K, Exhibit 10.1 | |||||||||||||
| *10.7 | April 8, 2016, Definitive Proxy Statement, Appendix A | |||||||||||||
| *10.8.1 | September 26, 2007, Form 8-K, Exhibit 99.2 | |||||||||||||
| *10.8.2 | March 1, 2017, Form 10-K, Exhibit 10.10.2 | |||||||||||||
| 10.9.1 | April 22, 2015, Form 8-K, Exhibit 10.1 | |||||||||||||
| 10.9.2 | April 22, 2015, Form 8-K, Exhibit 10.2 | |||||||||||||
103
| Exhibit Number | Description | Filings Referenced for Incorporation by Reference | ||||||||||||
| 10.9.3 | October 31, 2016, Form 8-K, Exhibit 10.2.1 | |||||||||||||
| 10.9.4 | November 26, 2018, Form 8-K, Exhibit 10.1 | |||||||||||||
| 10.9.5 | May 8, 2019, Form 10-Q, Exhibit 10.1 | |||||||||||||
| 10.9.6 | January 31, 2022, Form 8-K, Exhibit 10.1 | |||||||||||||
10.9.7 | March 5, 2024, Form 10-K, Exhibit 10.9.7 | |||||||||||||
10.9.8 | March 5, 2024, Form 10-K, Exhibit 10.9.8 | |||||||||||||
| 10.10.1 | November 5, 2015, Form 10-Q, Exhibit 10.3 | |||||||||||||
| 10.10.2 | February 26, 2019, Form 10-K, Exhibit 10.13.1 | |||||||||||||
| 10.10.3 | February 26, 2021, Form 8-K, Exhibit 10.1 | |||||||||||||
| 10.10.4 | August 25, 2022, Form 8-K, Exhibit 10.2 | |||||||||||||
10.10.5 | March 5, 2024, Form 10-K, Exhibit 10.10.5 | |||||||||||||
| 10.11 | March 1, 2017, Form 10-K, Exhibit 10.22 | |||||||||||||
| *†10.12 | March 1, 2017, Form 10-K, Exhibit 10.23 | |||||||||||||
| *10.13 | March 1, 2018, Form 10-K, Exhibit 10.26 | |||||||||||||
| *10.14 | March 1, 2017, Form 10-K, Exhibit 10.28 | |||||||||||||
| 10.15 | April 13, 2018, Form 8-K, Exhibit 10.1 | |||||||||||||
| 10.16 | September 21, 2018, Form 8-K, Exhibit 10.1 | |||||||||||||
| 10.17.1 | September 21, 2018, Form 8-K, Exhibit 10.2 | |||||||||||||
| 10.17.2 | July 16, 2021, Form 8-K, Exhibit 10.1 | |||||||||||||
| *10.18 | October 29, 2018, Form 8-K, Exhibit 10.3 | |||||||||||||
104
| Exhibit Number | Description | Filings Referenced for Incorporation by Reference | ||||||||||||
| *10.19 | October 29, 2018, Form 8-K, Exhibit 10.4 | |||||||||||||
| 10.2 | April 7, 2017, Definitive Proxy Statement, Appendix B | |||||||||||||
| 10.21.1 | November 26, 2018, Form 8-K, Exhibit 10.3 | |||||||||||||
| 10.21.2 | November 26, 2018, Form 8-K, Exhibit 10.4 | |||||||||||||
| 10.21.3 | March 14, 2023, Form 10-K, Exhibit 10.25.3 | |||||||||||||
| *10.22 | February 26, 2019, Form 10-K, Exhibit 10.47 | |||||||||||||
| 10.23 | July 9, 2019, Form 8-K, Exhibit 10.1 | |||||||||||||
| *10.24 | November 6, 2019, Form 10-Q, Exhibit 10.2 | |||||||||||||
| ††10.25.1 | February 24, 2020, Form 10-K, Exhibit 10.51 | |||||||||||||
| 10.25.2 | March 14, 2023, Form 10-K, Exhibit 10.25.3 | |||||||||||||
| 10.25.3 | August 8, 2023, Form 10-Q, Exhibit 10.2.2 | |||||||||||||
| 10.26.1 | July 1, 2020, Form 8-K, Exhibit 10.1 | |||||||||||||
| 10.26.2 | June 30, 2023, Form 8-K, Exhibit 10.1 | |||||||||||||
| 10.27 | July 1, 2020, Form 8-K, Exhibit 10.2 | |||||||||||||
| 10.28 | July 1, 2020, Form 8-K, Exhibit 10.3 | |||||||||||||
| 10.29 | July 1, 2020, Form 8-K, Exhibit 10.4 | |||||||||||||
| *10.30 | October 29, 2020, Form 8-K, Exhibit 10.1 | |||||||||||||
| *10.31 | December 6, 2023, Form 8-K, Exhibit 10.1 | |||||||||||||
| 10.32.1 | October 30, 2020, Form 10-Q, Exhibit 10.1 | |||||||||||||
| 10.32.2 | August 25, 2022, Form 8-K, Exhibit 10.1 | |||||||||||||
| *10.33 | March 31, 2021, Definitive Proxy Statement, Appendix A | |||||||||||||
| *10.34 | April 1, 2022, Form 8-K, Exhibit 10.1 | |||||||||||||
105
| Exhibit Number | Description | Filings Referenced for Incorporation by Reference | ||||||||||||
| *10.35 | April 1, 2022, Form 8-K, Exhibit 10.2 | |||||||||||||
| *10.36 | April 1, 2022, Form 8-K, Exhibit 10.3 | |||||||||||||
| *10.37 | April 1, 2022, Form 8-K, Exhibit 10.4 | |||||||||||||
| *10.38 | April 14, 2022, Definitive Proxy Statement, Appendix A | |||||||||||||
| 10.39 | March 5, 2024, Form 10-K, Exhibit 10.51 | |||||||||||||
| *10.40 | March 5, 2024, Form 10-K, Exhibit 10.52 | |||||||||||||
| *10.41 | March 5, 2024, Form 10-K, Exhibit 10.53 | |||||||||||||
| *†††10.42 | March 5, 2024, Form 10-K, Exhibit 10.54 | |||||||||||||
| *†††10.43 | March 5, 2024, Form 10-K, Exhibit 10.55 | |||||||||||||
| *10.44.1 | March 5, 2024, Form 10-K, Exhibit 10.56 | |||||||||||||
| *10.44.2 | March 5, 2024, Form 10-K, Exhibit 10.57 | |||||||||||||
| *10.45 | March 5, 2024, Form 10-K, Exhibit 10.58 | |||||||||||||
| *10.46.1 | March 5, 2024, Form 10-K, Exhibit 10.59 | |||||||||||||
| *10.46.2 | March 5, 2024, Form 10-K, Exhibit 10.60 | |||||||||||||
| *10.47 | March 5, 2024, Form 10-K, Exhibit 10.61 | |||||||||||||
| *10.48.1 | March 5, 2024, Form 10-K, Exhibit 10.62 | |||||||||||||
| *10.48.2 | March 5, 2024, Form 10-K, Exhibit 10.63 | |||||||||||||
| *10.49 | March 5, 2024, Form 10-K, Exhibit 10.64 | |||||||||||||
| *10.50 | January 4, 2024, Form 8-K, Exhibit 10.1 | |||||||||||||
| *10.51 | August 9, 2024, Form 8-K, Exhibit 10.1 | |||||||||||||
106
| Exhibit Number | Description | Filings Referenced for Incorporation by Reference | ||||||||||||
| *10.52 | August 9, 2024, Form 8-K, Exhibit 10.2 | |||||||||||||
| *10.53 | August 9, 2024, Form 8-K, Exhibit 10.3 | |||||||||||||
| *10.54 | April 24, 2024, Definitive Proxy Statement, Appendix A | |||||||||||||
| *10.55 | April 30, 2025, Definitive Proxy Statement, Appendix A | |||||||||||||
| *10.56 | May 8, 2025, Form 8-K, Exhibit 10.1 | |||||||||||||
| *10.57 | May 8, 2025, Form 8-K, Exhibit 10.2 | |||||||||||||
| *10.58 | December 5, 2025, Form 8-K, Exhibit 10.2 | |||||||||||||
| *†10.59 | May 8, 2025, Form 10-Q, Exhibit 10.1 | |||||||||||||
| *10.60 | May 8, 2025, Form 10-Q, Exhibit 10.2 | |||||||||||||
| 19.1 | February 26, 2025, Form 10-K, Exhibit 19.1 | |||||||||||||
| 21.1 | Filed herewith | |||||||||||||
| 23.1 | Filed herewith | |||||||||||||
| 24.1 | Included on signature page | |||||||||||||
| 31.1 | Filed herewith | |||||||||||||
| 31.2 | Filed herewith | |||||||||||||
| 32.1 | Furnished herewith | |||||||||||||
| 97 | March 5, 2024, Form 10-K, Exhibit 97 | |||||||||||||
| 101 | Inline XBRL Document Set for the consolidated financial statements and accompanying notes in Part II, Item 8, “Financial Statements and Supplementary Data” of this Annual Report on Form 10-K | Filed herewith | ||||||||||||
| 101.INS | XBRL Instance Document – The instance document does not appear in the Interactive Data File because its XBRL tags are embedded within the Inline XBRL document | Filed herewith | ||||||||||||
| 101.SCH | Inline XBRL Taxonomy Extension Schema Document | Filed herewith | ||||||||||||
| 101.CAL | Inline XBRL Taxonomy Extension Calculation Linkbase Document | Filed herewith | ||||||||||||
| 101.DEF | Inline XBRL Taxonomy Extension Definition Linkbase Document | Filed herewith | ||||||||||||
| 101.LAB | Inline XBRL Extension Labels Linkbase | Filed herewith | ||||||||||||
| 101.PRE | Inline XBRL Taxonomy Extension Presentation Linkbase Document | Filed herewith | ||||||||||||
107
| Exhibit Number | Description | Filings Referenced for Incorporation by Reference | ||||||||||||
| 104 | Inline XBRL for the cover page of this Annual Report on Form 10-K, included in the Exhibit 101 Inline XBRL Document Set | Filed herewith | ||||||||||||
| * | Management contract or compensatory plan or arrangement. | ||||
| † | Confidential treatment has been granted for portions of this Exhibit pursuant to Rule 24b-2 under the Securities Exchange Act of 1934, as amended. The confidential portions have been deleted and filed separately with the U.S. Securities and Exchange Commission. | ||||
| †† | Confidential treatment has been requested as to certain portions, which portions have been omitted and filed separately with the Securities and Exchange Commission. | ||||
| ††† | Portions of this exhibit have been omitted pursuant to Item 601(b)(10) of Regulation S-K because the information is not material and is the type of information that the Company treats as private or confidential. The Company agrees to furnish an unredacted copy of this exhibit on a supplemental basis to the SEC or its staff upon request. | ||||
ITEM 16. Form 10-K Summary
None.
108
SIGNATURES
Pursuant to the requirements of Section 13 or 15(d) of the Securities Exchange Act of 1934, as amended, the registrant has duly caused this report to be signed on its behalf by the undersigned, thereunto duly authorized.
| AXOGEN, INC | |||||
| /s/ Michael J. Dale | |||||
| Michael J. Dale | |||||
Chief Executive Officer, President and Director | |||||
| February 24, 2026 | |||||
KNOW ALL PERSONS BY THESE PRESENTS, that each person whose signature appears below constitutes and appoints Michael J. Dale, Lindsey Hartley and Marc A. Began (each with full power to act alone), as his or her true and lawful attorney-in-fact and agent, with full powers of substitution and re-substitution, for him and in his name, place and stead, in any and all capacities, to sign any and all amendments to the Annual Report on Form 10-K of Axogen, Inc., and to file the same, with all exhibits thereto, and other documents in connection therewith, with the Securities and Exchange Commission, granting unto said attorney-in-fact and agent full power and authority to do and perform each and every act and thing requisite or necessary to be done in and about the premises, as fully to all intents and purposes as he might or could do in person, hereby ratifying and confirming all that said attorney-in-fact and agent, or their substitute or substitutes, lawfully do or cause to be done by virtue hereof.
In accordance with the Securities Exchange Act of 1934, as amended, this report has been signed below by the following persons on behalf of the registrant and in the capacities and on the dates indicated.
| /s/ Michael J. Dale | February 24, 2026 | |||||||
Michael J. Dale, Chief Executive Officer, President and Director (Principal Executive Officer) | ||||||||
| /s/ Lindsey Hartley | February 24, 2026 | |||||||
| Lindsey Hartley, Chief Financial Officer (Principal Financial and Accounting Officer) | ||||||||
/s/ William Burke | February 24, 2026 | |||||||
William Burke Director | ||||||||
| /s/ John H. Johnson | February 24, 2026 | |||||||
John H. Johnson Director | ||||||||
| /s/ Alan M. Levine | February 24, 2026 | |||||||
| Alan M. Levine Director | ||||||||
/s/ Paul G. Thomas | February 24, 2026 | |||||||
| Paul G. Thomas Chairman of the Board | ||||||||
| /s/ Joseph A. Tyndall | February 24, 2026 | |||||||
Joseph A. Tyndall Director | ||||||||
| /s/ Kathy Weiler | February 24, 2026 | |||||||
| Kathy Weiler Director | ||||||||
| /s/ Amy Wendell | February 24, 2026 | |||||||
| Amy Wendell Director | ||||||||
109