EX-99.3 - CORPORATE PRESENTATION DATED APRIL 2026
Published on April 28, 2026

Making Nerve Repair an Expected Standard of Care A commitment to restoring health, improving quality of life, and advancing peripheral nerve care for every patient. April 2026

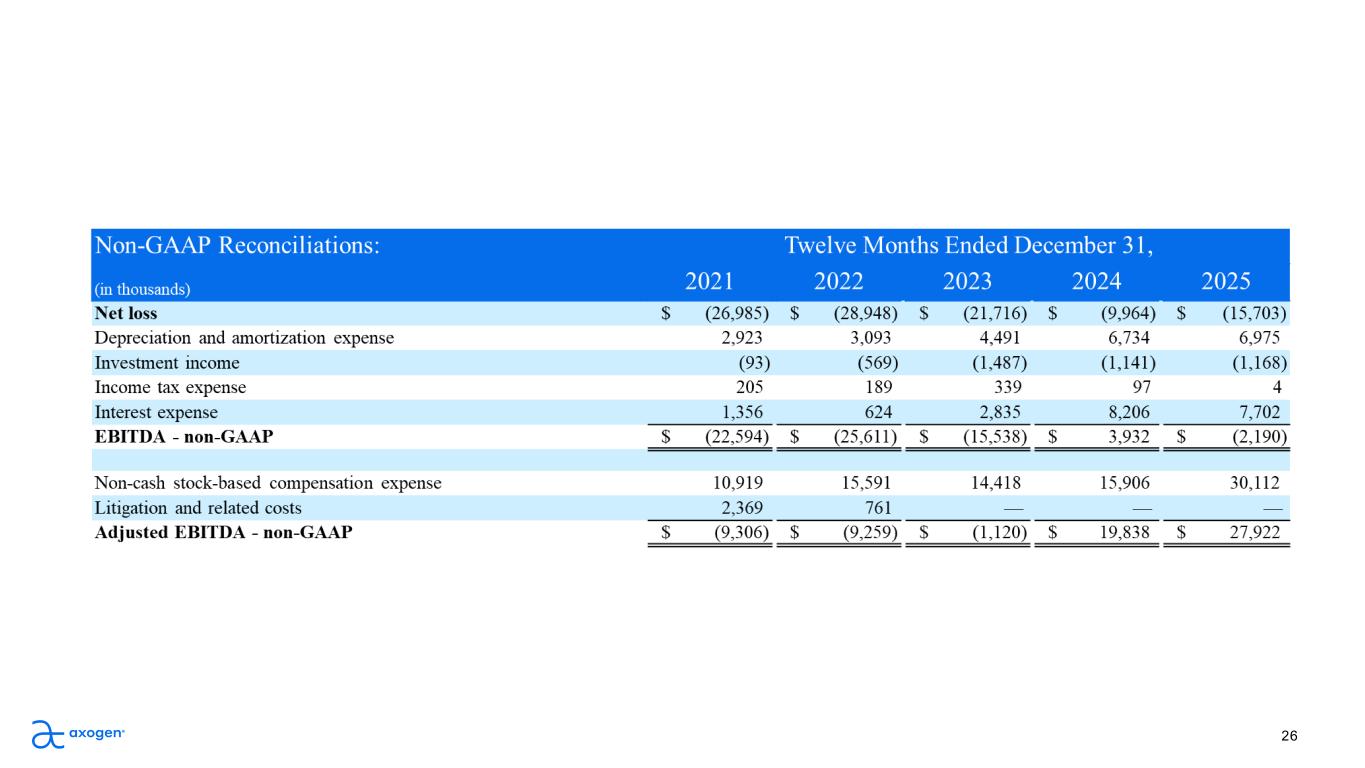
Forward-looking Statements This presentation contains “forward-looking” statements as defined in the Private Securities Litigation Reform Act of 1995, which are statements that are not historical facts and relate to future conditions, events, or results. These statements are based on management's current expectations or predictions of future conditions, events, or results based on various assumptions and management's estimates of trends and economic factors in the markets in which we are active, as well as our business plans. Words such as “expects,” “anticipates,” “objectives,” “intends,” “plans,” “believes,” “seeks,” “estimates,” “projects,” “forecasts,” “continue,” “may,” “should,” “will,” “goals,” and variations of such words and similar expressions are intended to identify such forward-looking statements. Forward-looking statements include, but are not limited to, statements related to: clinical development activities, including expansion into prostate applications; commercial growth initiatives, including planned expansion of breast and extremities sales specialists; market development opportunities; expectations regarding disciplined, profitable growth and margin improvement; financial guidance and outlook for 2026, including, but not limited to, projected revenue, growth, free cash flows, gross margins, and other operating performance and financial metrics; and statements regarding our training and education initiatives, reimbursement and market access efforts, and research and development activities. Actual results or events could differ materially from those described in any forward-looking statements as a result of various factors, including, without limitation, risks related to global supply chain conditions, inflationary pressures, hospital staffing challenges, product development and product potential, clinical enrollment timing and outcomes, regulatory processes and approvals, financial performance, sales growth, surgeon and product adoption, market awareness of our products, data validation, our visibility at and sponsorship of conferences and educational events, geopolitical and macroeconomic conditions, including armed conflicts and government actions or policies that may affect our business, tax position, or regulatory processes, as well as those risk factors described under Part I, Item 1A., “Risk Factors,” of our most recent Annual Report on Form 10-K, subsequent Quarterly Reports on Form 10-Q, and other filings made from time to time with the Securities and Exchange Commission. Forward-looking statements are not a guarantee of future performance, and actual results may differ materially from those projected. Forward-looking statements speak only as of the date they are made and, except as required by applicable law, we assume no responsibility to publicly update or revise any forward-looking statements. 2 About Non-GAAP Financial Measures To supplement our condensed consolidated financial statements, we use the non-GAAP financial measures of EBITDA, which measures earnings before interest, income taxes, depreciation and amortization, EBITDA margin, Adjusted EBITDA, which further excludes non-cash stock-based compensation expense and litigation related costs, and Adjusted EBITDA margin. We also use the Free Cash Flow metric, which corresponds to the net cash provided by (used in) operating activities less cash used for purchases of property and equipment and intangible assets. These non- GAAP measures are not based on any comprehensive set of accounting rules or principles and should not be considered a substitute for, or superior to, financial measures calculated in accordance with GAAP and may be different from non-GAAP measures used by other companies. In addition, these non-GAAP measures should be read in conjunction with our financial statements prepared in accordance with GAAP. The reconciliations of the non-GAAP measures to the most directly comparable financial measures calculated and presented in accordance with GAAP should be carefully evaluated. We use these non-GAAP financial measures for financial and operational decision-making and as a means to evaluate period-to-period comparisons. We believe that these non-GAAP financial measures provide meaningful supplemental information regarding our performance and that both management and investors benefit from referring to these non-GAAP financial measures in assessing our performance and when planning, forecasting, and analyzing future periods. We believe these non-GAAP financial measures are useful to investors because (1) they allow for greater transparency with respect to key metrics used by management in its financial and operational decision-making and (2) they are used by our institutional investors and the analyst community to help them analyze the performance of our business, the Company’s cash available for operations, and the Company’s ability to meet future capital expenditure and working capital requirements.

3 To restore health and improve quality of life by making restoration of peripheral nerve function an expected standard of care. Our Mission
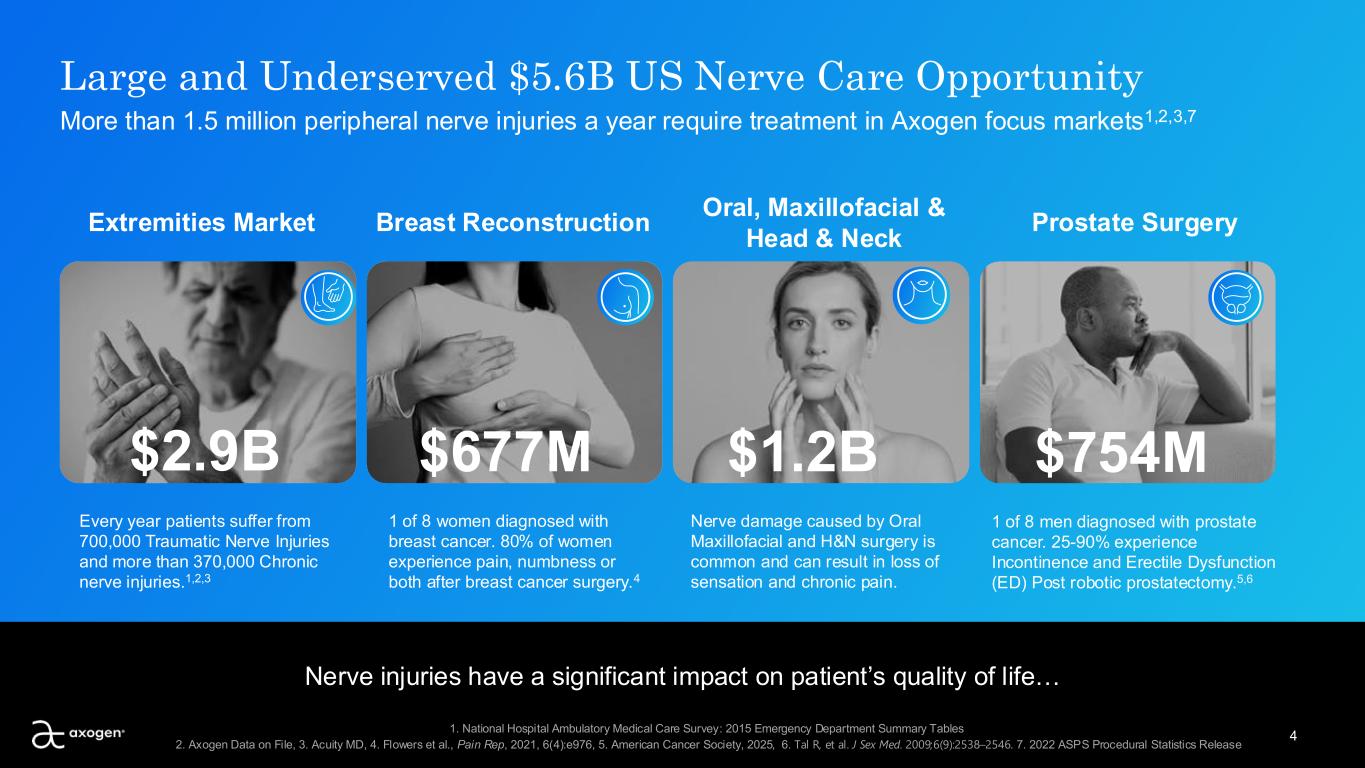
4 Large and Underserved $5.6B US Nerve Care Opportunity More than 1.5 million peripheral nerve injuries a year require treatment in Axogen focus markets1,2,3,7 Extremities Market Every year patients suffer from 700,000 Traumatic Nerve Injuries and more than 370,000 Chronic nerve injuries.1,2,3 Breast Reconstruction 1 of 8 women diagnosed with breast cancer. 80% of women experience pain, numbness or both after breast cancer surgery.4 Oral, Maxillofacial & Head & Neck Nerve damage caused by Oral Maxillofacial and H&N surgery is common and can result in loss of sensation and chronic pain. Prostate Surgery $1.2B$677M $754M Nerve injuries have a significant impact on patient’s quality of life… 1 of 8 men diagnosed with prostate cancer. 25-90% experience Incontinence and Erectile Dysfunction (ED) Post robotic prostatectomy.5,6 $2.9B 1. National Hospital Ambulatory Medical Care Survey: 2015 Emergency Department Summary Tables 2. Axogen Data on File, 3. Acuity MD, 4. Flowers et al., Pain Rep, 2021, 6(4):e976, 5. American Cancer Society, 2025, 6. Tal R, et al. J Sex Med. 2009;6(9):2538–2546. 7. 2022 ASPS Procedural Statistics Release
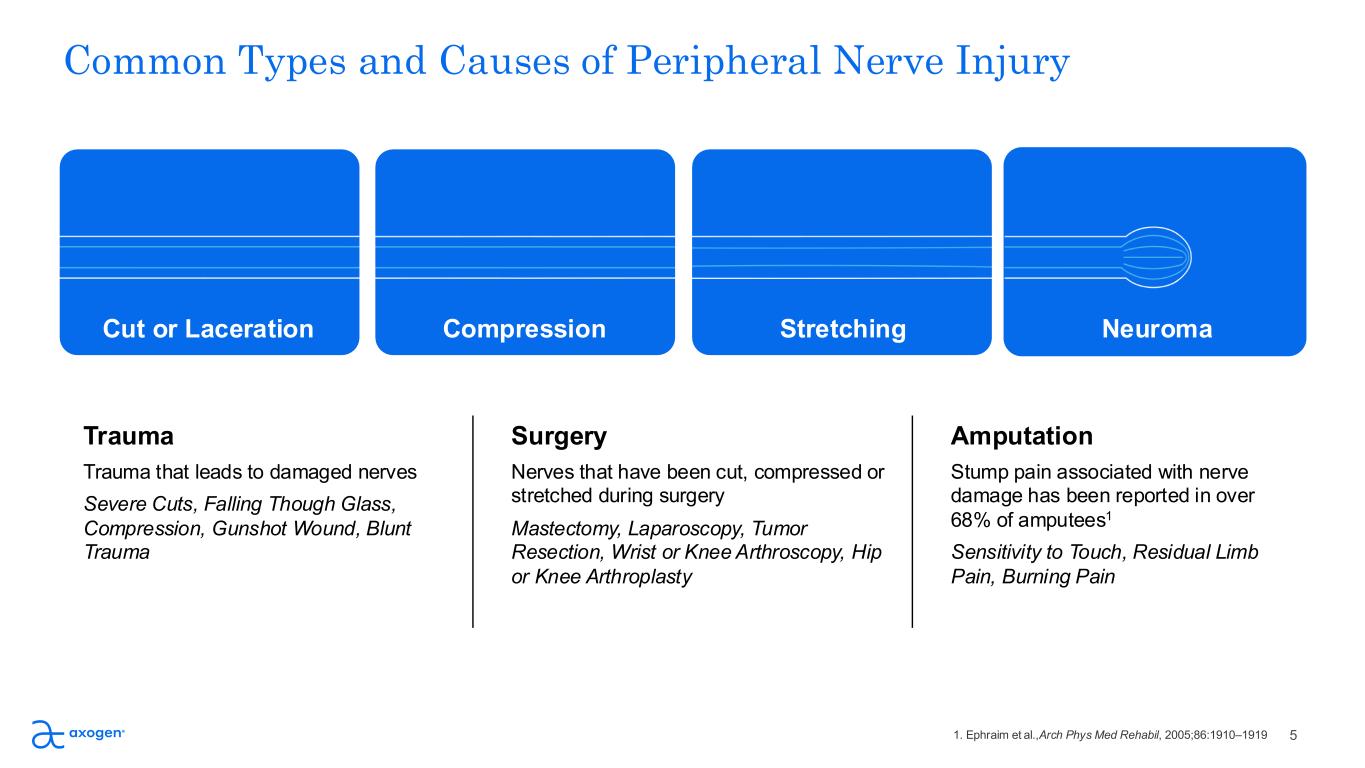
Common Types and Causes of Peripheral Nerve Injury Cut or Laceration Compression Stretching Neuroma Trauma Trauma that leads to damaged nerves Severe Cuts, Falling Though Glass, Compression, Gunshot Wound, Blunt Trauma Amputation Stump pain associated with nerve damage has been reported in over 68% of amputees1 Sensitivity to Touch, Residual Limb Pain, Burning Pain Surgery Nerves that have been cut, compressed or stretched during surgery Mastectomy, Laparoscopy, Tumor Resection, Wrist or Knee Arthroscopy, Hip or Knee Arthroplasty 51. Ephraim et al.,Arch Phys Med Rehabil, 2005;86:1910–1919
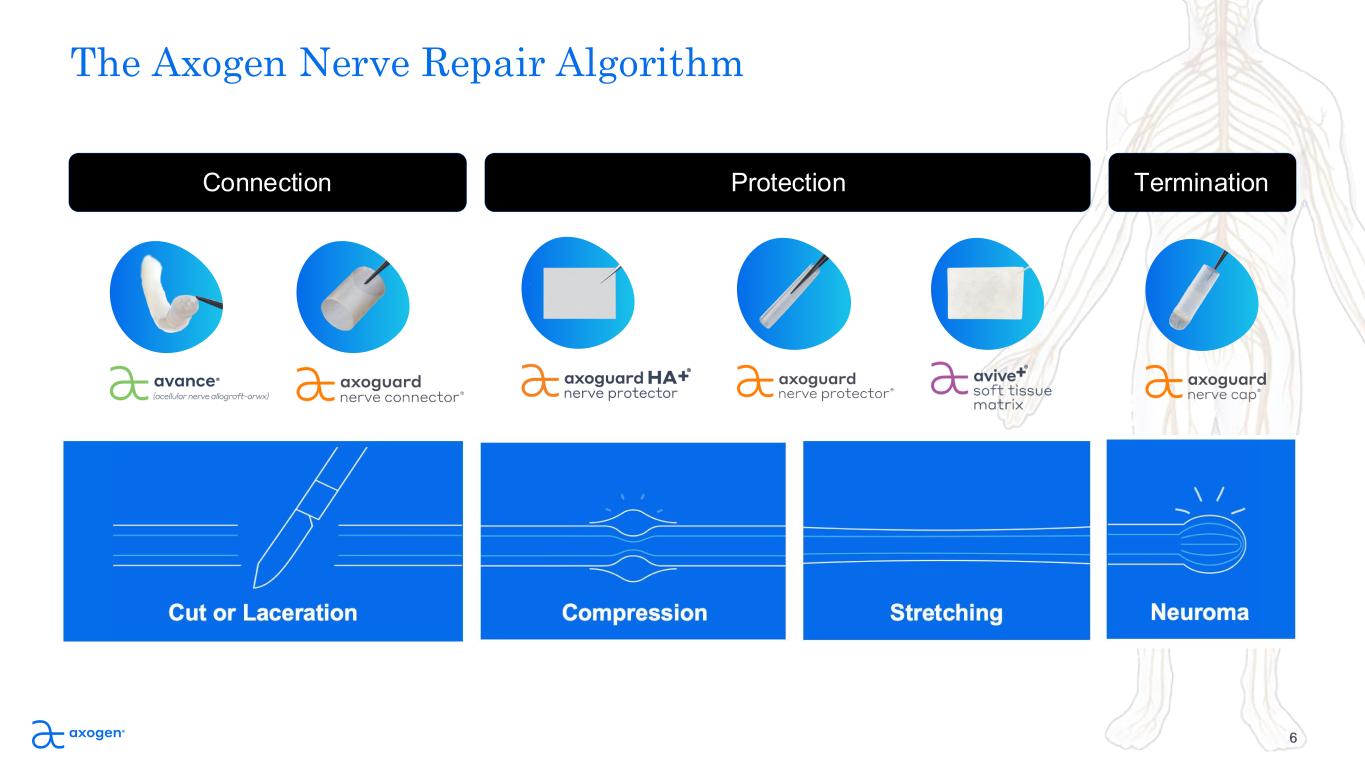
The Axogen Nerve Repair Algorithm 6 Connection Protection Termination
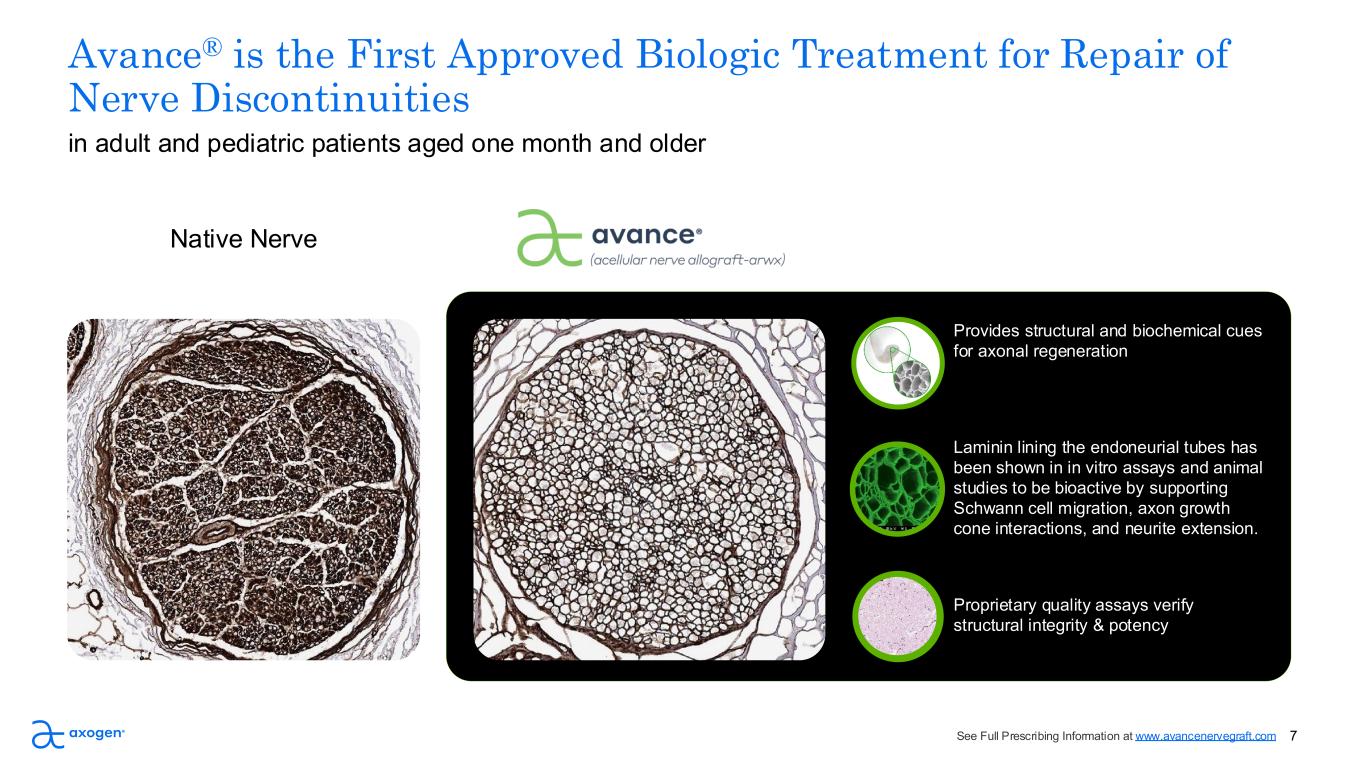
Avance® is the First Approved Biologic Treatment for Repair of Nerve Discontinuities i l i reat ent for e air of Provides structural and biochemical cues for axonal regeneration 7 Native Nerve in adult and pediatric patients aged one month and older Laminin lining the endoneurial tubes has been shown in in vitro assays and animal studies to be bioactive by supporting Schwann cell migration, axon growth cone interactions, and neurite extension. Proprietary quality assays verify structural integrity & potency See Full Prescribing Information at www.avancenervegraft.com
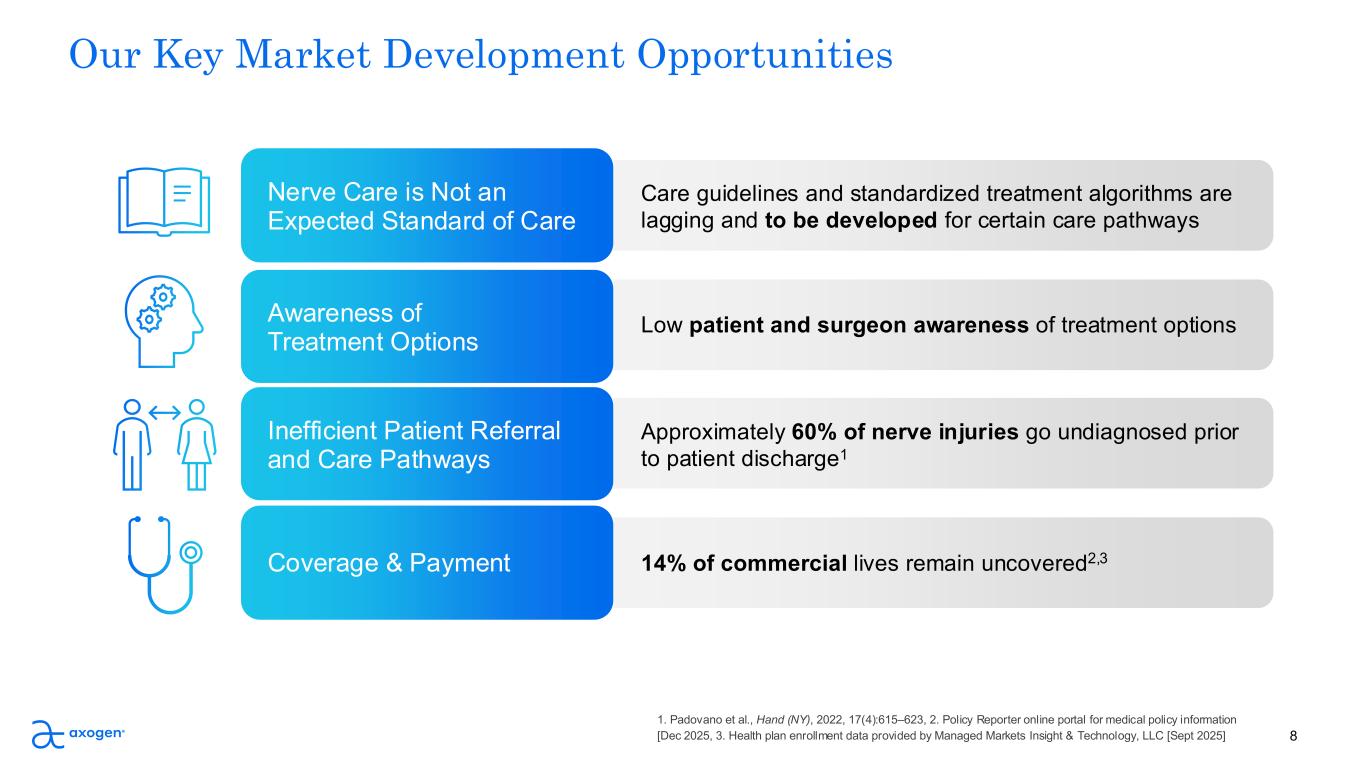
8 Our Key Market Development Opportunities Care guidelines and standardized treatment algorithms are lagging and to be developed for certain care pathways Nerve Care is Not an Expected Standard of Care Low patient and surgeon awareness of treatment options Awareness of Treatment Options Approximately 60% of nerve injuries go undiagnosed prior to patient discharge1 Inefficient Patient Referral and Care Pathways 14% of commercial lives remain uncovered2,3Coverage & Payment 1. Padovano et al., Hand (NY), 2022, 17(4):615–623, 2. Policy Reporter online portal for medical policy information [Dec 2025, 3. Health plan enrollment data provided by Managed Markets Insight & Technology, LLC [Sept 2025]

9 2025-2028 Strategic Plan Priorities Commercial infrastructure and salesforce expansion Continuous business model and customer creation process optimization by market Product development to drive better benefit versus risk profiles in nerve care Elective and planned procedure focus Prostate market development CAGR 15 - 20% Commercial Excellence Commercial Expansion Innovation MarketsGrowth Level 1 clinical evidence generation for societal support, standard of care & coverage requirements Standard of Care
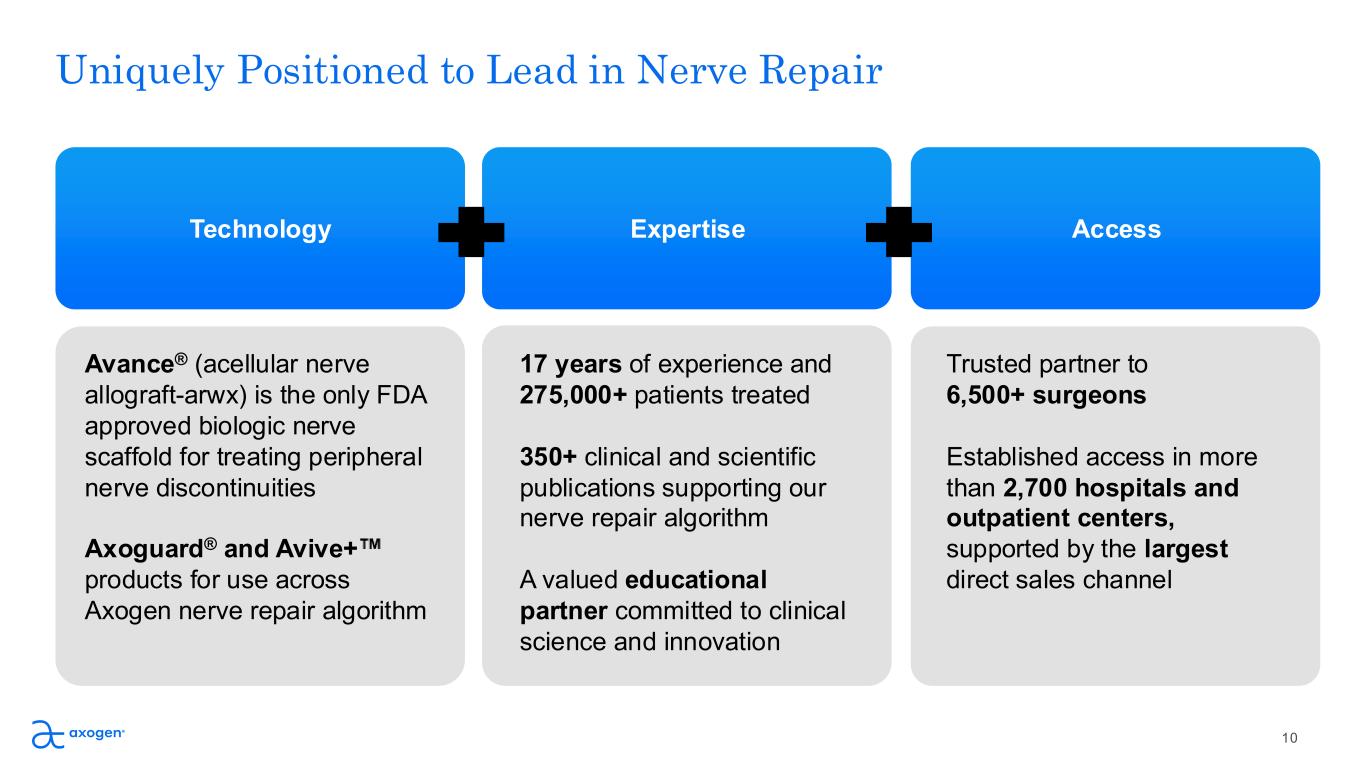
Uniquely Positioned to Lead in Nerve Repair Avance® (acellular nerve allograft-arwx) is the only FDA approved biologic nerve scaffold for treating peripheral nerve discontinuities Axoguard® and Avive+ products for use across Axogen nerve repair algorithm 17 years of experience and 275,000+ patients treated 350+ clinical and scientific publications supporting our nerve repair algorithm A valued educational partner committed to clinical science and innovation Trusted partner to 6,500+ surgeons Established access in more than 2,700 hospitals and outpatient centers, supported by the largest direct sales channel Technology Expertise Access 10

Elevating Nerve IQ 11 75% of hand fellows trained 13 Professional education programs across our markets with more than 225 surgeons trained 117 Regional programs helping to enhance the micro surgical skills of surgeons Surgeons trained on Axogen’s nerve repair algorithm 55 Faculty educational partners of leading nerve repair thought leaders 2025 ACTIVITY 1,400 Education & Training are Key to Market Development
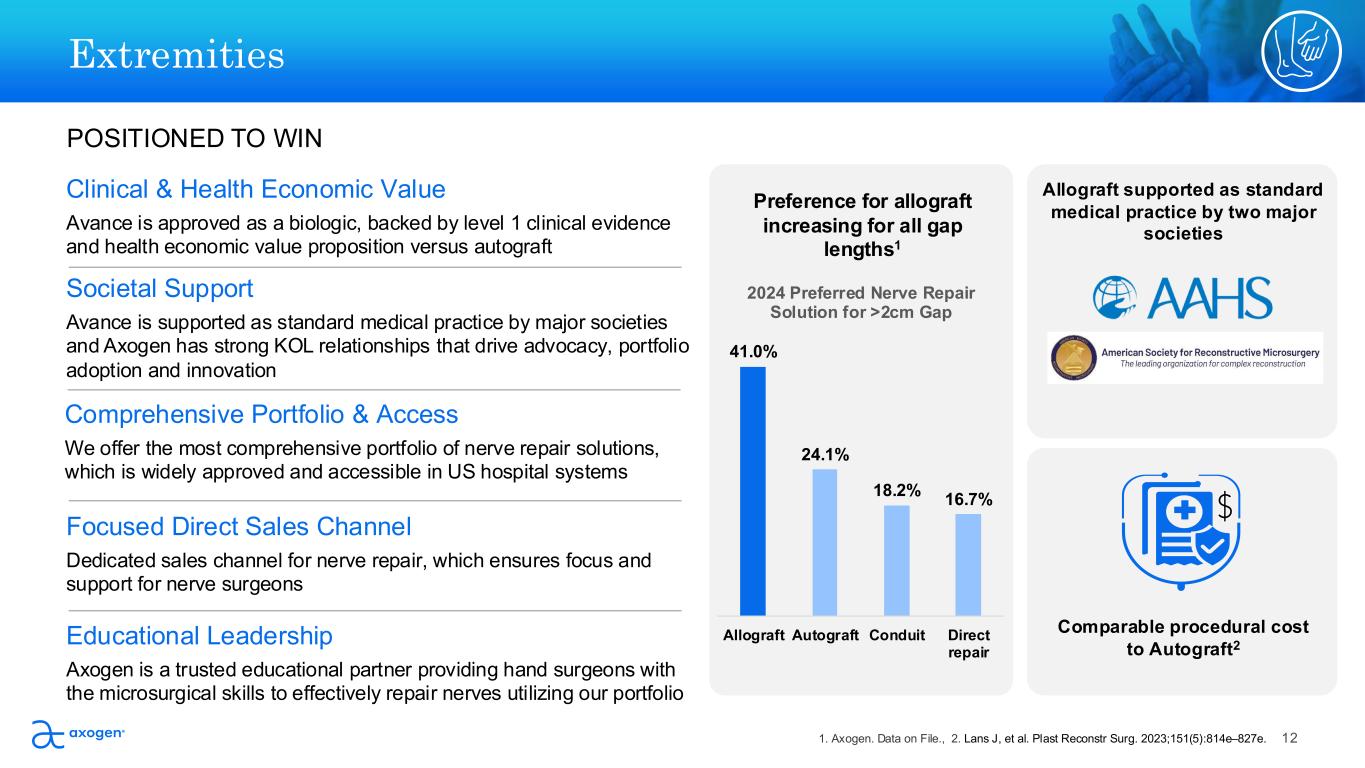
Societal Support Avance is supported as standard medical practice by major societies and Axogen has strong KOL relationships that drive advocacy, portfolio adoption and innovation POSITIONED TO WIN Preference for allograft increasing for all gap lengths1 Comparable procedural cost to Autograft2 Allograft supported as standard medical practice by two major societies Educational Leadership Axogen is a trusted educational partner providing hand surgeons with the microsurgical skills to effectively repair nerves utilizing our portfolio Comprehensive Portfolio & Access We offer the most comprehensive portfolio of nerve repair solutions, which is widely approved and accessible in US hospital systems Focused Direct Sales Channel Dedicated sales channel for nerve repair, which ensures focus and support for nerve surgeons Clinical & Health Economic Value Avance is approved as a biologic, backed by level 1 clinical evidence and health economic value proposition versus autograft Extremities 41.0% 24.1% 18.2% 16.7% Allograft Autograft Conduit Direct repair 2024 Preferred Nerve Repair Solution for >2cm Gap 121. Axogen. Data on File., 2. Lans J, et al. Plast Reconstr Surg. 2023;151(5):814e–827e.

POSITIONED TO WIN Growing body of evidence supporting the benefits or nerve reconstruction Grow presence in H&N oncologic procedures Opportunity to build patient and surgeon awareness of the patient QoL impact Expand educational capacity & programs Societal Support OMF societal support for nerve repair and included in AAOMS ParCare Guidelines can be leveraged to influence H&N societies Clinical Education Leadership Axogen has developed and executed on high quality national attending level professional education programs with proven post program adoption Clinical Evidence Independent clinical evidence with strong outcome data in benign mandible reconstruction and lingual nerve repair Direct Sales Channel Axogen has a large direct sales channel to service the highly concentrated market Oral Maxillofacial and Head & Neck 13

POSITIONED TO WIN Specialized Sales & Marketing Dedicated, deeply knowledgeable sales team enables effective surgeon development, support and market penetration Marketing expertise in the creation of strategies, tactics, tools, and resources support the sales process Established, predictable customer creation process Proven Patient Activation Strategy Axogen’s marketing team excels at translating complex medical information into patient-friendly content, raising awareness and driving demand for Resensation® Clinical Education Leadership A collaborative approach to training has resulted in surgeon advocacy, high adoption rates and strong customer loyalty Standardized, branded procedure 80%+ surgeon adoption after training Breast 14 25K+ monthly website visitors 3K+ monthly visitors to surgeon locator

POSITIONED TO WIN Clinical Education Leadership Extensive expertise in developing standardized surgical techniques and building comprehensive training courses to equip surgeons with the necessary skills and knowledge to successfully perform the procedures Nerve Repair Portfolio Axogen’s nerve repair portfolio has the potential to help surgeons address nerve protection and reconstruction needs in robotic assisted radical prostatectomy Patient Awareness & Activation Marketing team excels at executing campaigns that raise awareness of clinical problems and drives patient demand for new treatments 15 Prostate
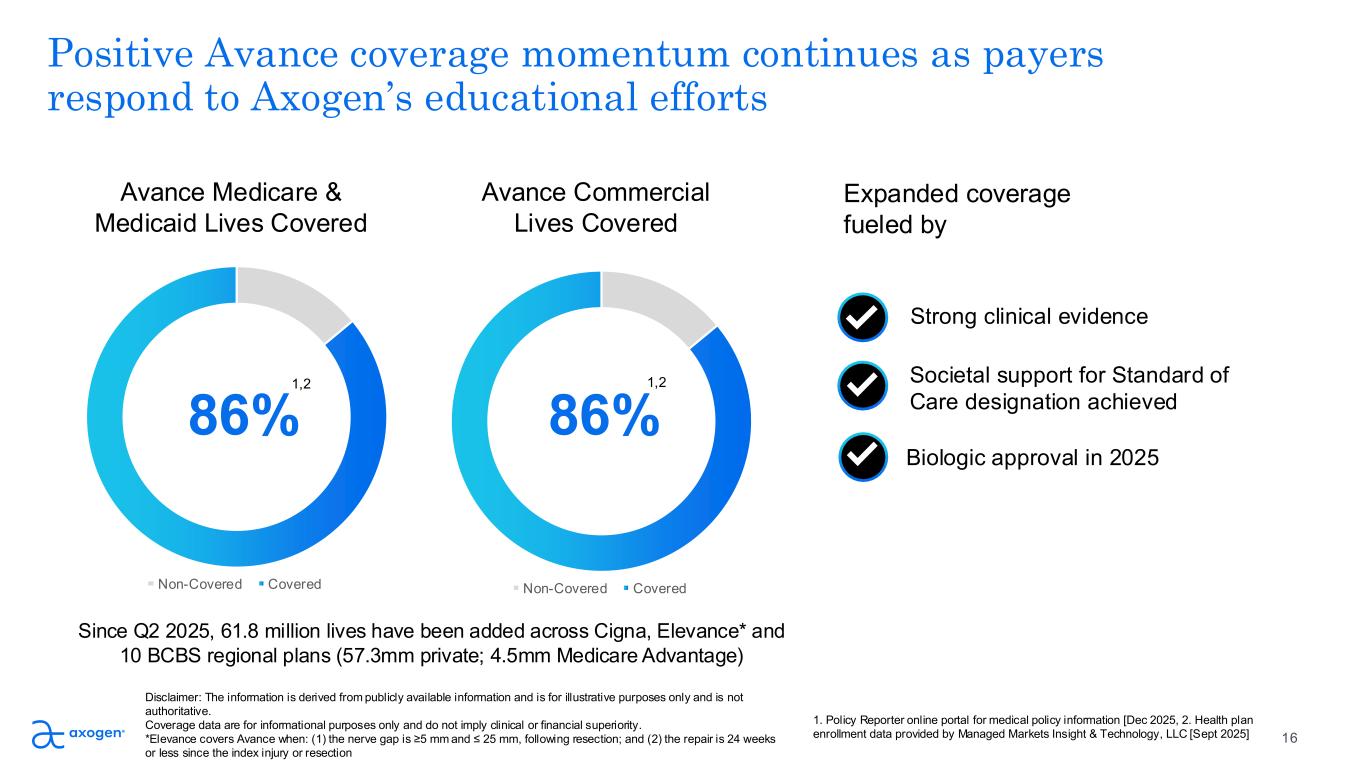
Non-Covered Covered Societal support for Standard of Care designation achieved Non-Covered Covered Positive Avance coverage momentum continues as payers respond to Axogen’s educational efforts 86% 86% Expanded coverage fueled by Biologic approval in 2025 Strong clinical evidence Avance Medicare & Medicaid Lives Covered Since Q2 2025, 61.8 million lives have been added across Cigna, Elevance* and 10 BCBS regional plans (57.3mm private; 4.5mm Medicare Advantage) 16 Disclaimer: The information is derived from publicly available information and is for illustrative purposes only and is not authoritative. Coverage data are for informational purposes only and do not imply clinical or financial superiority. *Elevance covers Avance when: (1) the nerve gap is ≥5 mm and ≤ 25 mm, following resection; and (2) the repair is 24 weeks or less since the index injury or resection 1,2 1,2 Avance Commercial Lives Covered 1. Policy Reporter online portal for medical policy information [Dec 2025, 2. Health plan enrollment data provided by Managed Markets Insight & Technology, LLC [Sept 2025]

National Average2025 FACILITY PAYMENT In 2026, CMS Improved Facility Payments by Creating a New Outpatient Code Group The code for Allograft 64912 (Avance) is not specific to a clinical application and can therefore be applied to nerve repair in all anatomical locations CPT Code Descriptor C-APC Reimbursement Hospital Outpatient Reimbursement Ambulatory Surgery Center 64912 Allograft nerve repair 5432 $8,965 $6,157* +40% YoY +96% Since 2019 +35% YoY +221% Since 2019 National Average Disclaimer: The information is derived from publicly available information and is for illustrative purposes only and is not authoritative. 17 * Device intensive status achieved in 2020 New Level 3 Nerve Procedure Code increases reimbursement for hospitals and ASC’s
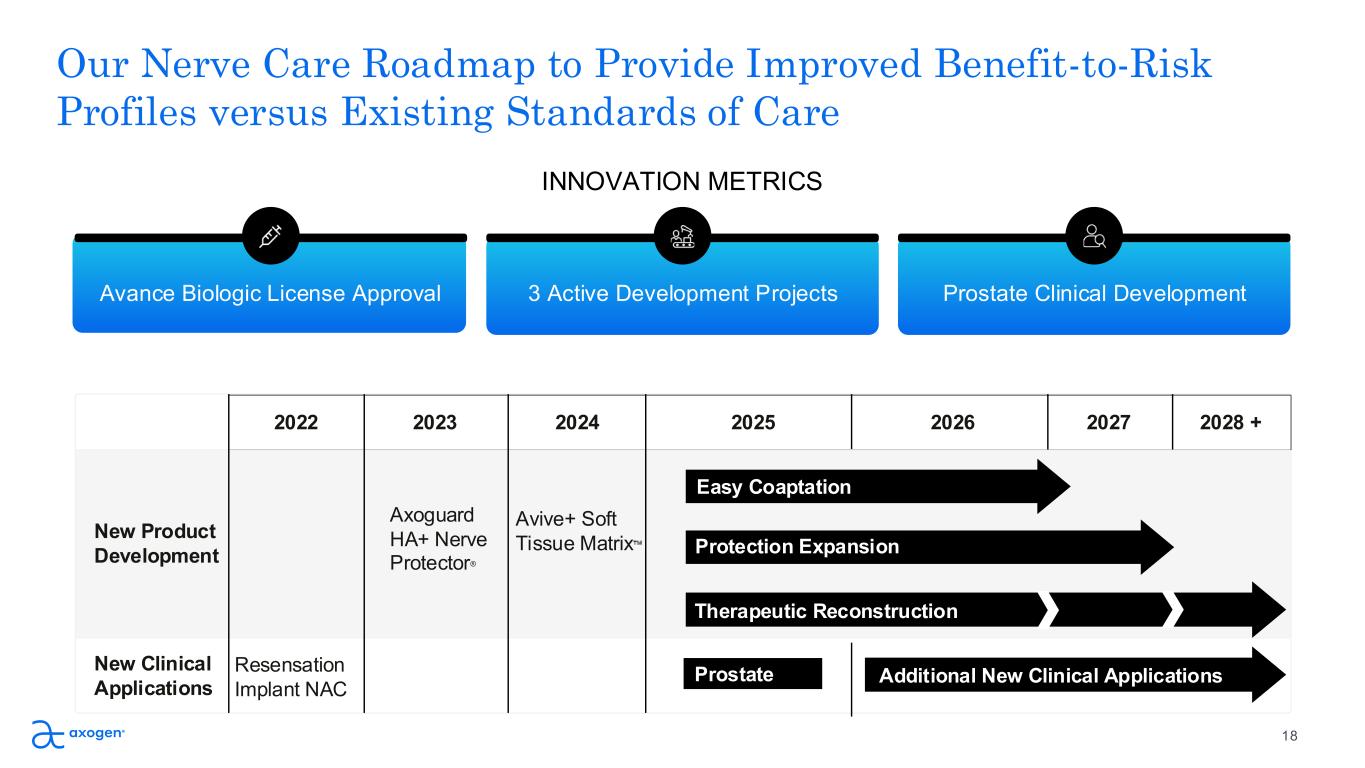
18 INNOVATION METRICS Avance Biologic License Approval 3 Active Development Projects Prostate Clinical Development 2022 2023 2024 2025 2026 2027 2028 + New Product Development Axoguard HA+ Nerve Protector® Avive+ Soft Tissue Matrix New Clinical Applications Resensation Implant NAC Prostate Our Nerve Care Roadmap to Provide Improved Benefit-to-Risk Profiles versus Existing Standards of Care Easy Coaptation Protection Expansion Therapeutic Reconstruction Additional New Clinical Applications
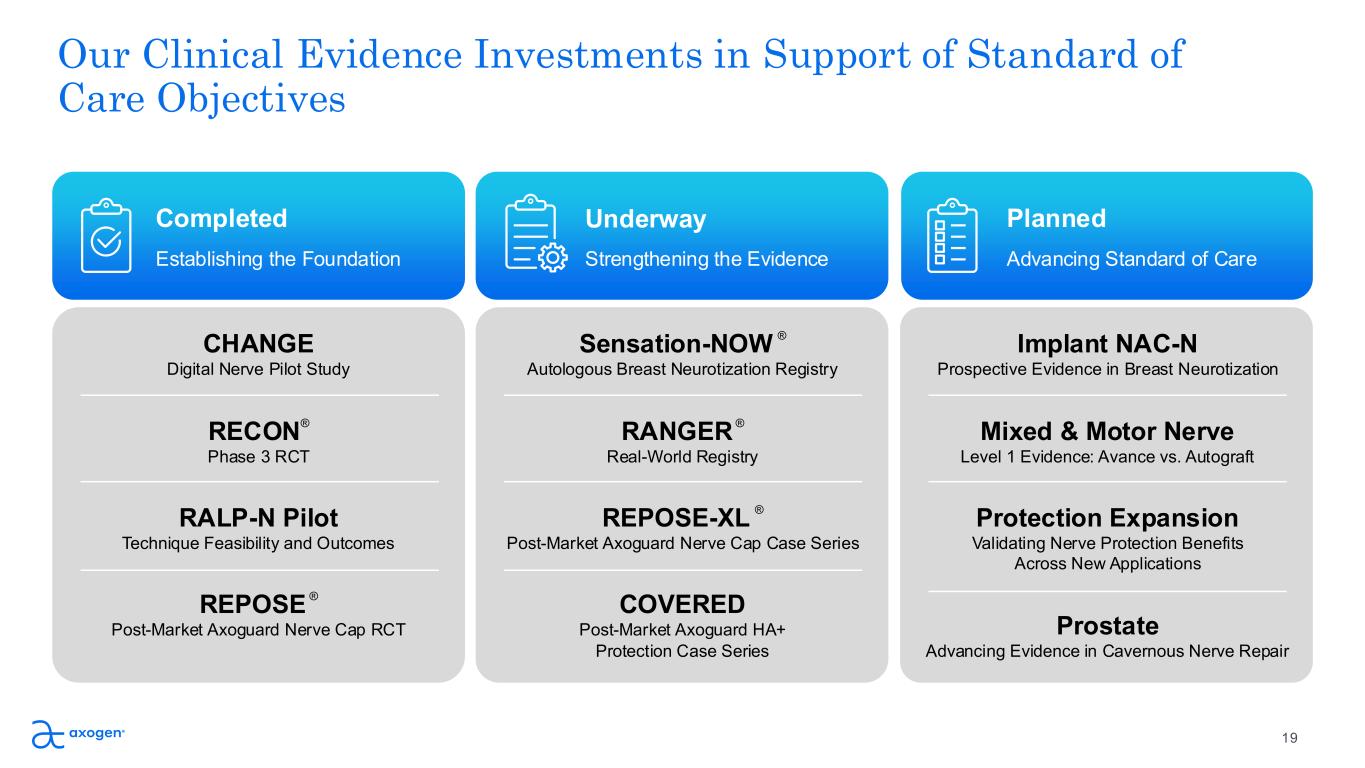
Our Clinical Evidence Investments in Support of Standard of Care Objectives Completed CHANGE Digital Nerve Pilot Study RECON ® Phase 3 RCT RALP-N Pilot Technique Feasibility and Outcomes REPOSE ® Post-Market Axoguard Nerve Cap RCT Underway Planned Establishing the Foundation Strengthening the Evidence Advancing Standard of Care Sensation-NOW ® Autologous Breast Neurotization Registry RANGER ® Real-World Registry REPOSE-XL ® Post-Market Axoguard Nerve Cap Case Series COVERED Post-Market Axoguard HA+ Protection Case Series Implant NAC-N Prospective Evidence in Breast Neurotization Mixed & Motor Nerve Level 1 Evidence: Avance vs. Autograft Protection Expansion Validating Nerve Protection Benefits Across New Applications Prostate Advancing Evidence in Cavernous Nerve Repair 19
Management Team with a Track Record of Success Michael Dale Chief Executive Officer and Board Director Marc Began Executive Vice President and General Counsel Lindsey Hartley Chief Financial Officer Erick DeVinney Chief Innovation Officer Jens Schroeder Kemp Chief Marketing Officer Ivica Ducic, M.D. Chief Medical Officer Craig Swandal Vice President, Operations Stacy Arnold Vice President of Product Development and Clinical Research Al Jacks Vice President of Quality Rick Ditto Vice President, Global Health Economics, Reimbursement & Policy Doris Quackenbush Vice President of Sales Jesse Bishop Vice President, Regulatory Prior Roles Include 20

Financial Overview 21
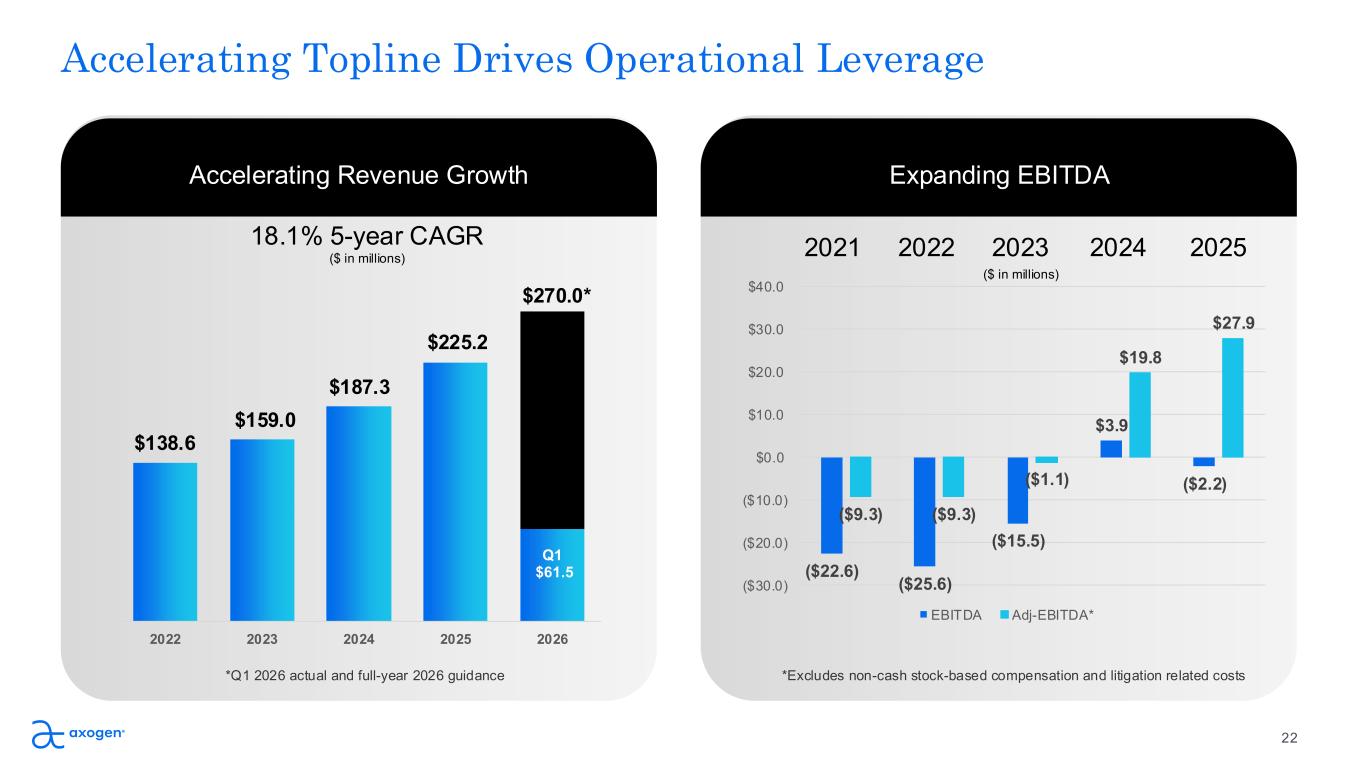
Accelerating Topline Drives Operational Leverage Accelerating Revenue Growth 22 Expanding EBITDA 2022 2023 2024 2025 2026 Q1 $61.5 $138.6 $159.0 $187.3 $225.2 ($22.6) ($25.6) ($15.5) $3.9 ($2.2) ($9.3) ($9.3) ($1.1) $19.8 $27.9 ($30.0) ($20.0) ($10.0) $0.0 $10.0 $20.0 $30.0 $40.0 EBITDA Adj-EBITDA* 2021 2022 2023 2024 2025 *Excludes non-cash stock-based compensation and litigation related costs 18.1% 5-year CAGR ($ in millions) $270.0* *Q1 2026 actual and full-year 2026 guidance ($ in millions)

Revenue growth of at least 20% or $270 million Gross margin of 74% to 76% 23 Free cash flow positive 2026 Guidance

Investment Highlights Big Market Opportunity $5.6B TAM with minimal current penetration 24 Clinical Leadership Unique comprehensive solution with strong evidence Multiple Growth Catalysts Four distinct market opportunities at different stages Reimbursement Tailwinds Expanding coverage and improving payment rates Scalable Infrastructure Proven commercial model ready to capture market share Financial Inflection Point Positive cashflow, expanding margins, accelerating growth

Thank you © 2026 Axogen Corporation. All rights reserved. The stylized "a" logo is a registered trademark of Axogen Corporation.
26